GreenLight XPS™
Laser Therapy System
Meet the Primary Investigator
Listen to one of the GOLIATH study Principal Investigators, Professor Alexander Bachmann, discuss some of the key findings of the GOLIATH study.
Equally Effective. Equally Safe.
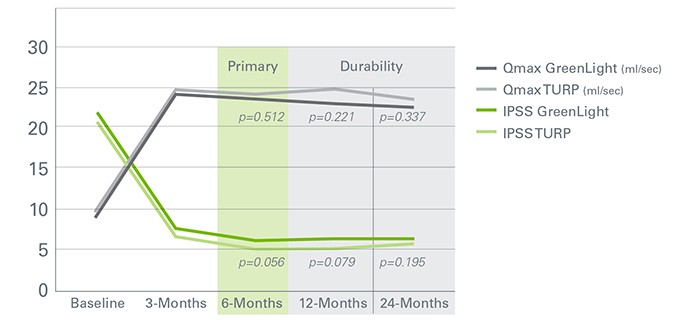
At the 6 month follow up, the GOLIATH Study showed that the GreenLight XPS system was equivalent to TURP in reducing benign prostatic hyperplasia (BPH) symptoms (IPSS) and prostate volume as well as improving quality of life (QoL). The two treatments also had similar Qmax and Dysuria rates. These clinical outcomes remained true in the 24 month follow up.
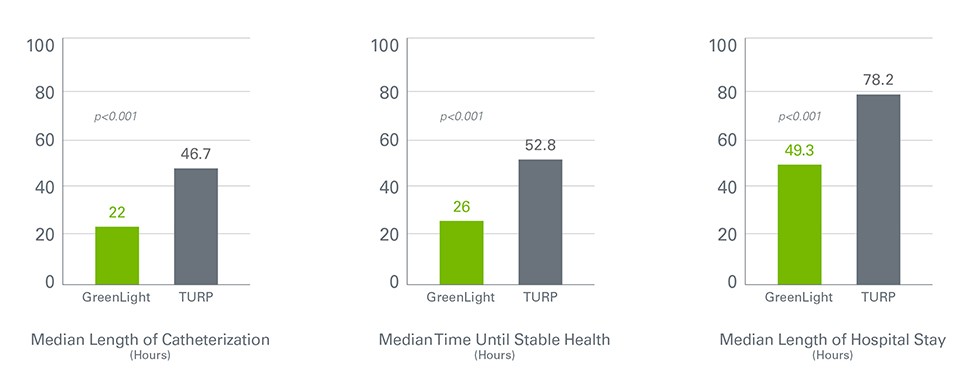
GreenLight Laser Therapy is superior to TURP for Healthcare Facility Length of Stay
Patients treated with GreenLight therapy had a significantly shorter mean length of catheterization, time until stable health and hospitalization.
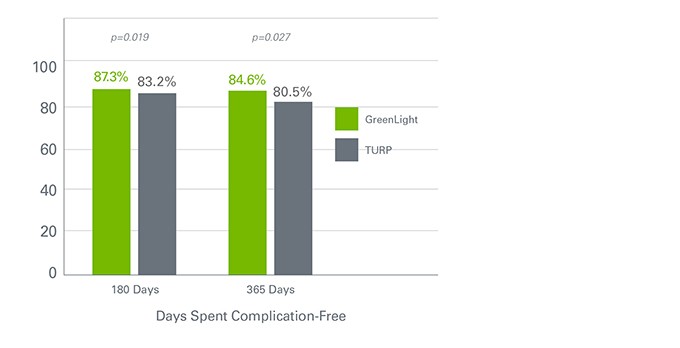
GreenLight Procedure Results in Fewer Complications
The proportion of patients who were complication-free during the first six months was comparable between the GreenLight XPS system and TURP. These results continued to be comparable at 24 months. However, there was a statistically significant difference in early adverse events (48 hours to 30 days), with 12 complications in the TURP group and zero in the GreenLight XPS arm.
Presentation Deck
Interested in sharing the GOLIATH Study for presenting GOLIATH at your facility?
Infographic
Learn more about the GOLIATH Study by downloading an infographic summary.