Boston Scientific accounts are for healthcare professionals only.

RESONATE™ Cardiac Resynchronization Therapy Defibrillator (CRT-D)
Reimbursement
Configure or select a product to continue to order
- Overview
- Shock polarity examples
- Clinical data
- Technical specifications
- Ordering information
- Training
- Resources
Innovative technology, powerful insights
Identify heart failure decompensation,1 increase CRT response2,3 and reduce costs3
How it works
RESONATE X4 and RESONATE HF are Boston Scientific’s most technically advanced CRT-Ds available with the following features:
Provides 8 shocks in the VF zone with the option to choose shock polarity and automatically invert shock polarity in a series of shocks
EnduraLife™ Battery Technology
HeartLogic™ Heart Failure Diagnostic
SmartCRT™ Technology
ImageReady™ MR-Conditional Systems
RightRate™ Minute Ventilation (available only with RESONATE HF)
Why choose RESONATE?

Extended device life
CRT-D devices with EnduraLife Battery Technology have up to 13.3 years projected longevity*, giving you the clinical freedom to make programming decisions that optimize therapy for the patient, not the device.

Actionable heart failure insights
The HeartLogic Heart Failure Diagnostic uses multiple sensors to track physiological trends, combines them into one composite index and sends a proactive alert of potential worsening heart failure.1

Tailored CRT programming
SmartCRT Technology helps you maximize patient response to CRT therapy by providing customized programming to obtain CRT response in most patients.3
RightRate Minute Ventilation
The only sensor clinically proven to restore chronotropic competence and maintain healthy heart rate variability.5,6
RightRate dual sensor adapts to changes in movement and respiration, regulating the patient's ability to increase their heart rate during exercise or other everyday activities. Hear directly from Don Hopper, an active patient, as he shares his journey with RESONATE HF in the video.
Available only with RESONATE HF CRT-D.
Unrestricted MRI access
Patients can safely get full body MRI scans at 1.5T or 3T with ImageReady MR-Conditional Systems devices without exclusion zone, scan duration, or patient height restrictions.7,8
ImageReady is compatible with the RESONATE X4 and RESONATE HF CRT-Ds.
Prevent sudden cardiac death
All our transvenous ICDs and CRT-Ds are available to implant provide 8 shocks in the ventricular fibrillation (VF) zone, safely support programmable shock polarity, and automatically invert polarity in a series of shocks.
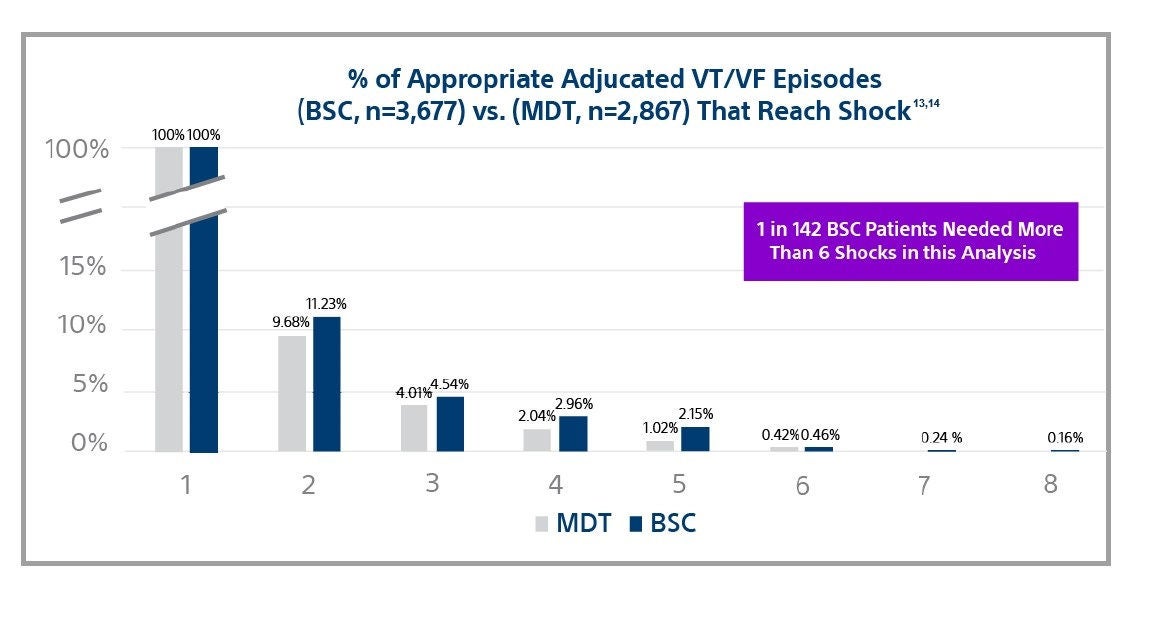
One in 142 patients needed more than 6 shocks to convert an episode of VT/VF9
Among leading manufacturers, shock efficacy is nearly identical for shocks 1-6 with 0.46% and 0.42% of adjudicated VT/VF episodes reaching the 6th shock.9,10 However, Boston Scientific devices provide 8 shocks in the VF zone while others only provide 6.
| Boston Scientific | Medtronic | Abbott | |
| Shocks in the VF Zone | 8 | 6 | 6 |
| Programmable and Recommended Bi-Directional Shock Polarity | Yes | No* | Yes |
| Automatic Shock Polarity Invert | Yes | No* | No |
*The Medtronic May 2023 Advisory and associated instructions for Use warn users that AX>B pathway is not recommended due to increased risk for reduced-energy output or no-energy output during high voltage therapy. The B>AX pathway is nominal and recommended for all high-voltage pathways in the Cobalt XT, Cobalt Crome, Claria, Amplia, Compia, Viva, Brava, Visia AF, Evera, Primo, and Mirro ICDs and CRT-Ds, which are also trademarks of Medtronic and its affiliates.
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Shock polarity examples
Large clinical trials show the mortality benefit of Implantable Cardiac Defibrillators (ICDs) is a result of successful shocks.11,12 See the value our transvenous ICDs and CRT-Ds deliver as they prevent sudden cardiac death in these real-world examples.
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Proven cardiac care
MANAGE-HF trial
Investigated the clinical integration and safety of the FDA-approved HeartLogic™ Heart Failure Diagnostic in the management of patients with heart failure.
HeartLogic was safely integrated into clinical care 13
Early treatment augmentation was associated with more rapid recovery of the HeartLogic index compared to patients with no change in treatment following an alert13
HeartLogic was associated with a 67% reduction in heart failure hospitalization compared to pre-study, 12-month HF hospitalization rate14
67%
reduction in HF hospitalizations associated with HeartLogic*
*Compared with pre-study hospitalization rate (12 months)
SMART-MSP trial
The SMART-MSP clinical trial exceeded both of its end points:
Safety endpoint: The MultiSite Pacing (MSP) feature-related complication-free rate at 180 days post MSP was 99%
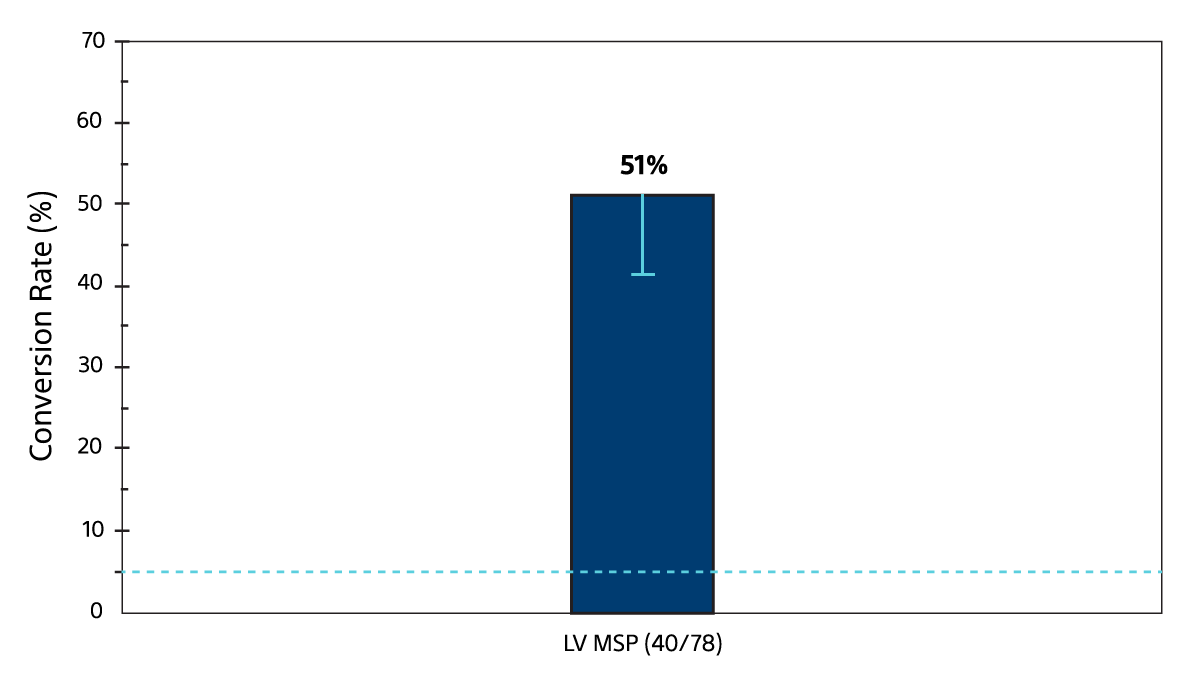
Effectiveness endpoint: 51% of the non-responders at 6 months converted to responders at 12 months3
MSP Effectiveness
NAVIGATE X4 trial
The NAVIGATE X4 trial was a prospective, non-randomized, multi-center, single-arm clinical study that included 764 patients in 88 implant centers.
50% of ACUITY™ X4 leads were placed in 6 minutes or less and required no reoperation due to pacing capture or thresholds
77.3% of ACUITY X4 spiral leads were programmed with a proximal electrode as the pacing cathode15

ACUITY X4 LEADS
REQUIRED ZERO REOPERATIONS
DUE TO PACING CAPTURE OR THRESHOLDS
PREEMPT-HF study
The PREEMPT-HF study evaluated the correlation between HeartLogic sensor data and the likelihood of readmission after hospitalization for heart failure.16 Key findings from the study were:
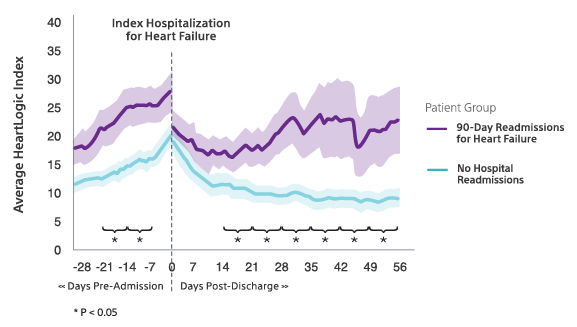
Patients in a HeartLogic alert state 14 days after hospital discharge had triple the risk of 90-day readmission for heart failure compared to those not in alert.
HeartLogic trends were significantly different for patients who were readmitted following an index heart failure hospitalization compared to patients without readmissions.
Battery longevity without sacrificing performance
The Heart Rhythm Journal's article, The variability of implantable cardioverter-defibrillator battery longevity evaluated 43,000+ single chamber ICD, dual chamber ICD and biventricular ICDs (CRT-Ds). The study concluded that the factors most associated with longevity were the manufacturers, programmed pulse width, and programmed output.17 Boston Scientific consistently had the longest battery life across all ICD types.17
Longevity of ICDs reaching replacement index
Observed median and (IQR) values given in months
| Device Type | BIO | BSX | MDT | ABT | P Value |
| Single-Chamber ICDs | 84.0 (64.6-100.9) | 151.3 (136.6-163.8) | 107. (96.1- 115.7) | 129.5 (115.0- 141.4) | <.001 |
| Dual-Chamber ICDs | 92.9 (80.5- 100.7) | 134.1 (118.3- 143.4) | 91.7 (84.4- 101.0) | 108.6 (100.7- 118.8) | <.001 |
| Biventricular ICDs (CRT-Ds) | 76.0 (61.5- 85.7) | 110.6 (96.9-123.1) | 78.2 (67.4- 88.2) | 83.1 (71.6- 91.8) | <.001 |
ABT= Abbot, BIO= Biotronik, BSX= Boston Scientific, MDT= Medtronic, IQR= interquartile range
Median lifetime lead amplitude, pulse width, impedance, pacing percentage, and lower rate limit as well as shock therapies delivered and shock therapies aborted were similar across device manufacturers for each device type, except for small differences in pacing percentages.
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Mechanical specifications
| Model | Type | Size (cm) (W x H x D) | Mass (g) | Volume (cc) | Connector Type (RA RV LV) |
| G547 (HF) | X4 CRT-D | 5.37 x 8.18 x 0.99 | 73.8 | 32.5 | RA: IS-1; RV; DF4; LV: IS4 |
| G447 | X4 CRT-D | 5.37 x 8.18 x 0.99 | 73.8 | 32.5 | RA: IS-1; RV; DF4; LV: IS4 |
EasyView™ Header and Feedthrough System
Discover the innovation behind the RESONATE family of ICDs and CRT-Ds EasyView Header & Feedthrough System —engineered for precision, durability, and seamless integration. The system preserves the electrical isolation of the lead wires as they pass into the internal circuitry while concurrently maintaining the hermeticity of the pulse generator.
Longevity information
The following tables represent sample pulse generator life expectancy estimation (implant to explant) with EnduraLife™ batteries as provided in product labeling.
| Projected longevityᵃ | Ventricular Chambers | RA/RV | LV | LVbᵈ | 500Ω with LATITUDE™ᵇ | 700Ω with LATITUDEᵇ | 700Ω with LATITUDE RS, or HFSSᶜ |
| MultiSite Pacing Off | |||||||
| Typical programmed setting | BiV | 2.5 V | 3.0 V | Off | 9.7 | 10.5 | 11.3 |
| Maximum labeled longevity | LV-Only | 2.0 V / Off | 2.0 V | Off | 12.9 | 13.2 | 14.7 |
| MultiSite Pacing On | |||||||
| Typical programmed setting | BiV MSP | 2.5 V | 3.0 V | 3.0 V | 8.2 | 9.1 | 9.7 |
| Maximum labeled longevity | LV-Only MSP | 2.0 V / Off | 2.0 V | 2.0 V | 11.15 | 12.1 | 13.3 |
| Projected longevity ᵃ | Ventricular Chambers | RA/RV | LV | LVbᵈ | 500Ω with LATITUDE™ᵇ | 700Ω with LATITUDEᵇ | 700Ω with LATITUDE RS, or HFSS ᶜ |
| MultiSite Pacing Off | |||||||
| Typical programmed setting | BiV | 2.5 V | 3.0 V | Off | 9.7 | 10.5 | 11.3 |
| Maximum labeled longevity | LV-Only | 2.0 V / Off | 2.0 V | Off | 12.9 | 13.2 | 14.7 |
| MultiSite Pacing On | |||||||
| Typical programmed setting | BiV MSP | 2.5 V | 3.0 V | 3.0 V | 8.2 | 9.1 | 9.7 |
| Maximum labeled longevity | LV-Only MSP | 2.0 V / Off | 2.0 V | 2.0 V | 11.15 | 12.1 | 13.3 |
Assumes 70 PPM LRL; DDDR mode; 0.4 ms Pulse Width (RA, RV, LV); sensors On, Heart Failure Sensor Suite On.
Projected longevity is calculated assuming 2 maximum energy charging cycles per year, including automatic capacitor reforms and therapeutic shocks. These calculations also assume 3-channel EGM Onset is on and that the pulse generator spends 3 months in Storage mode during shipping and storage.
a. Assumes ZIP telemetry use for 2 hours at implant and for 40 minutes annually for in-clinic follow-up checks.
b. Assumes standard use of the LATITUDE Communicator as follows: Daily Device Check on, quarterly scheduled remote follow-ups, and other typical interrogations.
c. Assumes LATITUDE Communicator is not used, Respiratory Sensor is Off, and Heart Failure Sensor Suite is Off.
d. Applies to models with MultiSite Pacing (MSP).
Boston Scientific devices have corporate warranties at 6 years in available geographies. Warranty information available at www.bostonscientific.com/warranty.
Devices use Li/MnO2 chemistry.
The Usable Battery Capacity is 1.9 Amp-hours (typical implant to battery capacity depleted).
Shelf life is 2 years (before use by date).
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Ordering information
| Model | Description |
| G547 | RESONATE HF CRT-D |
| G447 | RESONATE X4 CRT-D |
Reimbursement
The C-Code for RESONATE is C1882. C-codes are used for hospital outpatient device reporting for Medicare and some private payers.
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Education for RESONATE

Expand your knowledge of all Cardiac Rhythm Management and Diagnostics topics, including EMBLEM™ S-ICD, LUX-Dx™ ICMs, the LATITUDE™ Programming System Model 3300, MRI conditional systems, and more.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Resources
Subscribe for updates on RESONATE
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
References:
1. Boehmer JP, Hariharan R, Devecchi FG, et al. A Multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. JACC Heart Fail. 2017 Mar;5(3):216-25.
2. Ellenbogen KA, Gold MR, Meyer TE, et al. Primary results from the SmartDelay determined AV optimization: a comparison to other AV delay methods used in cardiac resynchronization therapy (SMART-AV) trial: a randomized trial comparing empirical, echocardiography-guided, and algorithmic atrioventricular delay programming in cardiac resynchronization therapy. Circulation. 2010;122(25):2660-2668.
3. Saba S, Nair D, Ellis CR, et al. Usefulness of Multisite Ventricular Pacing in Nonresponders to Cardiac Resynchronization Therapy. Am J Cardiol. 2022;164:86-92.
4. Williams J, et al. Modeling Long-term Effect of Biventricular Defibrillator Battery Capacity on Major Complications and Costs Associated with Replacement Procedures. Poster presented at: 2021 Heart Rhythm Society; July 2021; Boston, MA.
5. Chronotropic competence is defined by the Model of the Cardiac Chronotropic Response to Exercise. Wilkoff B, Corey J, Blackburn G. A mathematical model of the cardiac chronotropic response to exercise. J Electrophysiol. 1989 June;3(3):176-80.
6. Richards, et al., The Addition of Minute Ventilation to Rate Responsive Pacing Improves Heart Rate Score More than Accelerometer Alone. Heart Rhythm 2018.https://doi.org/10.1016/j.hrthm.2018.06.02.
7. Reference ImageReady MRI Technical Guide
8. Only when Conditions of Use are met
9. Cha YM, Hayes DL, Asirvatham SJ, Powell BD, Cesario DA, Cao M, Gilliam FR 3rd, Jones PW, Jiang S, Saxon LA. Impact of shock energy and ventricular rhythm on the success of first shock therapy: the ALTITUDE first shock study. Heart Rhythm. 2013 May;10(5):702-8. doi: 10.1016/j.hrthm.2013.01.019. Epub 2013 Jan 19. PMID: 23337541.
10. Medtronic White Paper, Determining the Efficacy of Monophasic Shocks, https://www.hrsonline.org/documents/medtronic-white-paperdetermining- efficacy-reduced-energy-monophasic-shocks/download.
11. G.H. Bardy, K.L. Lee, D.B. Mark, et al., for the SCD-HeFT Investigators. Amiodarone or an ICD for congestive heart failure: N Engl J Med, 352 (2005), pp. 225-237
12. Moss AJ , Zareba W, Hall WJ, et al. Prophylactic implantation of a defibrillator in patients with myocardial infarction and reduced ejection fraction. N Engl J Med. 2002;346:877–883 *Assumes 70 PPM LRL; DDDR mode; 0.4 ms Pulse Width (RA, RV, LV); sensors On, Heart Failure Sensor Suite On.
13. Hernandez AF, Albert NM, Allen LA, et al. Multiple cArdiac seNsors for mAnaGEment of Heart Failure (MANAGE-HF) - Phase I Evaluation of the Integration and Safety of the HeartLogic Multisensor Algorithm in Patients With Heart Failure. J Card Fail. 2022;28(8):1245-1254. doi:10.1016/j.cardfail.2022.03.349
14. Hernandez AF, Albert NM, Allen LA et al. Multiple cardiac sensors for management of heart failure (MANAGE-HF) Phase I results. Abstract presented/published at: ESC-HF 2021. June 29-July 1, 2021. Virtual. https://onlinelibrary.wiley.com/doi/epdf/10.1002/ejhf.2297 (Pg 159)
15. Clinical Summary: NAVIGATE X4 Study 358487-022 EN US 2016-01
16. Sauer AJ, Stolen CM, Shute JB, et al. Results of the Precision Event Monitoring for Patients With Heart Failure Using HeartLogic Study (PREEMPT-HF). JACC Heart Fail. 2025;13(6):973-983. doi:10.1016/j.jchf.2025.01.028
17. Freeman JV, Torre M, Sanders P, et al. Variability in implantable cardioverter-defibrillator battery longevity. Heart Rhythm. Published online May 22, 2025. doi:10.1016/j.hrthm.2025.05.031
†When conditions of use are met.