ADVANTAGE AF trial overview
The ADVANTAGE AF US IDE Phase I clinical trial1 studied the safety and effectiveness of the FARAWAVE™ Pulsed Field Ablation (PFA) Catheter in patients with persistent atrial fibrillation (PersAF), combining pulmonary vein isolation (PVI) and posterior wall ablation (PWA).
The ADVANTAGE AF US IDE Phase II clinical trial2 included the addition of cavo-tricuspid isthmus (CTI) isolation with the FARAPOINT™ PFA Catheter and continuous monitoring with the LUX-Dx™ Insertable Cardiac Monitor System (ICM) for continuous heart rhythm monitoring.
The ADVANTAGE AF CTI sub-analysis3 compared the safety and effectiveness of the FARAPOINT™ PFA Catheter (Phase II) to radiofrequency ablation (RFA) (Phase I)—the standard of care—for adjunctive CTI ablation in PersAF patients.
- ADVANTAGE AF US IDE Phase I
- ADVANTAGE AF US IDE Phase II
- ADVANTAGE CTI sub-analysis
About the study: ADVANTAGE AF US IDE Phase I
The ADVANTAGE AF US IDE Phase I clinical trial1 studied the safety and effectiveness of the FARAWAVE™ Pulsed Field Ablation (PFA) Catheter in patients with persistent atrial fibrillation (PersAF), combining pulmonary vein isolation (PVI) and posterior wall ablation (PWA).
ADVANTAGE AF USE IDE Phase I primary endpoints met
ADVANTAGE AF US IDE clinical trial discussion
Hear Dr. Vivek Reddy (Principal Investigator) discuss ADVANTAGE AF Phase I results with Dr. Brad Sutton (Chief Medical Officer for AF Solutions at Boston Scientific).
Key findings: ADVANTAGE AF US IDE Phase I
2.3%
[5.1% UCL] Primary safety event rate at 12 months, which met the 12.0% performance goal
63.5%
[57.3% LCL] Primary effectiveness rate at 12 months, which met 40.0% acceptance criteria
4.6%
Left artial (LA) re-ablation rate, with 84.4% pulmonary vein (PV) durability and 68.8% pulmonary wall (PW) durability
85.3%
Patients free from documented symptomatic AA recurrence at 12 months
Study design and methods: ADVANTAGE AF US IDE Phase I
- 43 US/OUS sites
- 87 investigators (67% of the investigators had no prior FARAWAVE experience)
- 260 PersAF patients underwent PVI and PWA with post-blanking period monitoring of 2x monthly TTM, symptomatic and 24-hour Holter monitoring at 6 and 12 months, and 12-lead ECGs at 3 and 12 months
Primary endpoints met: ADVANTAGE AF US IDE Phase I
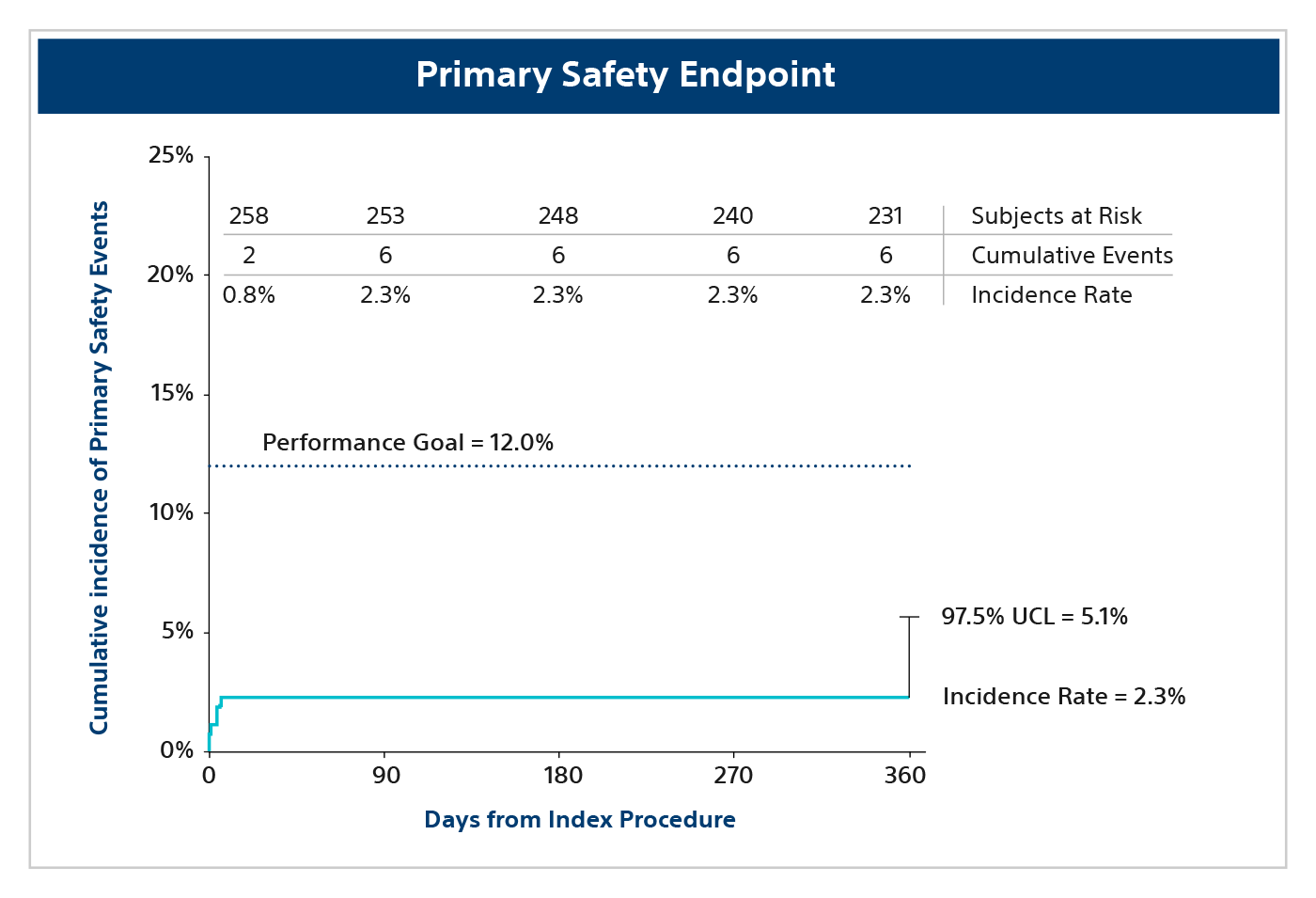
Primary safety endpoint (Phase I)
A composite endpoint defined as serious adverse events related to either the use of an ablation catheter or the ablation procedure with onset within 7 days of the primary procedure and pulmonary vein stenosis or atrio-esophageal fistula out to 12 months.
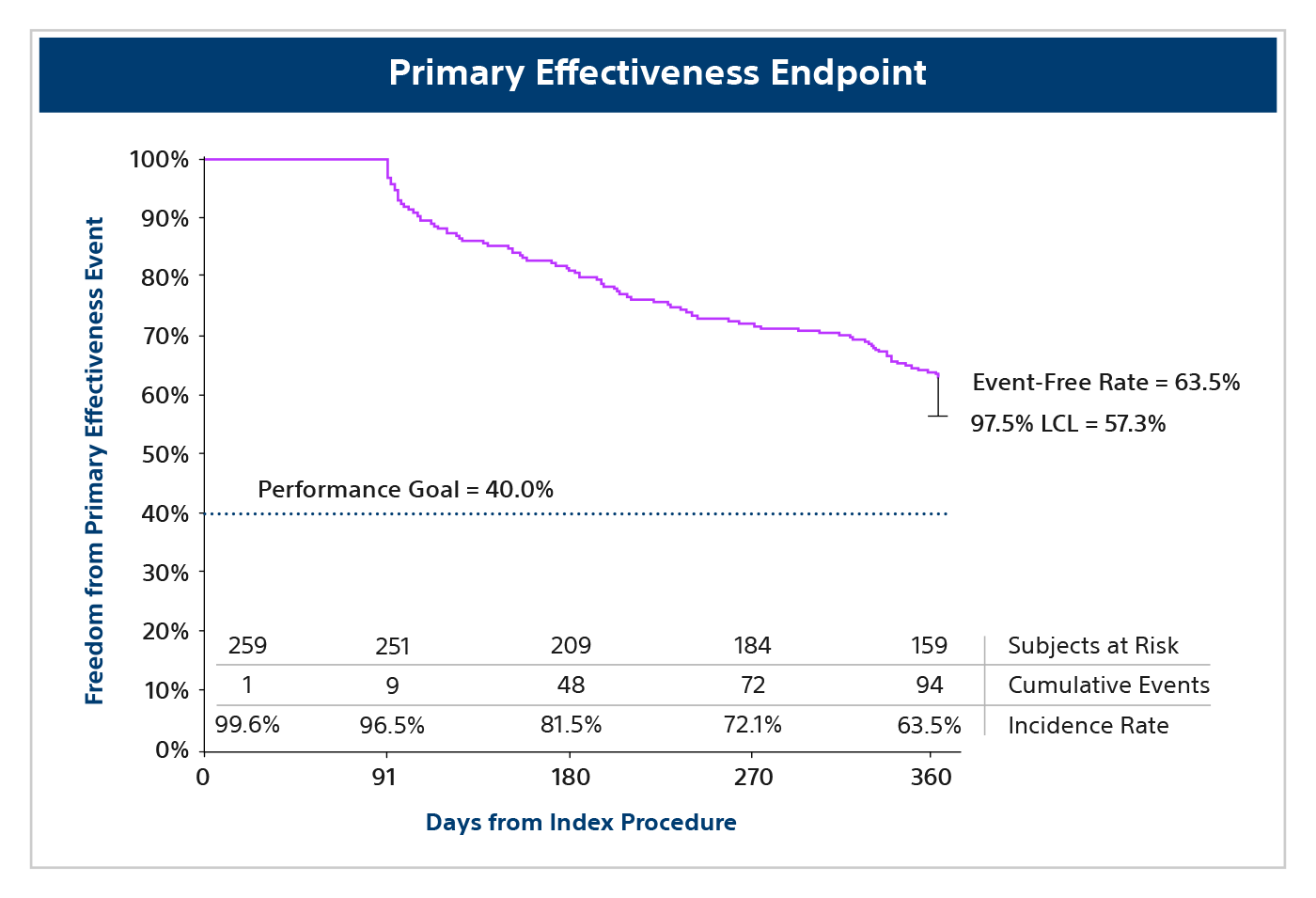
Primary effectiveness endpoint (Phase I)
A composite endpoint defined as acute and chronic ablation success through 12 months. After the 90-day blanking period, chronic success was defined as freedom from AF, AFL or AT, re-ablation, cardioversion and use of a new or escalated dose of Class I/III AADs or Amiodarone.
About the study: ADVANTAGE AF US IDE Phase II
The ADVANTAGE AF US IDE Phase II clinical trial2 included the addition of cavo-tricuspid isthmus (CTI) isolation with the FARAPOINT™ Pulsed Field Ablation (PFA) Catheter and continuous monitoring with the LUX-Dx™ Insertable Cardiac Monitor System (ICM) for continuous heart rhythm monitoring.
Key findings: ADVANTAGE AF US IDE Phase II
Primary endpoints met
2.4%
[97.5% UCL = 5.2%]
Primary safety event rate at 3 months
73.4%
[97.5% LCL = 67.5%]
Primary effectiveness event-free rate at 12 months
96.4%
[97.5% LCL = 91.7%]
CTI primary effectiveness event-free rate at 12 months
AA episodes detected with LUX-Dx ICM
52%
No AA recurrence
at 12 months
70%
<1 hour AA episode duration
at 12 months
Patients were monitored with the LUX-Dx ICM which eliminated monitoring bias giving a more comprehensive assessment of ablation efficacy. LUX-Dx detected atrial arrhythmia episodes were adjudicated by BeatLogic™. The BeatLogic algorithm is a cloud-based ECG analysis platform that leverages artificial intelligence (AI) algorithms and deep learning to automate ECG interpretation.
Study design and methods: ADVANTAGE AF US IDE Phase II
29 US sites
49 Investigators
255 PersAF patients
PVI and PWA with FARAWAVE, CTI with FARAPOINT (per protocol)
Continuous monitoring with LUX-Dx ICM and 12-lead ECG at 3 and 12 months
LUX-Dx was used to simulate traditional, symptomatic and Holter monitoring. LUX-Dx detected atrial arrhythmia episodes were adjudicated by BeatLogic to calculate AA burden.
Primary safety endpoint (Phase II)
A composite endpoint defined as: 1) serious adverse event related to either the use of an ablation catheter or the ablation procedure with onset within 7 days of the primary procedure, 2) death, cardiac tamponade/perforation, pericarditis, cardiovascular or pulmonary adverse event related to either the use of the ablation system or procedure out to 30 days and 3) pulmonary vein stenosis or atrio-esophageal fistula out to 3 months.
Primary effectiveness endpoint (Phase II)
A composite endpoint defined as acute procedural success (isolation of PV and LAPW) and chronic ablation success through 12 months. After the 90-day blanking period, chronic success was defined as freedom from AF/AFL/AT recorded on the LUX-Dx ICM (≥30 seconds on simulated bimonthly cardiac event monitor periods ≥30 second or 24-hour Holter monitor periods at Day 180 and 360, and ≥10 seconds on ECG), re-ablation, cardioversion and use of a new or escalated dose of Class I/III AADs or Amiodarone.
Primary CTI effectiveness endpoint (Phase II)
Composite endpoint of CTI dependent AFL acute (bidirectional conduction block at index procedure) and chronic (freedom from documented CTI dependent AFL (≥ 10 sec on ECG) without a repeat CTI ablation) procedural success.
About the study: ADVANTAGE CTI sub-analysis
The ADVANTAGE AF CTI sub-analysis3 compared the safety and effectiveness of the FARAPOINT PFA Catheter (Phase II) to RFA (Phase I)—the standard of care—for adjunctive CTI ablation in PersAF patients.
- All patients received PVI and PWA with the FARAWAVE PFA Catheter
- At operator discretion, patients were additionally treated for AFL with adjunctive CTI ablation using either:
- RFA in Phase I (n=50, 19%)
- FARAPOINT PFA in Phase II (n=141, 55%)
Key findings: ADVANTAGE CTI sub-analysis
The FARAPOINT PFA Catheter demonstrated similar safety and effectiveness to RFA, while enabling significantly greater procedural predictability.
CTI ablation time*
Phase I (RFA-CTI)
14 min
(9-19 min)
n=50
Phase II (FARAPOINT-CTI)
5 min
(4-8 min)
n=141
*Median CTI ablation time (min); p<0.001
The FARAPOINT PFA Catheter provided comparable freedom from atrial flutter recurrence and safety outcomes.
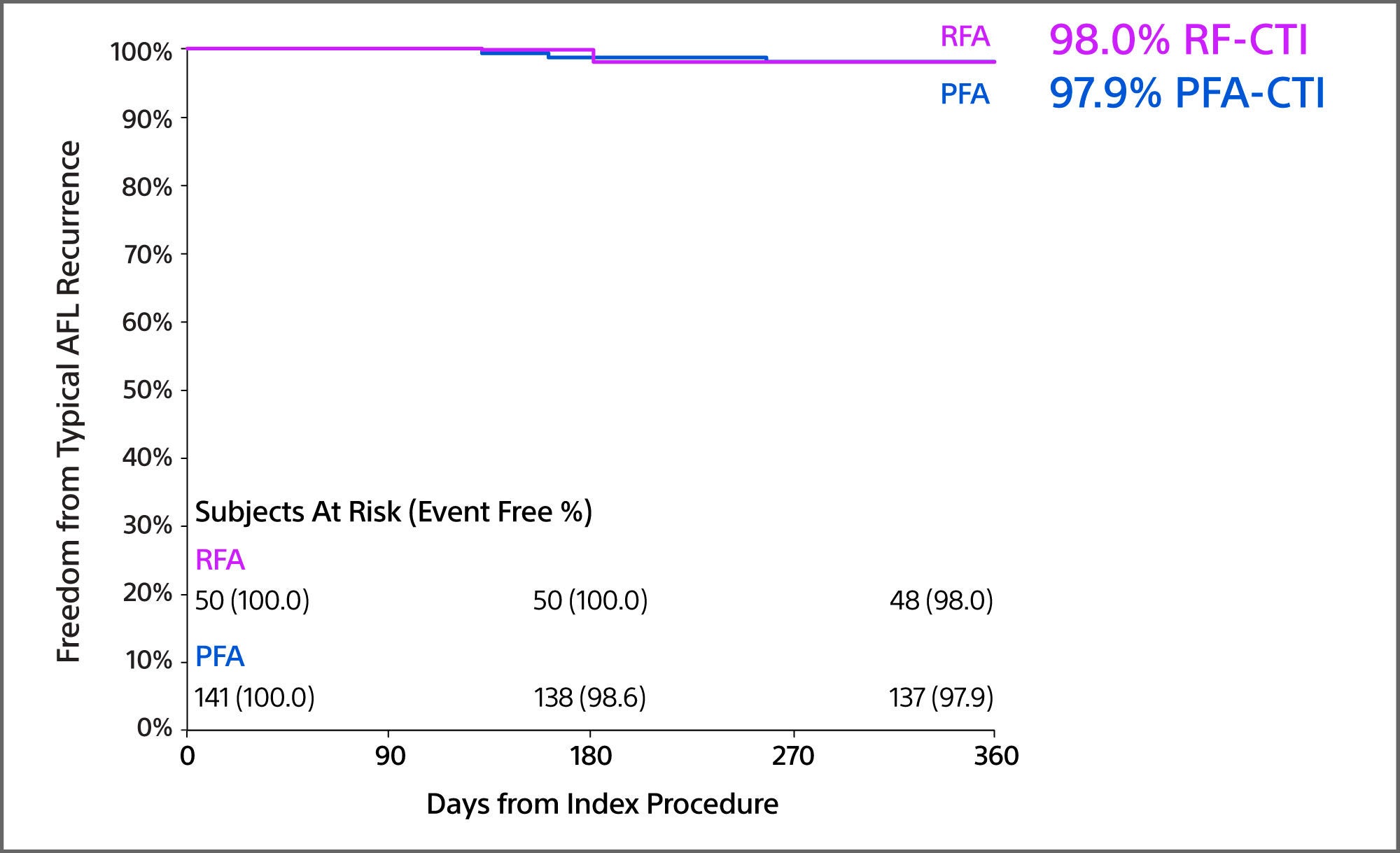
Freedom from Typical AFL Recurrence
p=0.95
CTI Primary Safety Event Rate
p = 0.96
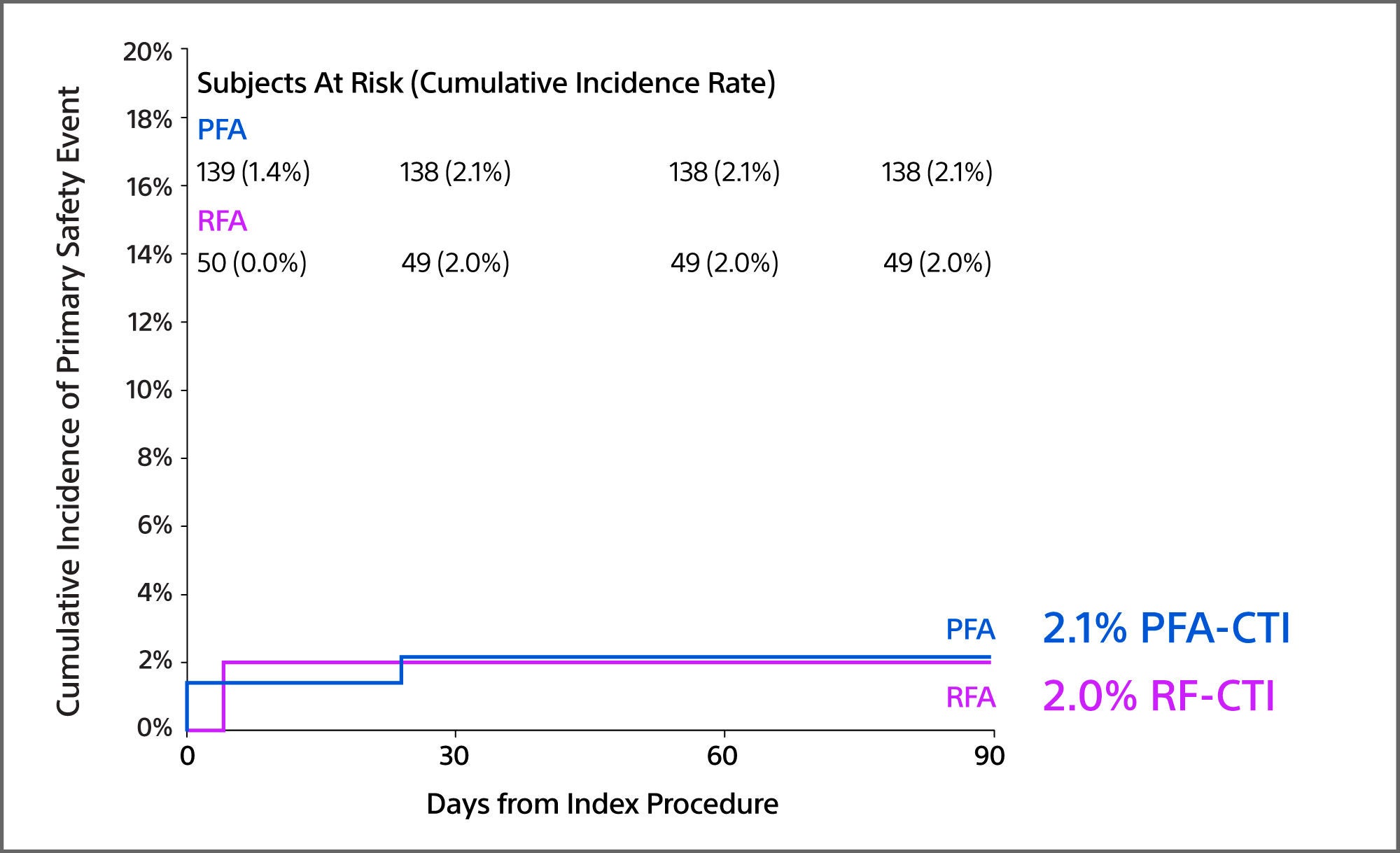
Conclusions: ADVANTAGE CTI sub-analysis
- Significantly greater predictability in CTI ablation time with FARAPOINT PFA (5 min (4-8) vs RFA (14 min (9-19)) (p<0.001)
- Safety rates were similar (PFA=2.1% vs RFA=2.0%; p=0.96). There were no instances of clinical coronary artery spasm in those who were treated with FARAPOINT (99.0% received prophylactic NTG per protocol with 54.0% receiving only 3 mg).
- The freedom from recurrence of typical AFL was similar in the two cohorts at 97.9% for FARAPOINT-CTI and 98.0% for RFA-CTI (p=0.95).