TARGET study overview
A global evaluation of advanced dosimetry in transarterial radioembolization of hepatocellular carcinoma with Yttrium-90: the TARGET study.
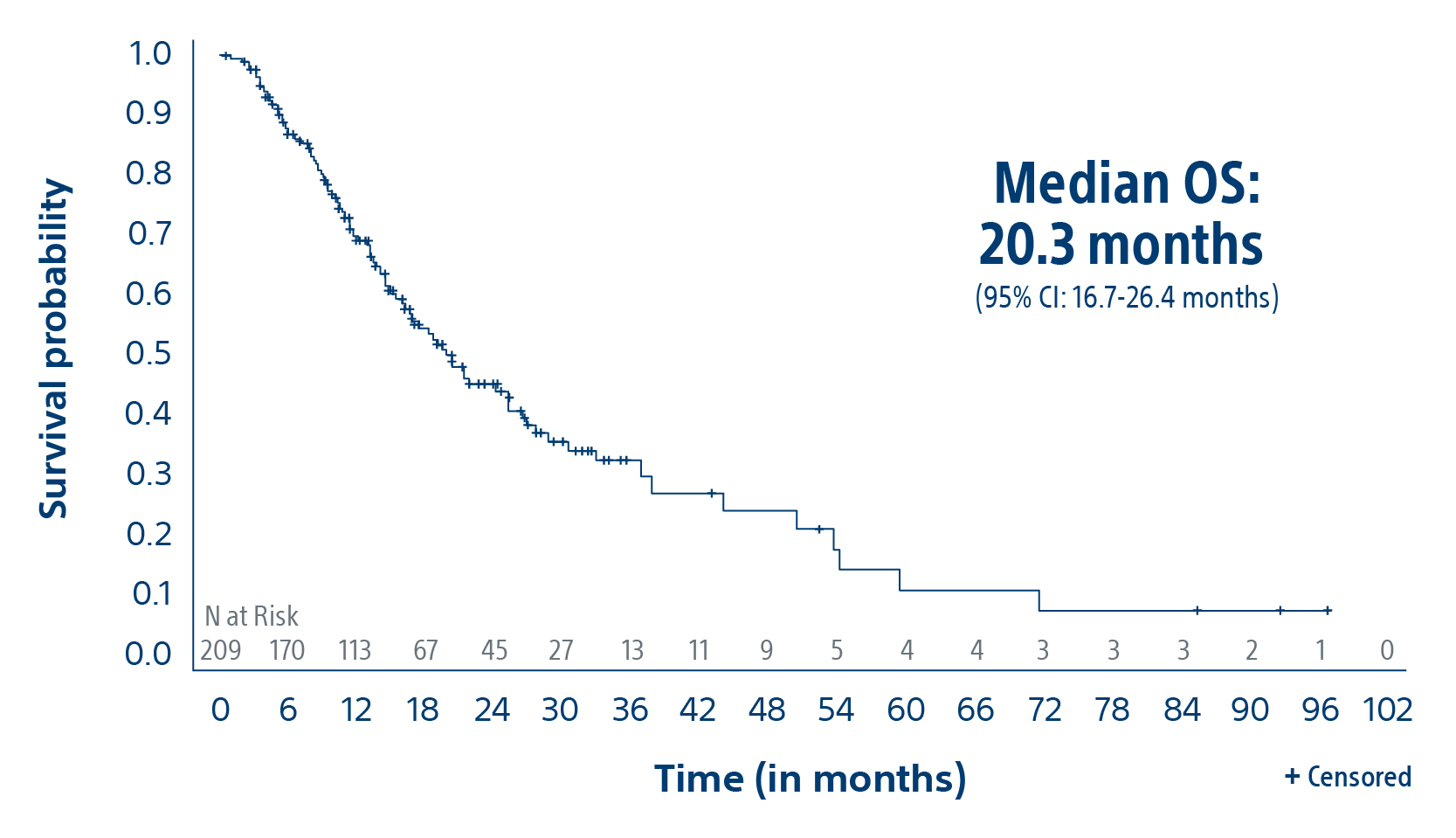
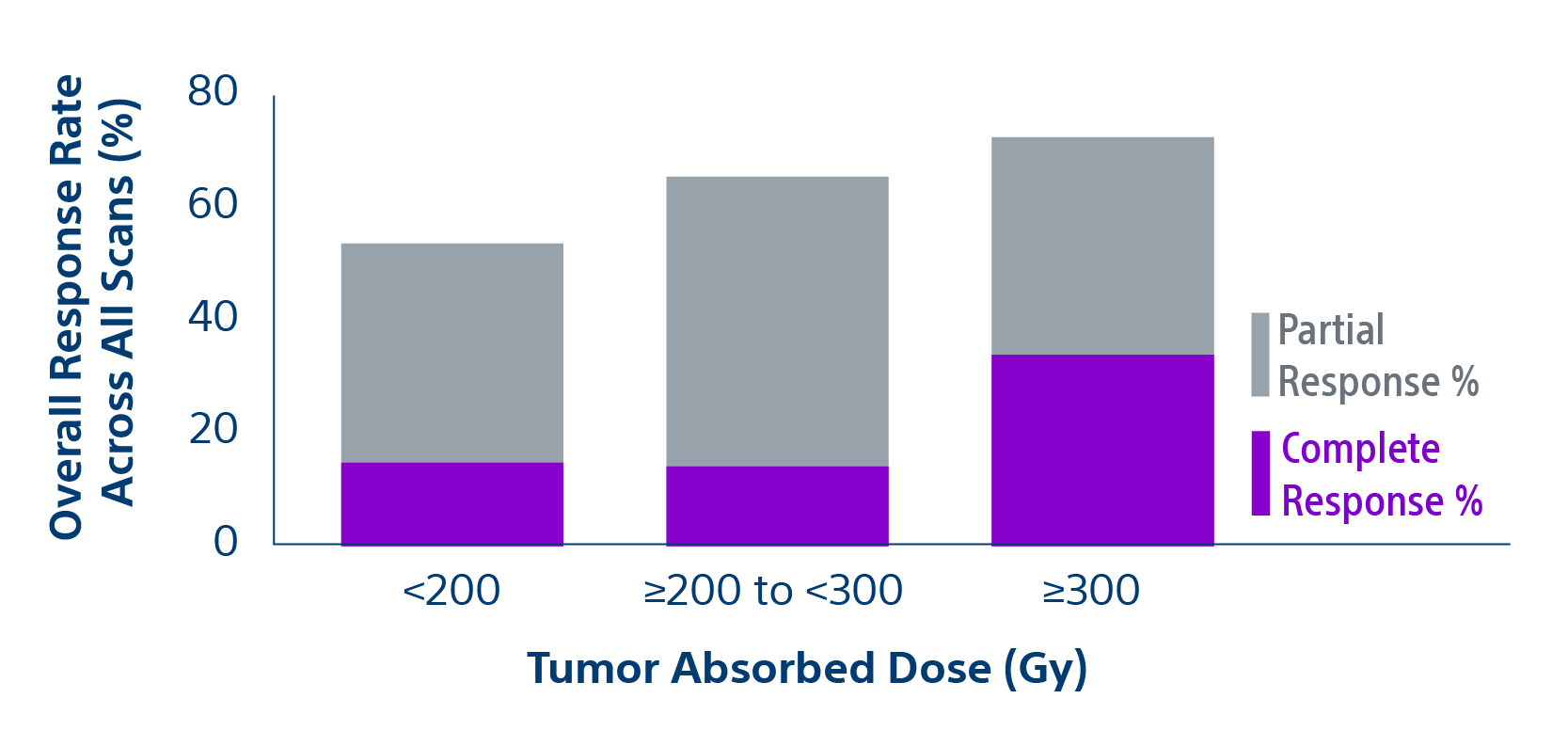
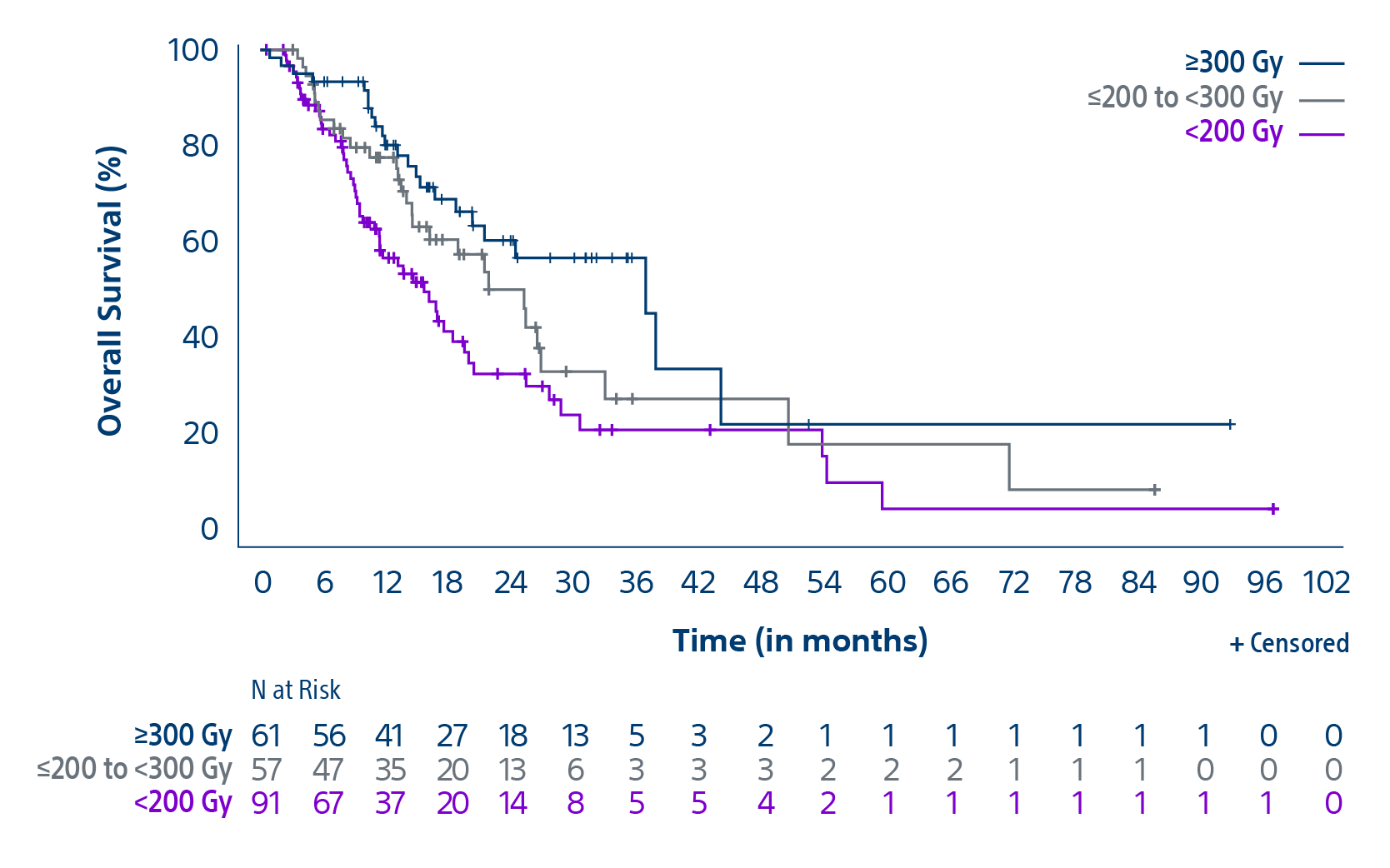
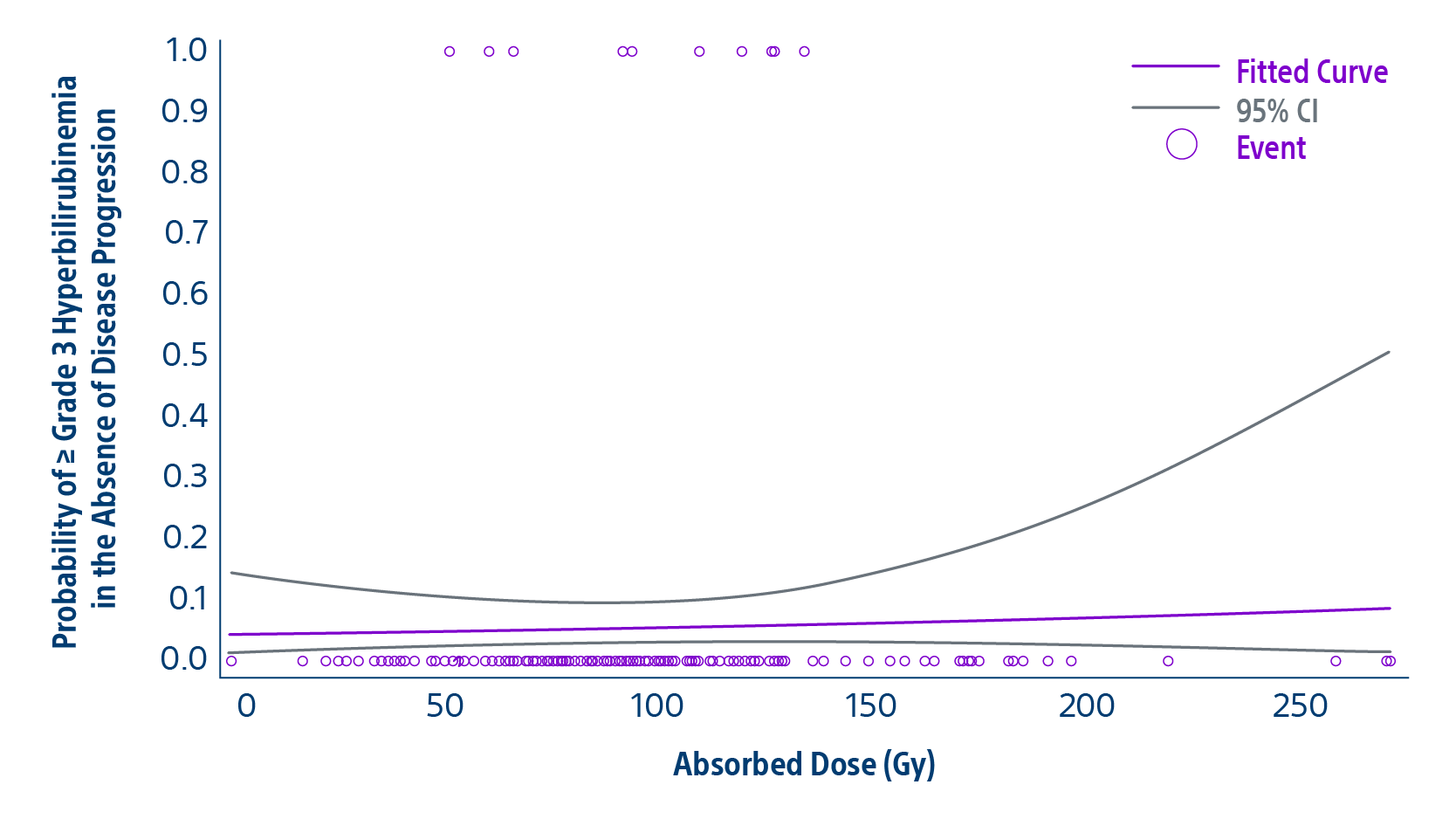
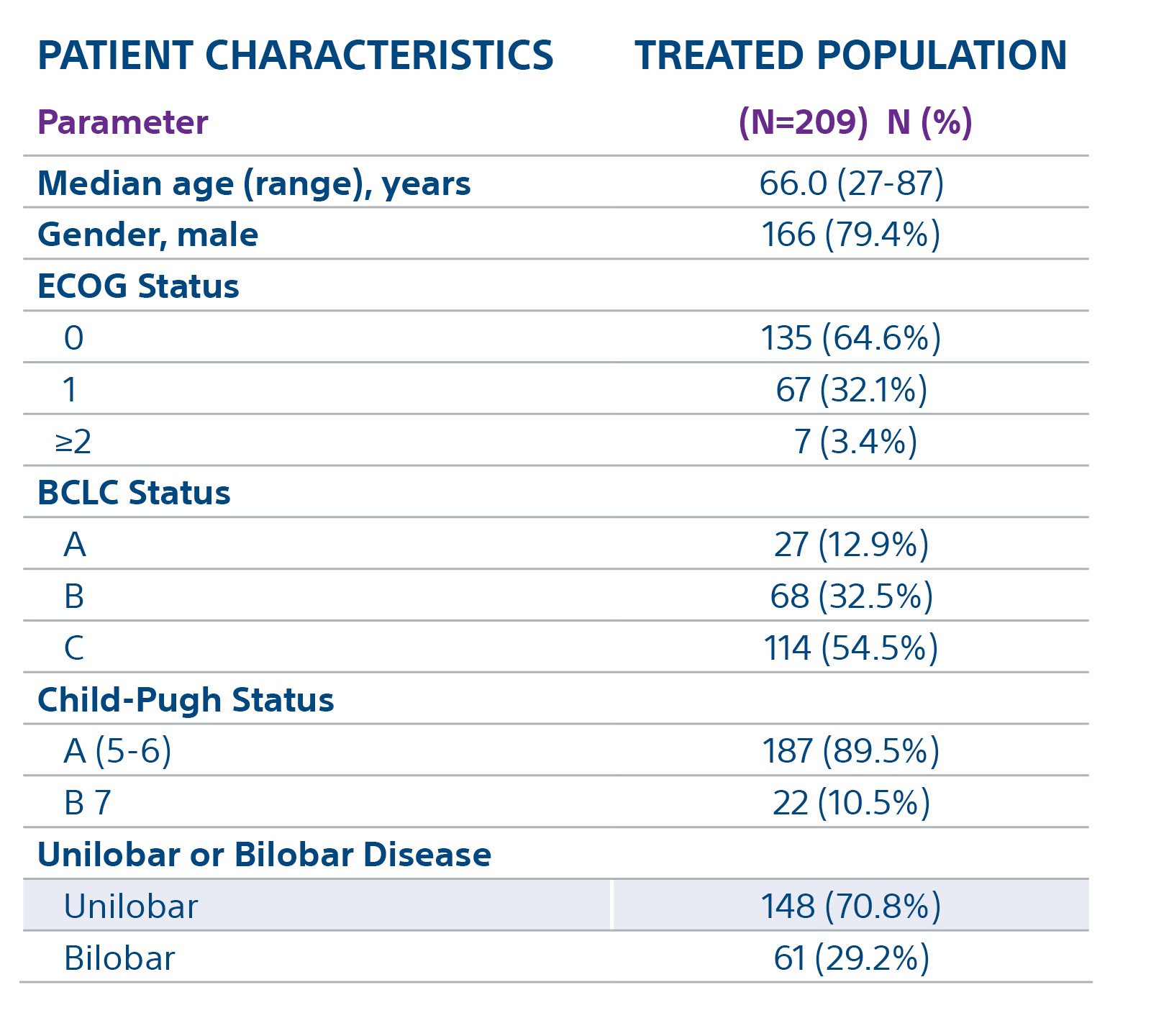
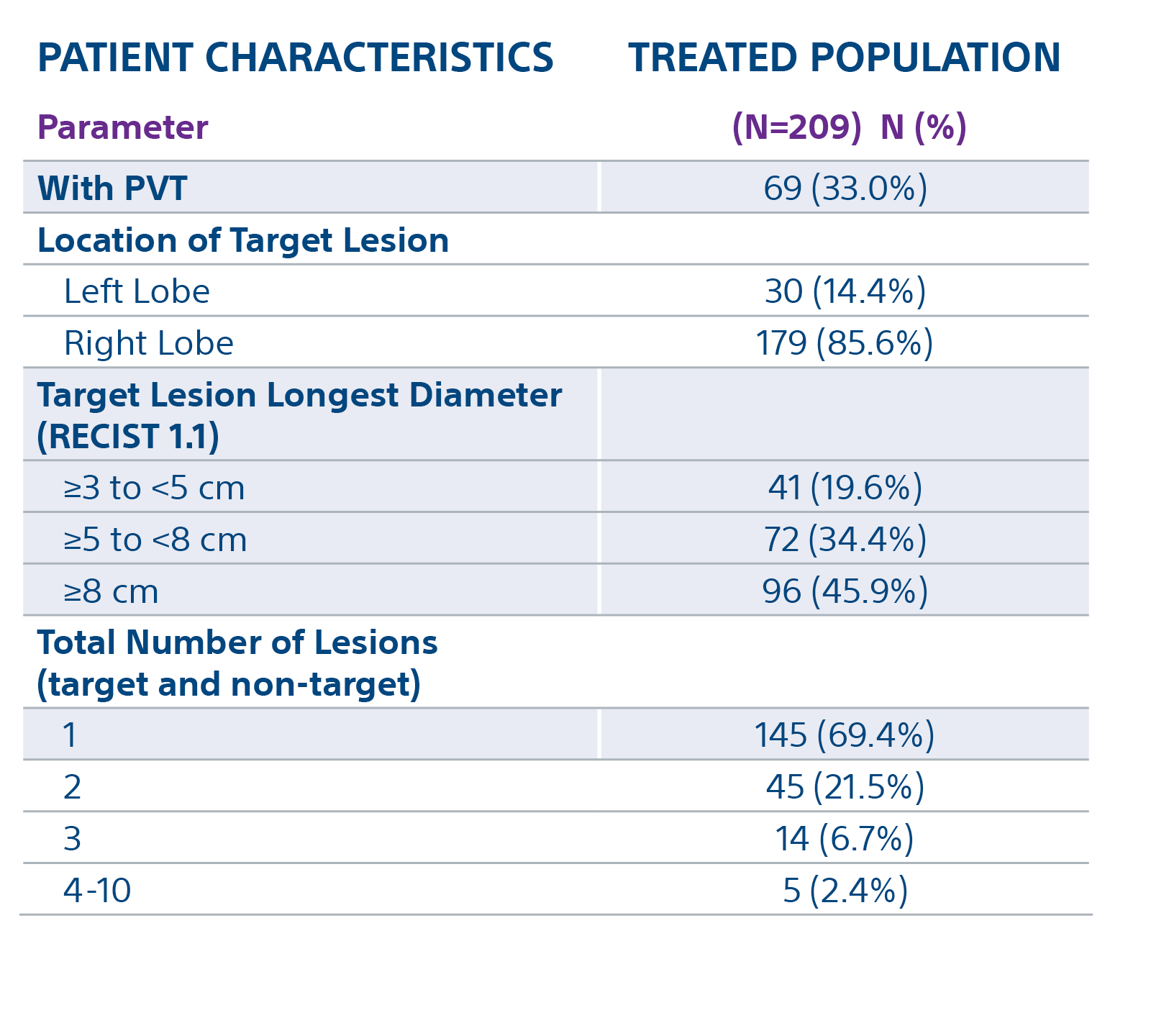
The TARGET study confirms TheraSphere treatment for HCC as safe and effective in select patient populations. Predictable clinical outcomes from this real-world study span a broad group across 8 countries.