Boston Scientific accounts are for healthcare professionals only.

Express™ SD Renal and Biliary Premounted Stent System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical Data
- Training
- Resources
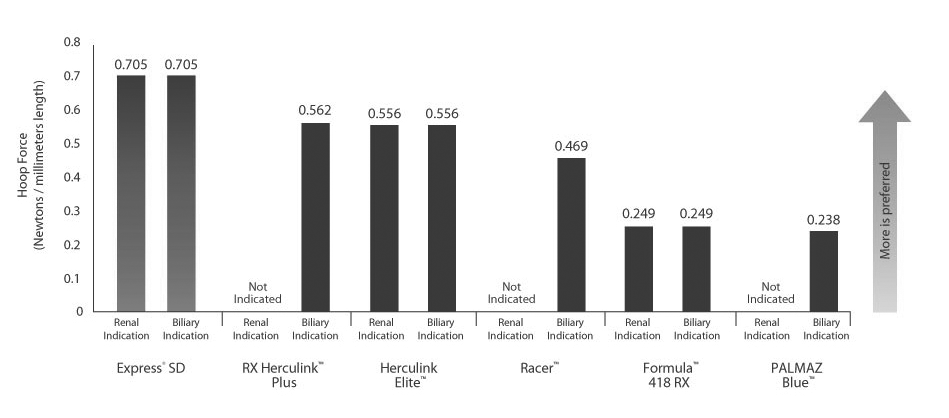
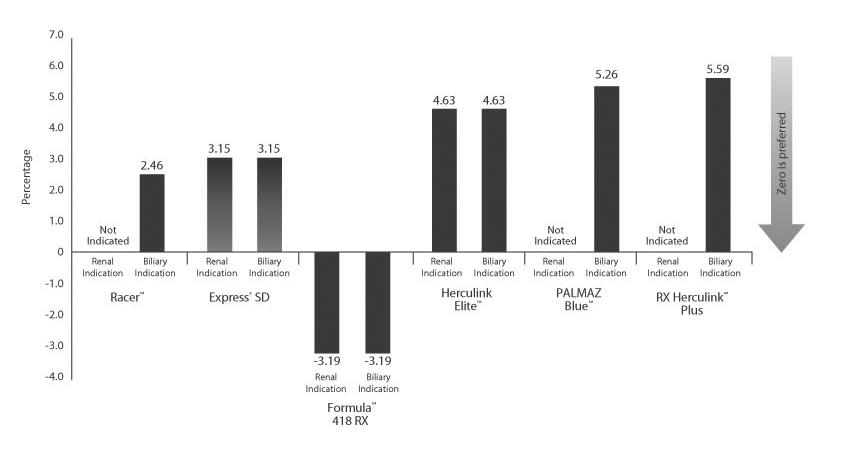
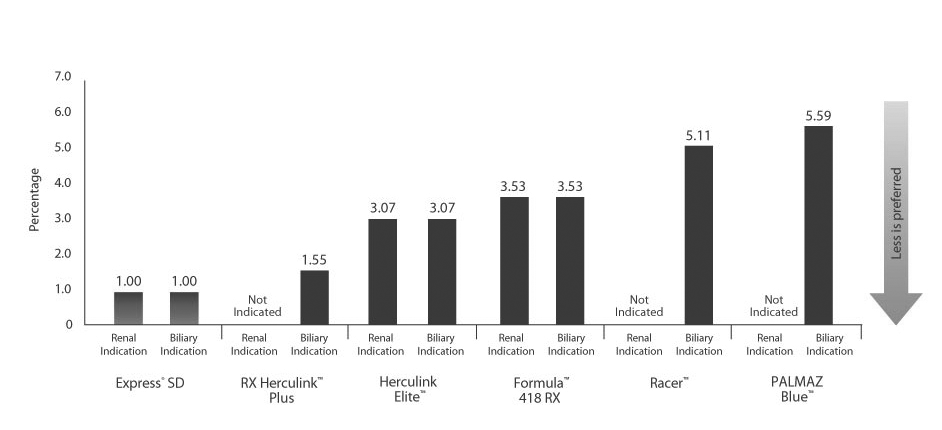
Excellence in compression resistance
The Express SD Stent couples excellent compression resistance with lower stent foreshortening and recoil. Only Express SD has the ideal balance of strength and precision for acute success.

How it works
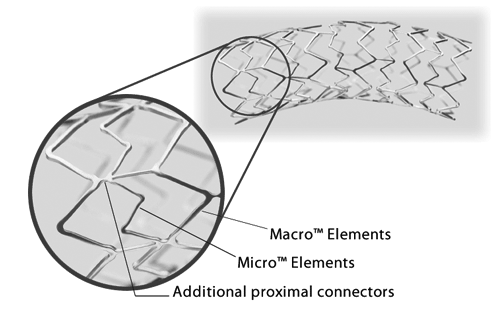
Why choose Express SD
Featured clinical data
RENAISSANCE Clinical Trial
Overview
The RENAISSANCE Trial was a prospective, multi-center, single arm study designed to evaluate the Express SD Renal Premounted Stent System in the treatment of atherosclerotic lesions in the aortorenal ostium.
The primary objective was to demonstrate superiority of the duplex triggered, angiographically-confirmed binary restenosis rate at 9-months over the 40% pre-specified Objective Performance Criterion (OPC) representative of PTRA.
Trial Design
The RENAISSANCE Trial enrolled 100 patients (117 lesions) with de novo or restenotic ostial atherosclerotic lesions ≤15mm long in vessels between 4.0 and 7.0mm diameter with diameter stenosis ≥70%.
Outcomes
Primary Endpoint: 21.3% binary in-stent restenosis rate at 9-months, defined as the proportion of target lesions with ≥50% diameter stenosis based on angiographic core lab assessment.
5-Year Data
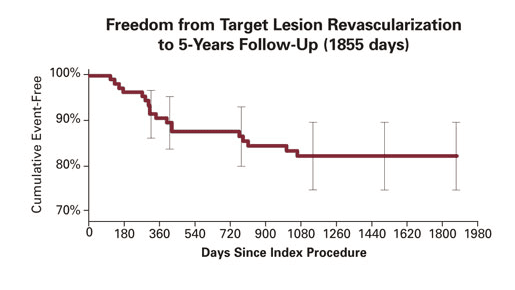
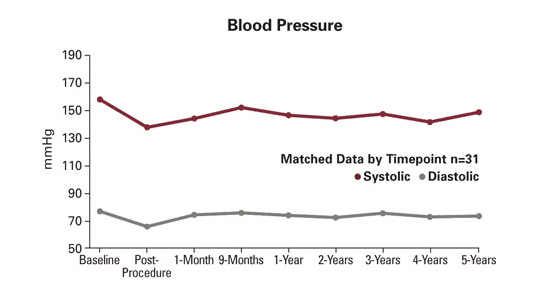
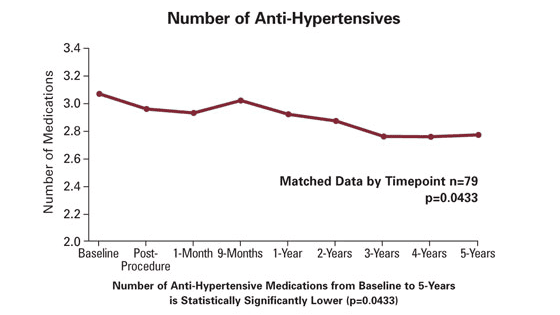
Additional results
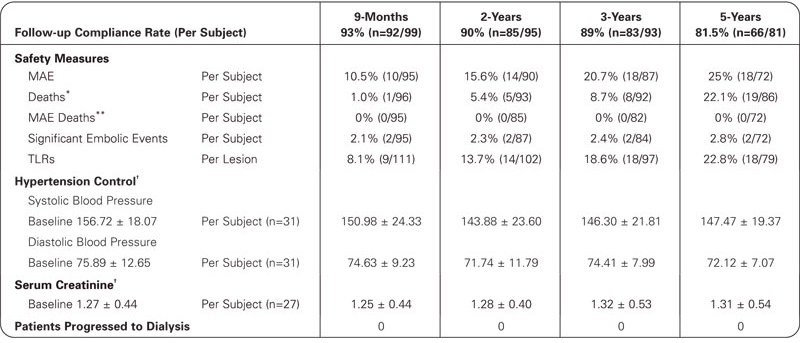
References:
*Denominator consists of number of subjects who either died prior to late follow-up window and/or had follow-up to at least the early follow-up window.
**Denominator consists of number of subjects who either had CEC adjudicated device or procedure related death prior to late follow-up window and/or had follow-up to at least the early follow-up window.
†Matched data by time point.
For additional information, please see Rocha-Singh K, Jaff M, Kelley E.L. Renal Artery Stenting with Non-Invasive Duplex Ultrasound Follow-up: 3-Year Results from the RENAISSANCE Renal Stent Trial. Catheterization and Cardiovascular Interventions 2008; 72:853-862.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Resources list
Express is a registered or unregistered trademark of Boston Scientific Corporation or its affiliates. All other trademarks are the property of their respective owners.