Boston Scientific accounts are for healthcare professionals only.
Boston Scientific accounts are for healthcare professionals only.
Create an account to access online training and education on EDUCARE, manage your customer profile, and connect with customer support and service teams.
My Boston Scientific account
Access your online applications and manage your customer profile.
Quick Links
Call customer care

Download the latest bench test comparing the rheological properties of SpaceOAR Vue™ and Barrigel® hydrogels. This bench test reveals how each hydrogel responds to stress. In this bench test performed by Boston Scientific, Barrigel showed a greater variability in volume change compared to SpaceOAR Vue Hydrogel at lower pressures (below 80 mmHg) and at higher pressures (above 80 mmHg).
Whether you’re looking to get started with SpaceOAR Hydrogel or have general questions about the product, procedure, or clinical data, our team is here to help.
When you choose SpaceOAR Hydrogel for your prostate cancer patients, you’re not only getting a device with proven clinical outcomes — you’re choosing a world-class customer experience.

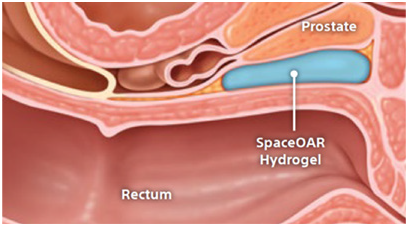
For prostate cancer patients undergoing radiation therapy, maintaining quality of life may be just as important as treating the cancer.
*Of men who had erections sufficient for intercourse at baseline.
†Number of patients is based on units shipped and a BSC proprietary algorithm.
‡Data based on sales for SpaceOAR™ Hydrogel or SpaceOAR Vue™ Hydrogel to institutions in 2022 and 2023
**CPT copyright © 2023 American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association.
References
Caution: U.S. Federal law restricts this device to sale by or on the order of a physician.
Health economic and reimbursement information provided by Boston Scientific Corporation is gathered from third-party sources and is subject to change without notice as a result of complex and frequently changing laws, regulations, rules, and policies.
This information is presented for illustrative purposes only and does not constitute reimbursement or legal advice.
Boston Scientific encourages providers to submit accurate and appropriate claims for services. It is always the provider’s responsibility to determine medical necessity, the proper site for delivery of any services, and to submit appropriate codes, charges, and modifiers for services rendered. It is also always the provider’s responsibility to understand and comply with Medicare national coverage determinations (NCD), Medicare local coverage determinations (LCD), and any other coverage requirements established by relevant payers which can be updated frequently. Boston Scientific recommends that you consult with your payers, reimbursement specialists, and/or legal counsel regarding coding, coverage, and reimbursement matters.
Boston Scientific does not promote the use of its products outside their FDA-approved label.
Payer policies will vary and should be verified prior to treatment for limitations on diagnosis, coding, or site of service requirements.
CPT Copyright © 2023 American Medical Association. All rights reserved. CPT is a registered trademark of the American Medical Association. Applicable FARS/DFARS Restrictions Apply to Government Use. Fee schedules, relative value units, conversion factors and/or related components are not assigned by the AMA, are not part of CPT, and the AMA is not recommending their use. The AMA does not directly or indirectly practice medicine or dispense medical services. The AMA assumes no liability for data contained or not contained herein.” All trademarks are the property of their respective owners.
All images are the property of Boston Scientific. All trademarks are the property of their respective owners.