Innovative portfolio with differentiated technologies
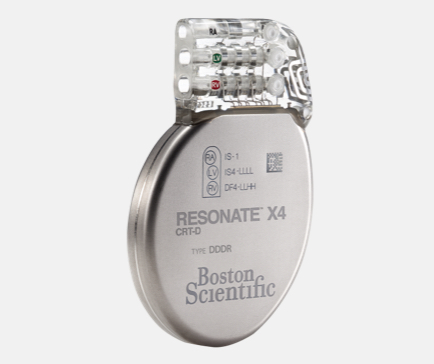
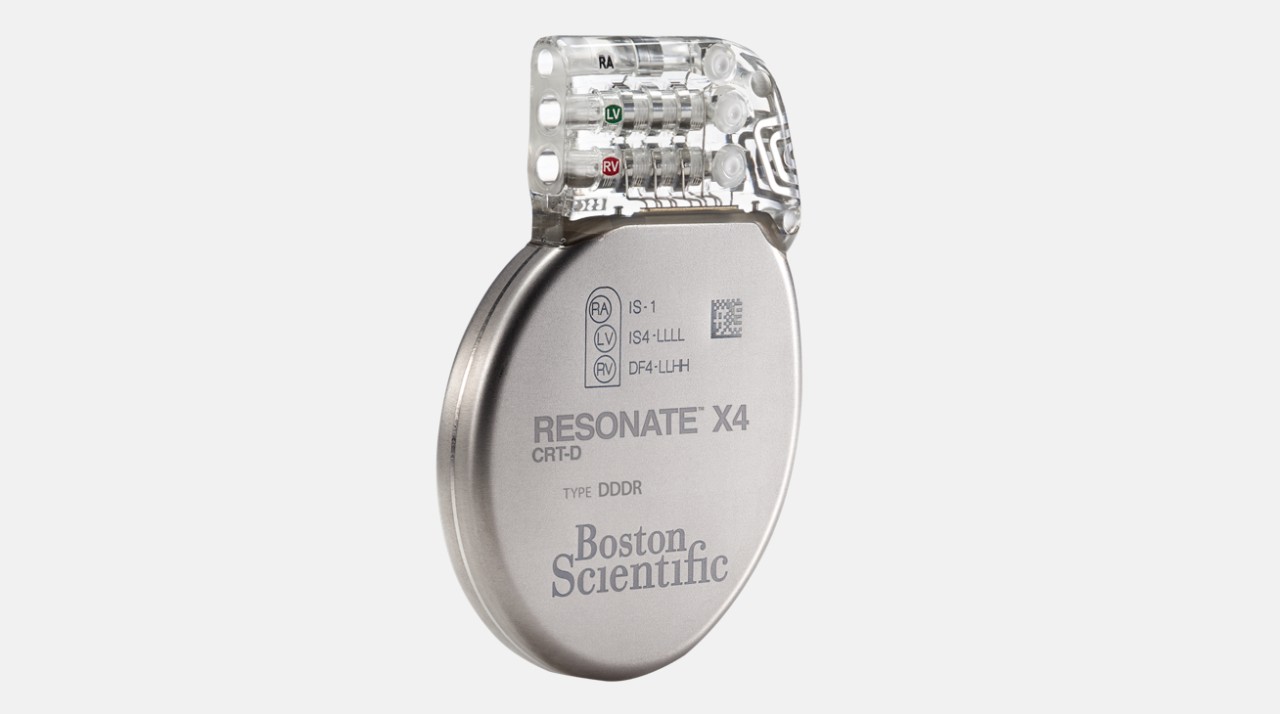
RESONATE™ Heart Failure Diagnostic
Demonstrated 67% reduction in heart failure hospitalizations

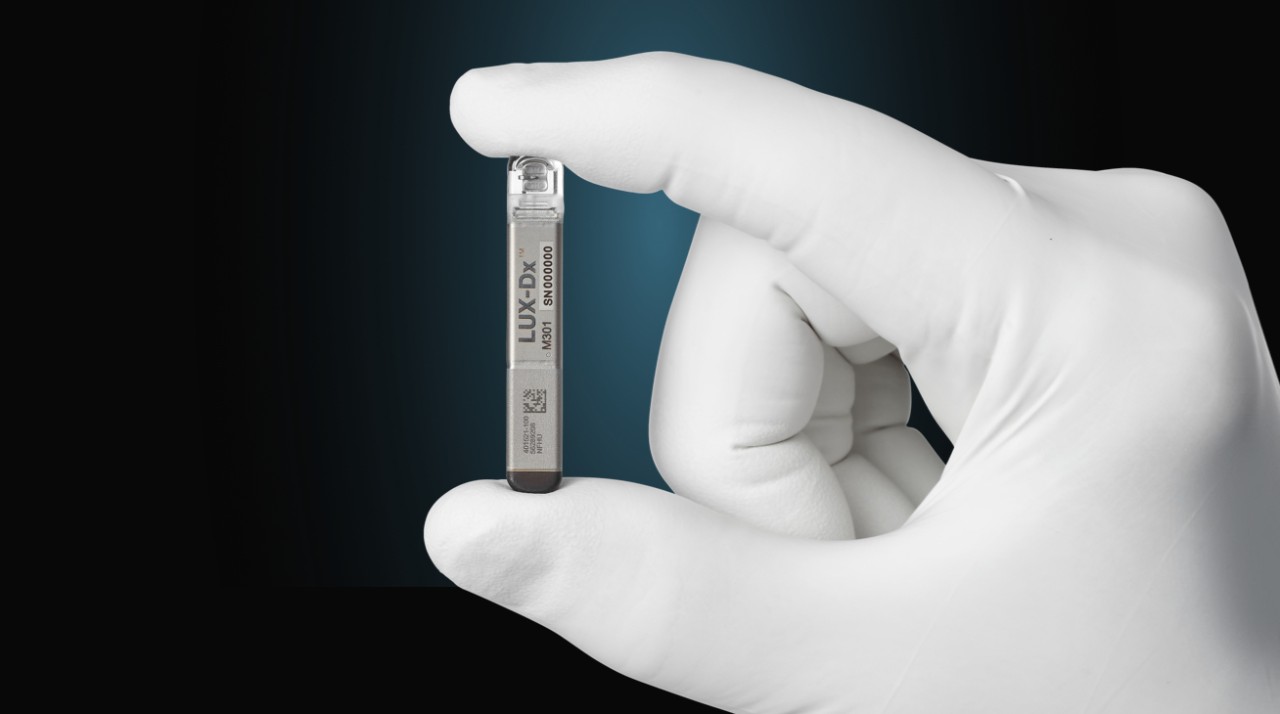
LUX-DX™ ICM
53% reduction in false positives
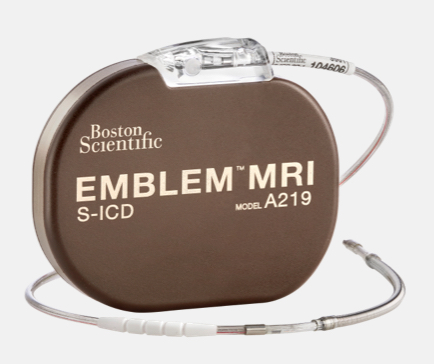
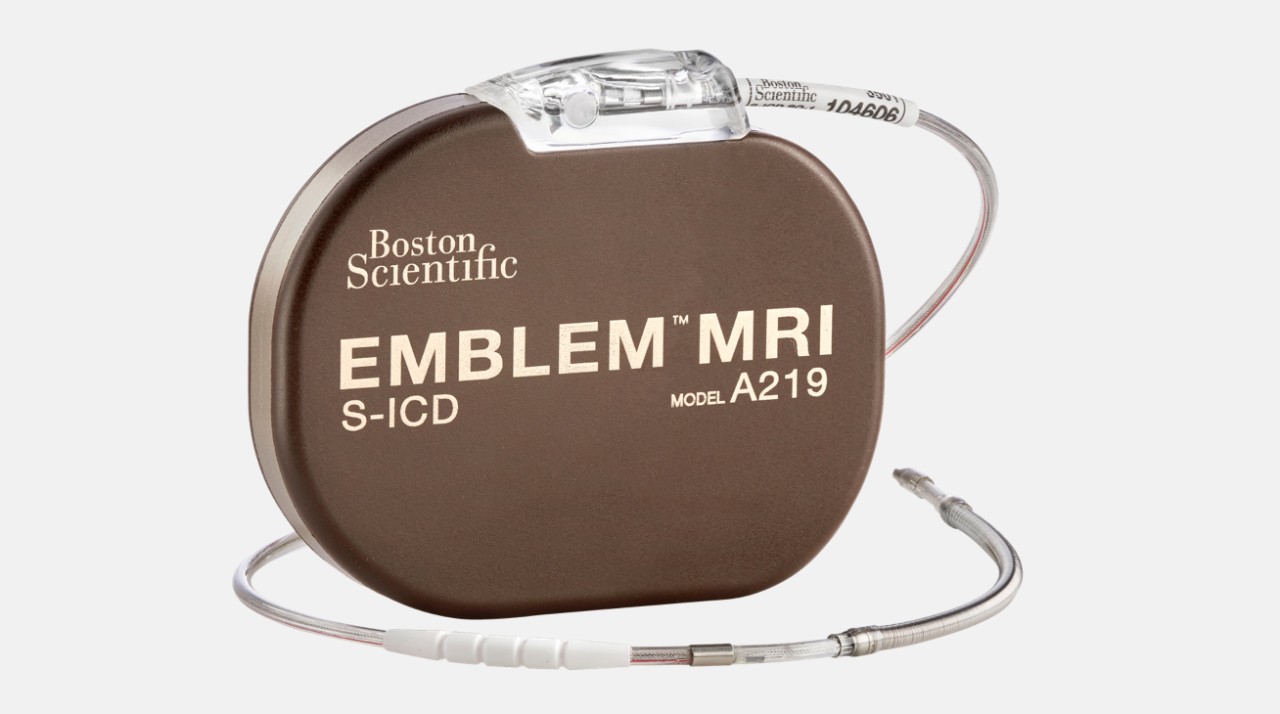
EMBLEM™ MRI S-ICD
Only FDA-approved subcutaneous ICD device
With the broadest diagnostics portfolio and differentiated ICD/CRT-D and S-ICD technologies, we can help you strengthen quality outcomes, increase operational efficiencies, enhance patient experience and improve the financial health of your cardiovascular service line.
Multiple Sensors
Heart Sounds
Reveals signs of elevated filling pressure and weakend ventricular contraction
Thoracic Impedance
Measures fluid accumulation and pulmonary edema
Respiration
Monitors rapid shallow breathing pattern associated with shortness of breath
Heart Rate
Indicates cardic status and arrhythmias
Activity
Shows activity levels and reflects the patient's overall status and fatigue
The impact of heart failure on quality of life and healthcare economics
1.1 Million+
hospitalizations attributed to heart failure each year6
25%
of heart failure patients are readmitted within 30 days of initial hospitalization7,8
$100 Billion
approximate expected heart failure spend by 20309
Improve efficiencies and patient outcomes
The MulitSENSE study assessed more than 900 patients and validated that the HeartLogic algorithm provides a sensitive and timely predictor of impending heart failure decompensation.16
70% sensitivity
in detecting heart failure events
A median of 34 days
advance notice of worsening heart failure
<2 total alerts
per patient per year
Watch to discover how HeartLogic™ can reduce heart failure-related hospitalizations.
The need for better detection is clear
5.3 M
People in the U.S. have atrial fibrillation1
13%
Of AF cases go undiagnosed1
450k+
Hospitalizations each year with AF as the primary diagnosis2
500k+
Patients remotely monitored with diagnostics each year in the U.S4
100k
Studies with an AF diagnosis5
- Identify clinically significant arrhythmias with BeatLogic® deep-learning and artificial intelligence
- Identify arrhythmias earlier, to improve clinical outcomes
- Obtain near real-time analysis of AECG data with PatientCare cloud-based platform
- Increase efficiency with Split-Bill and Patient Care models
Minimize risks associated with transvenous leads
200k+
De Novo transvenous ICDs are implanted worldwide each year11
14x
Higher rate of lead complications requiring surgical intervention with transvenous ICDs than S-ICD12
54.8%
Of first time ICD recipients were candidates for S-ICD13
- High conversion success of 98.4%14, similar to that reported with TV-ICD trials
- Protection from risks associated with transvenous leads, S-ICD demonstrated fewer lead and device-related complications than TV-ICD over a 4-year follow up15
- Reduces the need for fluoroscopy during the implant procedure, helping to improve efficiencies