- Primary endpoint
- Enrollment
- Treatment protocol
- Trial collaboration
7-day composite of:
- PE-related mortality
- Cardiorespiratory decompensation or collapse
- Symptomatic PE recurrence
9
Countries
59
Enrollment Sites
544
Patients
Eligibility criteria:
Intermediate-risk PE with elevated risk of early death or hemodynamic collapse.
- Infusion time: 7 hours
- Unilateral 9mg ƚPA (2mg bolus + 1mg/hr)
- Bilateral: 18mg ƚPA (2mg bolus + 1mg/hr)
- Standardized anticoagulation
Trial Collaborators
HI-PEITHO is a joint research study led by Boston Scientific, in partnership with The PERT Consortium and University Medical Center Mainz, to establish advanced therapy with EKOS as a first line treatment for intermediate-risk PE patients.
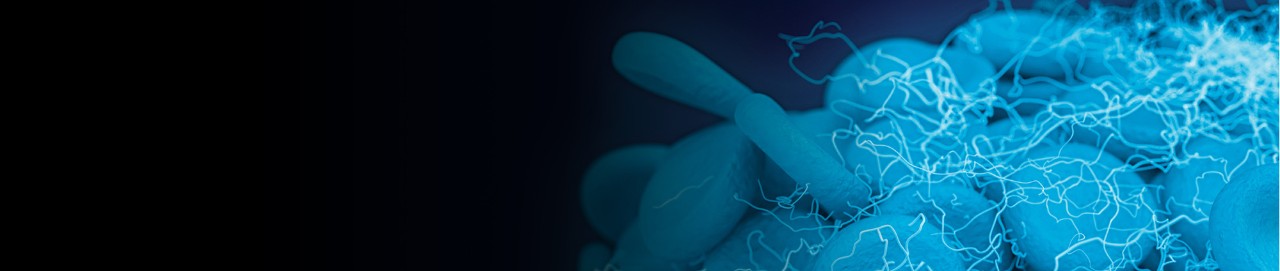
EKOS demonstrated superiority to AC alone for patients with intermediate-risk PE
Primary endpoint
EKOS demonstrated superiority to AC alone, resulting in a significantly lower rate of the primary composite endpoint (4.0% vs. 10.3%; p=0.005).
Composite of PE-related mortality, cardiorespiratory decompensation or collapse, and symptomatic PE recurrence at 7 days
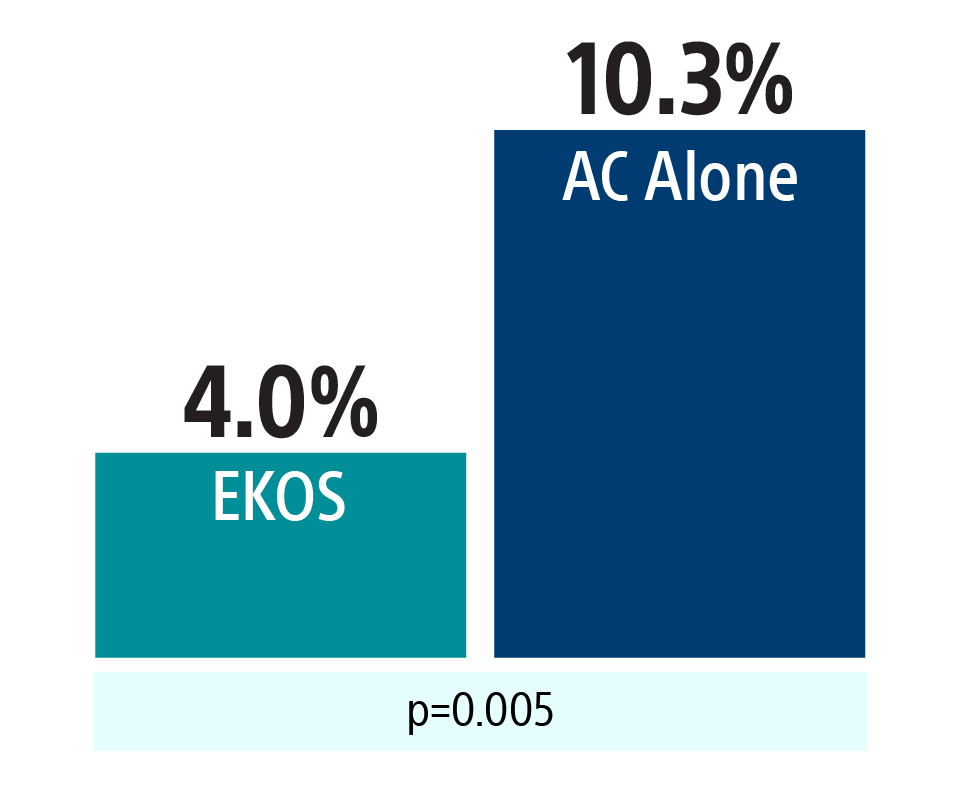
Primary endpoint components
64% reduction in cardiorespiratory decompensation or collapse with EKOS
Other primary endpoint components: PE-related mortality (EKOS 1.1%, AC alone 0.4%), symptomatic PE recurrence (EKOS 0.4%, AC alone 0.4%)
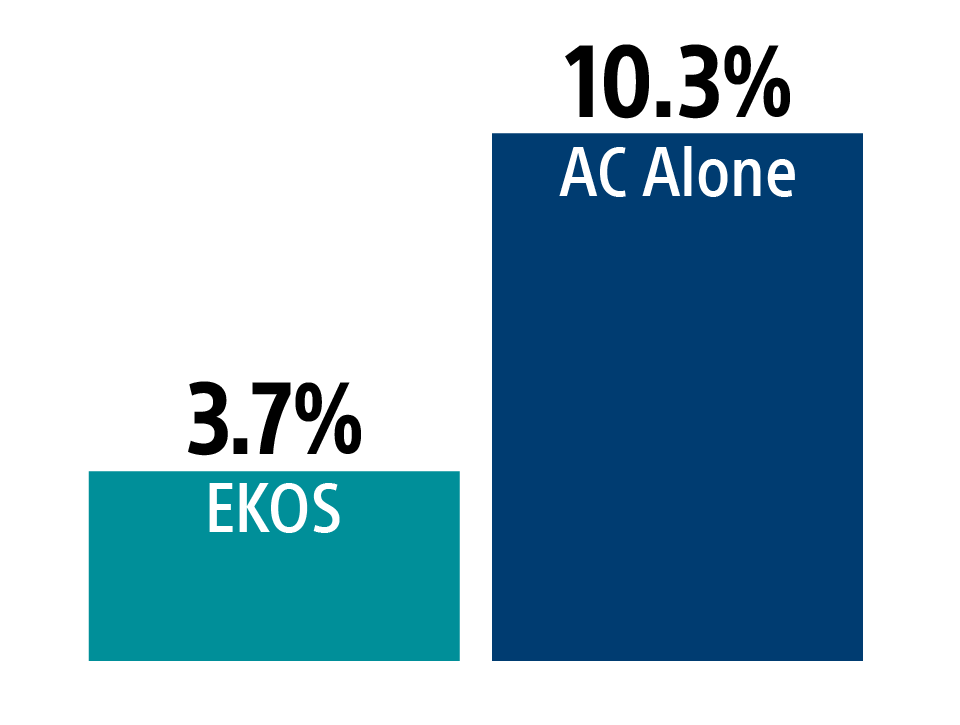
Major bleeding at 7 days
ISTH

There was no significant difference in major bleeding between treatment arms
Safety outcomes
There was no significant difference in major bleeding between treatment arms and zero ICH events.
Major bleeding at 30 days
ISTH1
4.1%
EKOS
n=271
3.0%
AC Alone
n=271
0
ICH Events
In either study arm
p=0.64
Hospital length of stay
Total hospital length of stay was shorter for patients treated with EKOS.
Hospital total length of stay
(Mean Days)
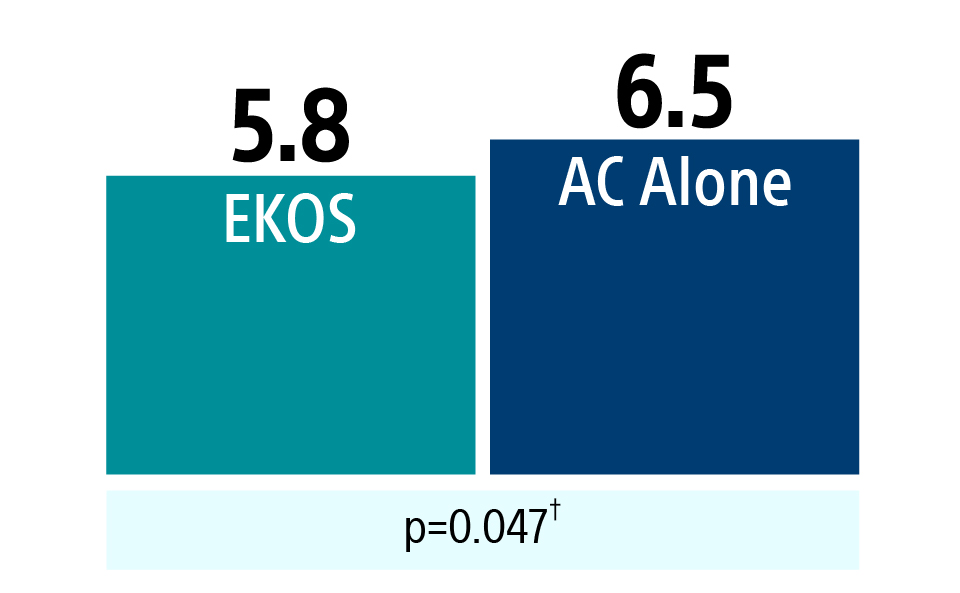
† One-sided t-test
NEWS score
HI-PEITHO is the first PE randomized controlled trial to include NEWS in the trial protocol.
The National Early Warning Score (NEWS) is a standardized and easy-to-use tool that determines the degree of illness and mortality risk of a patient and can be used to prompt critical care intervention.
NEWS provides a scoring system for physiological measurements that are routinely recorded at a patient’s bedside. A score ranging from 0-3 is allocated to each parameter, with higher scores indicating the parameter is further from the normal range. The scores for each parameter are then added together to determine the NEWS Score.
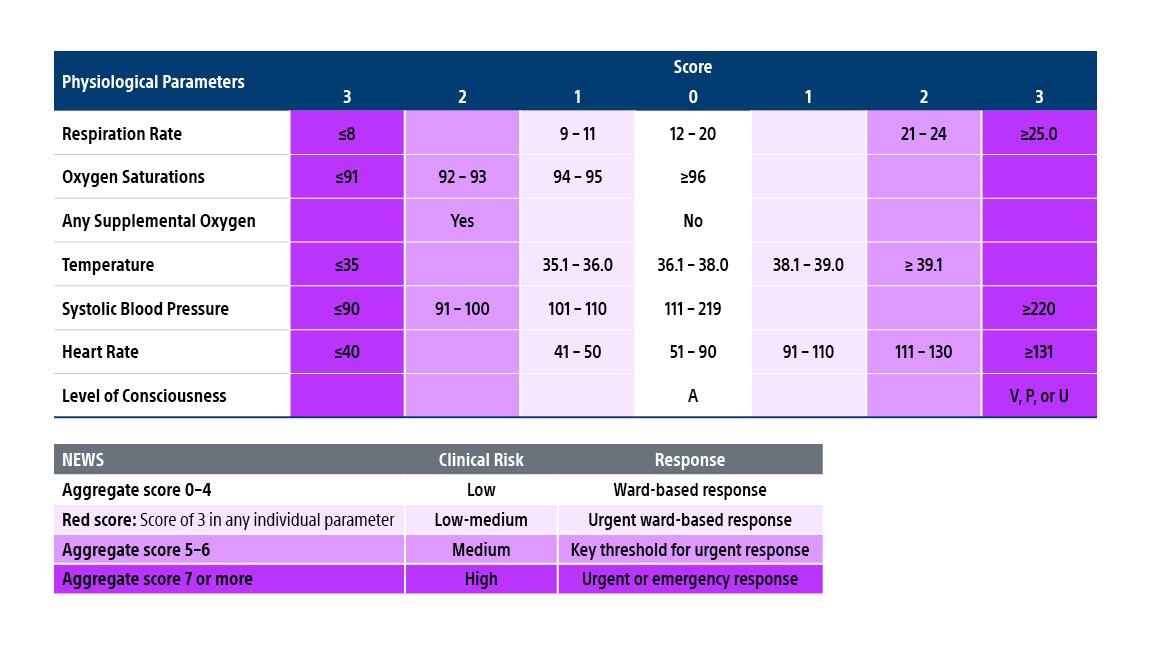
Reproduced from: Royal College of Physicians. National Early Warning Score (NEWS): Standardising the assessment of acute-illness severity in the NHS. Report of a working party. London: RCP, 2012
1. ISTH: International Society on Thrombosis and Haemostasis