Boston Scientific accounts are for healthcare professionals only.
EluPro™ Antibiotic-Eluting BioEnvelope
Reimbursement
Configure or select a product to continue to order
- Overview
- Implant Technique
- Ordering information
EluPro Antibiotic-Eluting BioEnvelope
EluPro BioEnvelope provides localized antibiotic protection, reduces pocket fibrosis, and supports natural healing for safer, more stable cardiac implants.1
How it works
EluPro delivers the trusted antibiotics rifampin and minocycline directly at the implant site, providing broad-spectrum protection against pathogens linked to CIED infections.2,3 The antibiotics elute locally over at least seven days4, maintaining effective levels during the period of highest infection risk.
This targeted delivery system creates a protected healing environment around the device. By combining antimicrobial defense with biocompatibility, EluPro helps reduce pocket fibrosis, supports healthy tissue regrowth, and promotes long-term device stability, all without increasing systemic antibiotic exposure.1
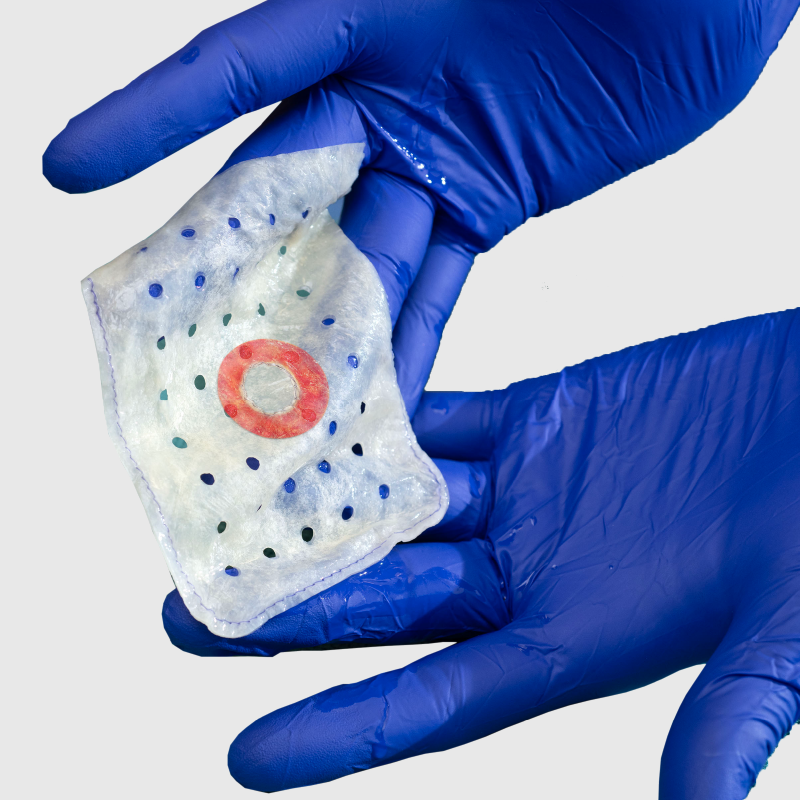
Why choose the EluPro BioEnvelope?
EluPro combines proven infection protection with regenerative healing in one bioenvelope, helping reduce post-implant complications while supporting healthy, vascularized tissue growth.

100% bacterial kill in preclinical testing
Successfully eliminated bacterial colonization, thus reducing the potential for infection.4,5
Soft feel and natural lubricity
Conforms to the shape of the CIED to enhance the implantation experience with no modification necessary to the envelope or incision.1,6

Supports natural biologic healing
Reduces foreign-body response, which mitigates inflammation and fosters vascularization and integration.1,7
Key features and specifications
Localized antibiotic protection
EluPro delivers rifampin and minocycline directly at the implant site, providing broad-spectrum antimicrobial protection against pathogens most often associated with CIED infections.2,3 The antibiotics elute gradually for at least seven days4, maintaining effective levels during the period of highest infection risk.
Biologic matrix for natural healing
Constructed from a biocompatible, biologic material, the EluPro BioEnvelope supports natural tissue remodeling and vascularization.6-9 This regenerative scaffold minimizes inflammation and foreign-body response, helping create a stable implant pocket with healthy tissue.
Enhanced surgical handling
The soft, conformable biomatrix easily adapts to various CIED shapes and sizes. Its natural lubricity supports device placement, pocket closure, and procedural efficiency while maintaining structural integrity during handling.1,6
Reduced reoperation complexity
In clinical studies, Elutia’s bioenvelope technology has been shown to decrease lead adhesions and simplify reoperative procedures.7-9 The result is easier device replacement and less disruption to surrounding tissue.
Extended local efficacy without systemic impact
By concentrating antibiotic activity at the surgical site, EluPro reduces reliance on systemic antibiotic dosing.4 This targeted approach may provide high local efficacy while limiting systemic exposure and potential antibiotic resistance concerns.
Implant technique
EluPro offers a quick, easy implant technique that fits your workflow.
Step 1:
To use, select the appropriate size EluPro for the CIED. Handle the sterile bioenvelope using aseptic technique.
Step 2:
Hydrate EluPro by immersing it completely in sterile solution (e.g. saline, water) for 1-2 minutes.
Step 3:
Slide the CIED into EluPro, with lead wires emerging out of the opening. Secure the bioenvelope with one stitch.
Step 4:
Implant the CIED with the bioenvelope into the pocket and secure it to the fascia per standard practice.
Ordering information
| Description | Model | Specs | Quantity |
| Medium | CMCV-124-MED | 6.9cm x 6.5cm | Single pack |
| Large | CMCV-124-LRG | 6.9cm x 8.0cm | Single pack |
EluPro BioEnvelope size M

EluPro BioEnvelope size L
Subscribe for updates on EluPro
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
References:
1. Catanzaro JN, et al. Front Cardiovasc Med. 2025;12:1638929. doi:10.3389/fcvm.2025.1638929
2. Chemaly RF, Athan E, Mack J, et al. Antibiotic prophylaxis for cardiac implantable electronic device procedures: review of clinical evidence. Int J Infect Dis. 2010;14(6):e548-e552.
3. Sohail MR, Uslan DZ, Khan AH, et al. Management and outcome of permanent pacemaker and implantable cardioverter-defibrillator infections. J Am Coll Cardiol. 2007;49(18):1851-1859. doi:10.1016/j.jacc.2007.01.072
4. Garrigos ZE, Catanzaro JN, Deegan D, Zhang J, Sohail MR. Preclinical evaluation of a novel antibiotic-eluting BioEnvelope for CIED infection prevention. Front Drug Deliv. 2024;4:1441956. doi:10.3389/fddev.2024.1441956
5. Büttner H, Kramer A, Fux M, et al. Bacterial colonization and infection prevention in implantable devices: insights from preclinical models. Front Cell Infect Microbiol. 2015;5:14.
6. Catanzaro JN, Wood MM, Kramer KB, et al. Clinical outcomes of bioenvelope use during CIED changeouts: the HEAL study. Heart Rhythm. 2023;20(5 suppl):S712-S713.
7. Deegan D, Piasecki SK, Riebman JB. An acellular biologic extracellular matrix envelope for cardiovascular implantable electronic devices: preclinical evaluation. J Regen Med. 2022;11(5):Article 5. doi:10.1097/01.mat.0000841052.84245.36
8. Srivastava A, Nayak HM. Regenerative response following CIED reimplantation with a biologic envelope. HeartRhythm Case Rep. 2023;9(12):797-799.
9. Catanzaro JN, Wood MM, Kramer KB, et al. Comparative tissue response with biologic vs non-biologic device envelopes. Circulation. 2022;146(suppl 1):A11052.