SYNERGY™
Everolimus-Eluting Platinum Chromium Coronary Stent System
SENIOR Trial Safety Data with SYNERGY BP-DES in 1-Month DAPT* Cohort at 2-Years1
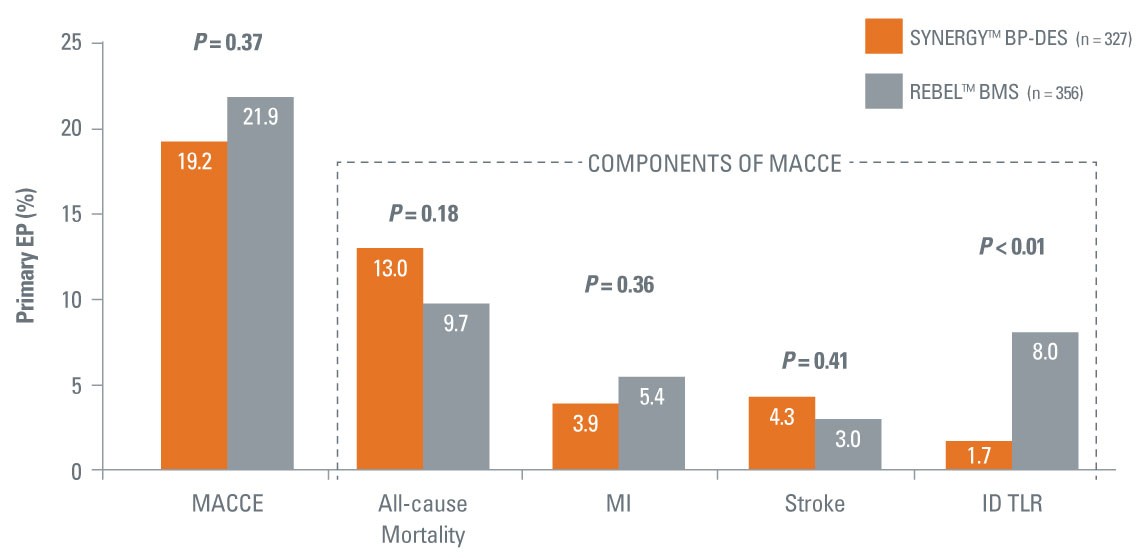
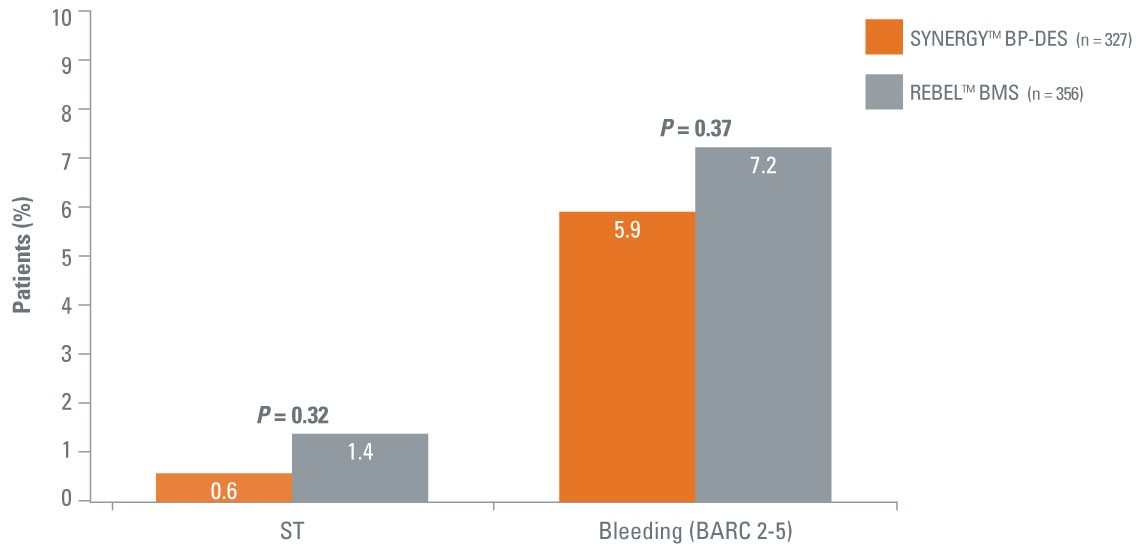
- Randomized, single-blind trial evaluating the SYNERGY BP-DES and the REBEL™ BMS in patients 75 years or older with a short duration of DAPT
- Over 50% of patients enrolled received a 1-month DAPT regimen – largely stable patients
- Statistical significant 4.5x reduction in ID-TLR with SYNERGY BP-DES vs. best-in-class REBEL™ BMS
- 0.6% ST with SYNERGY BP-DES
**All-cause mortality, MI, stroke, ischemia-driven TLR
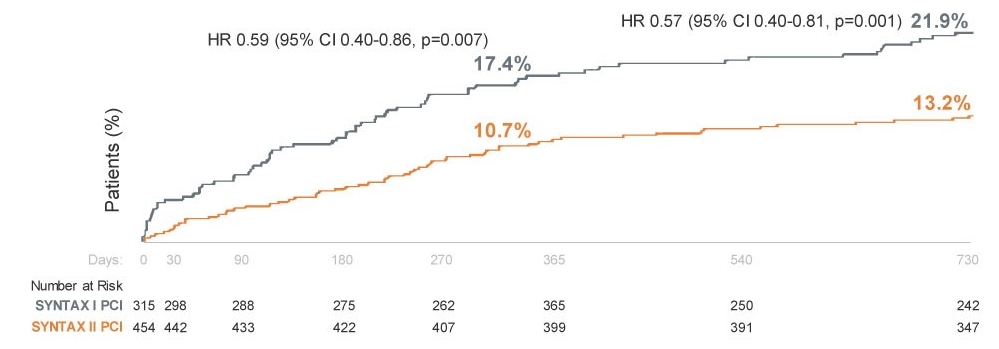
SYNTAX II Trial 2-Year Results2‡
- Prospective, single-arm, multicenter, open-label trial of patients with de novo three-vessel CAD
- The MACCE rate (primary endpoint) was significantly lower in SYNTAX II (13.2%) vs SYNTAX I (21.9%). (P=0.001)
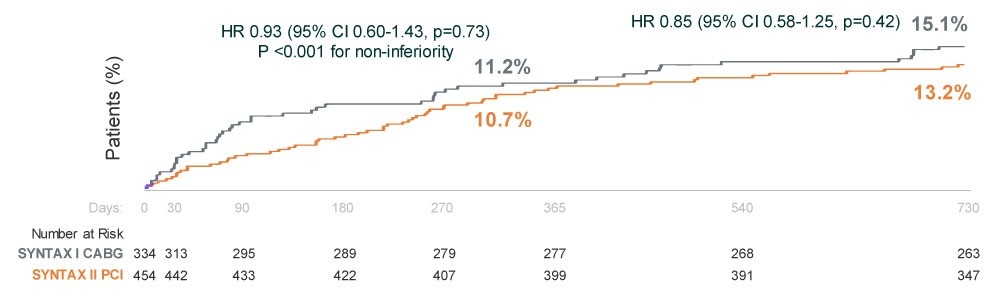
- SYNERGY BP-DES, physiological assessment, IVUS guidance, and contemporary PCI techniques demonstrated equivalent outcomes to CABG in patients with three-vessel disease.† (MACCE rate of 13.2% PCI vs 15.1% CABG, P<0.001 for non-inferiority)
MACCE vs. Historical PCI - 2 Year Results
MACCE PCI vs. Historical CABG - 2 Year Results
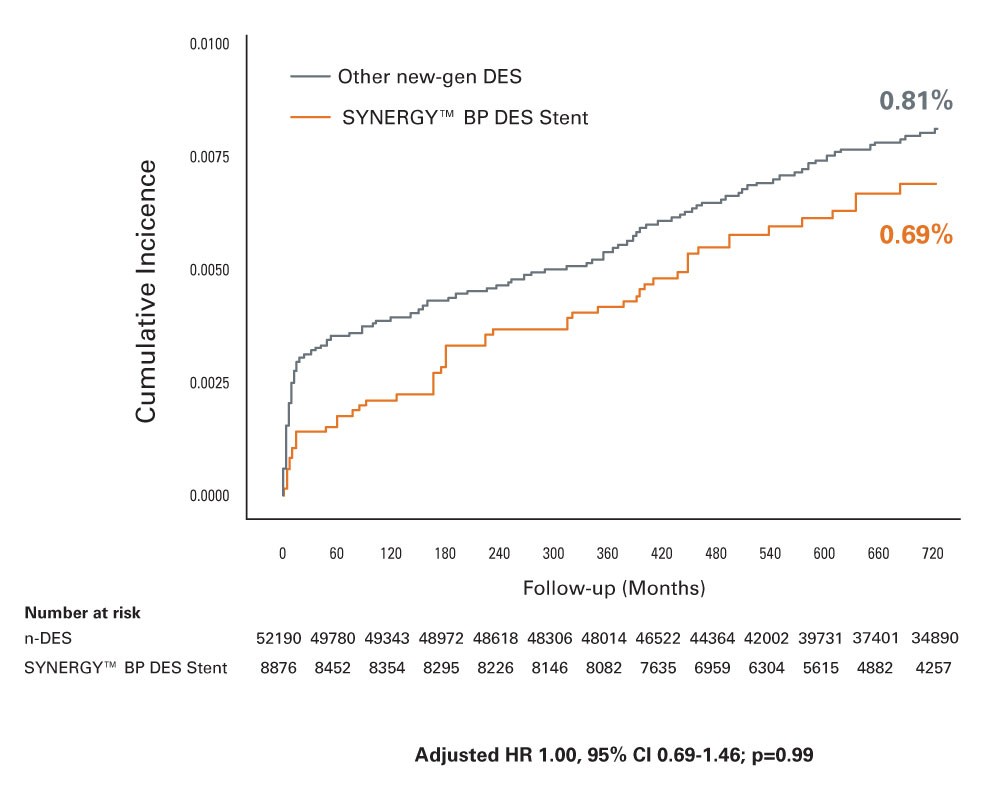
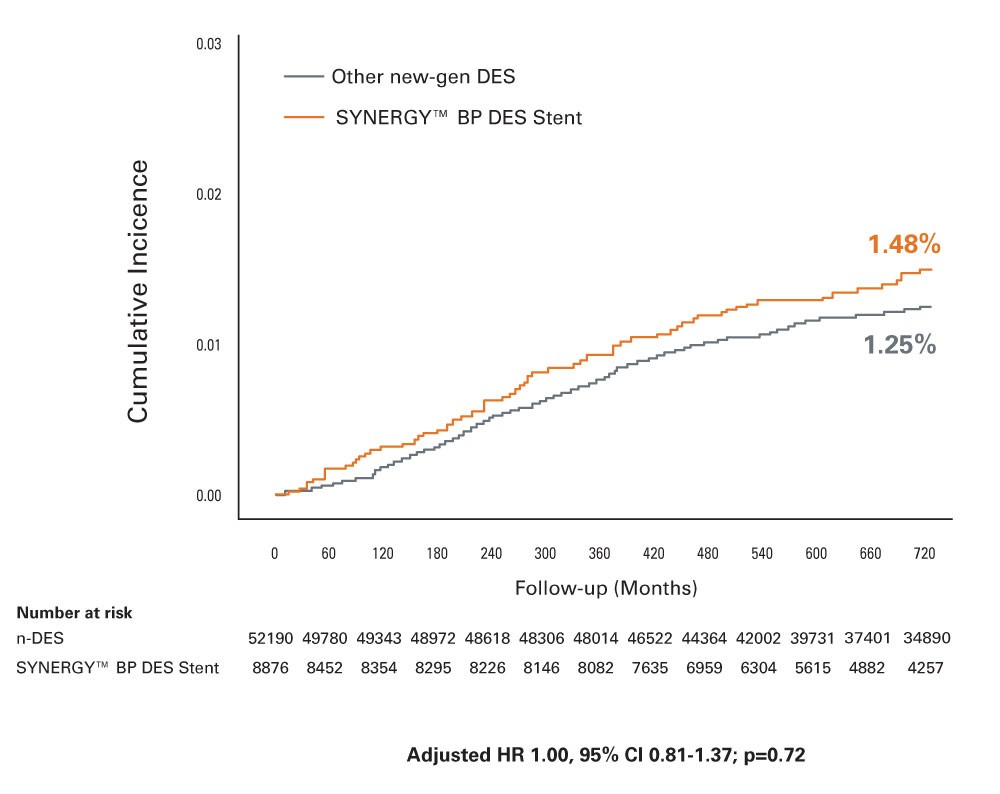
SCAAR AMI Substudy 2-Year Results3‡
- Evaluated SYNERGY BP-DES compared to all other new-generation DES in the SCAAR Registry in 36,292 patients with AMI at 2-Years
- Lowest ST rate with SYNERGY BP-DES in AMI patients despite use in more complex patients and lesions
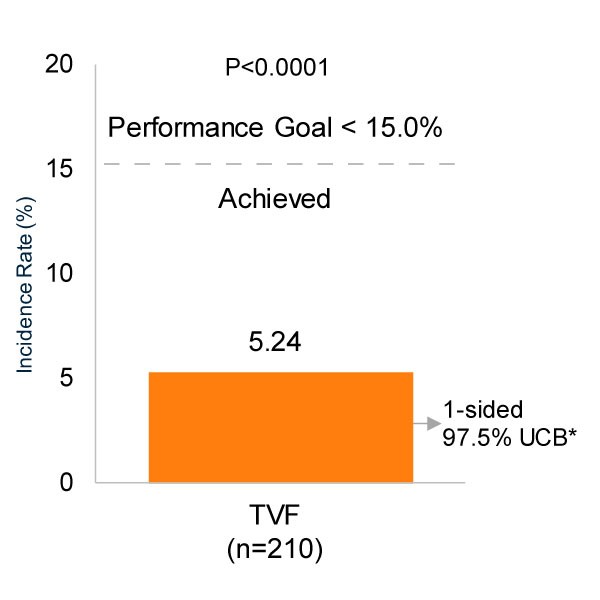
CONSISTENT CTO Study 12-Month Results4‡
- Single-arm, 6 center study of 210 CTO patients evaluating primary end-point of TVF versus performance goal with the SYNERGY BP-DES
- Low event rates in complex lesions (mean stent length 85.6 mm) and very symptomatic patients
Key Trial Details4
- 90.5% of patients had pre-PCI IVUS
- 90% CTO success rate
- Very complex and symptomatic patients
- JCTO Score of 2.4
- Mean stent length 85.6 mm
- 21% diabetic patients
- Significant QoL gains at 12-months following successful CTO PCI
CELTIC Bifurcation Study 9-Month Results5‡
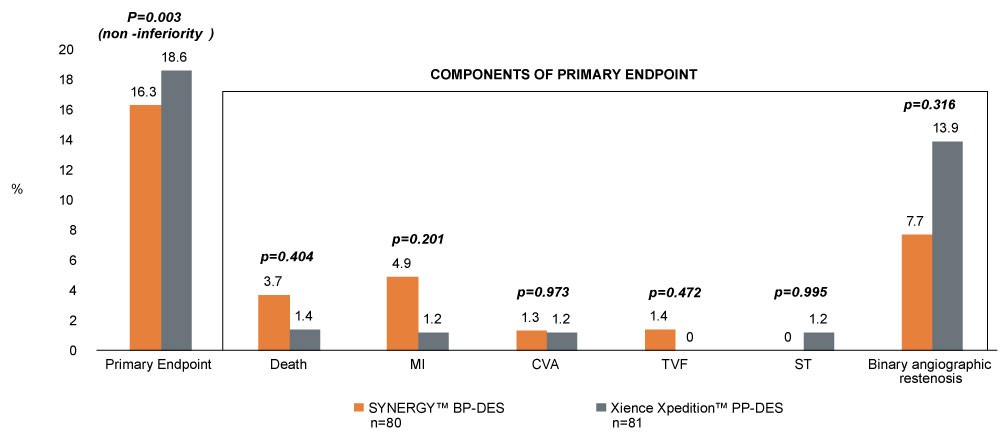
- Examined outcomes of 170 patients at 8 sites in the UK treated by planned culotte stenting with the SYNERGY BP-DES and the Xience Xpedition™ PP-DES
- Results showed lower composite primary endpoint, including 0% ST and low restenosis with the SYNERGY BP-DES
Composite Primary Endpoint at 9-Months