Boston Scientific accounts are for healthcare professionals only.
About EnduraLife Battery Technology
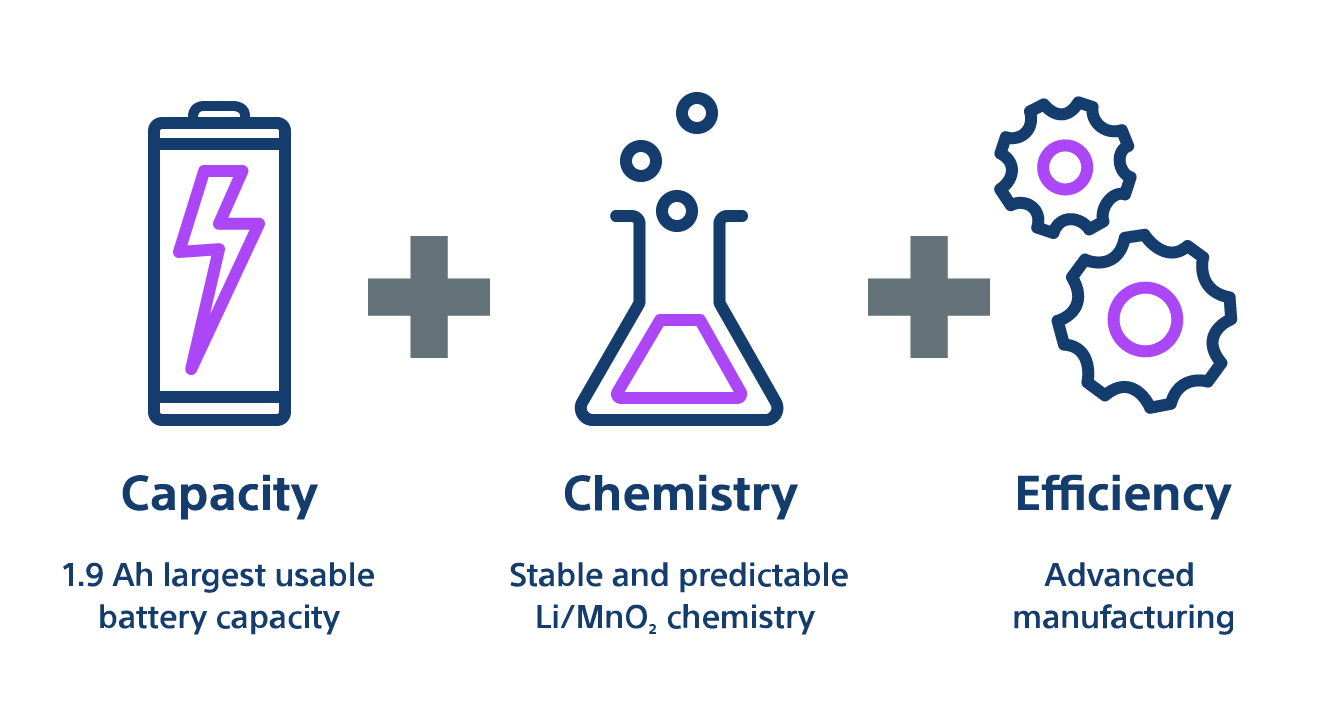
Battery longevity without sacrificing performance
- Capacity — 1.9 Amp-hours of usable battery capacity - highest in the industry1
- Chemistry – Lithium manganese dioxide maintains voltage and resistance stability for battery reliability
- Efficiency — Up to 8% smaller and 24% thinner devices2
Minimizing risks
Longer lasting battery means fewer replacements and reduced risks.3
Reducing costs
Fewer device replacements means reduced cost for patients, payers, and the healthcare system.4
Performance
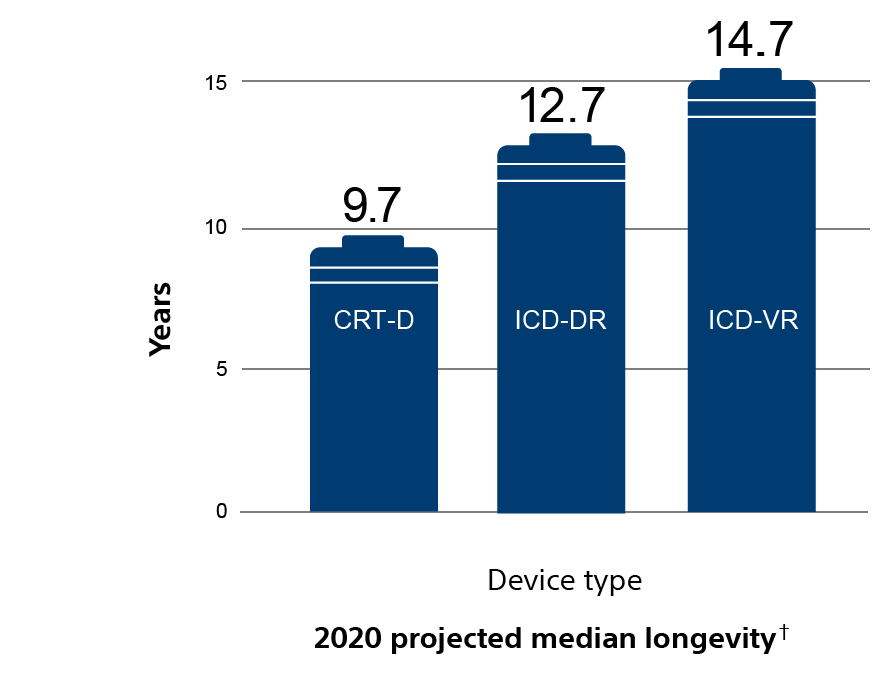
EnduraLife Battery Technology works for up to 13.2 years, outlasting the competition.*
EnduraLife Battery Technology offers substantial cost savings and reduces major clinical complications for patients
Physician perspective on device longevity
Clinical Data
Outlasting the competition
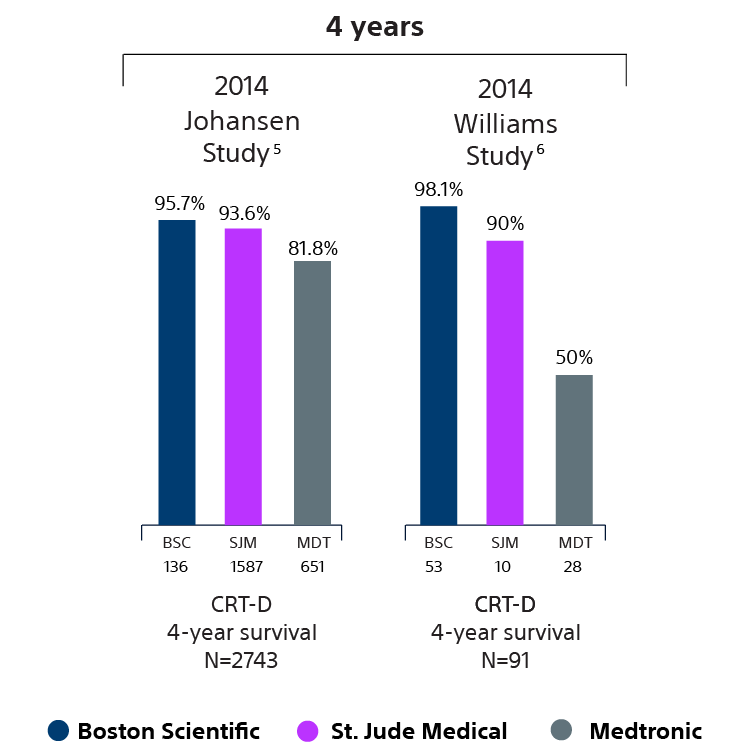
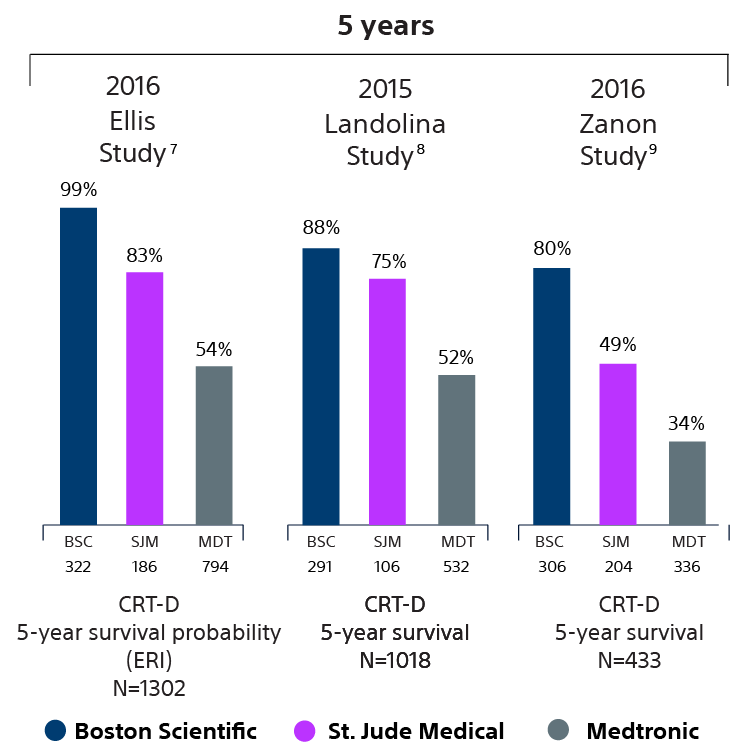
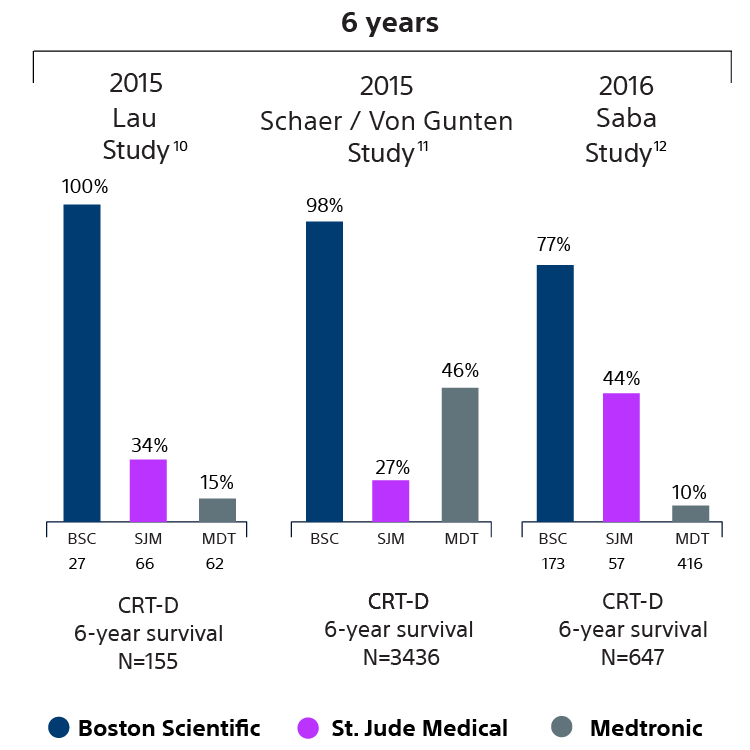
Devices with EnduraLife Battery Technology are lasting longer than comparable devices from both Medtronic and Abbott, as shown in nine independent clinical studies with over 11,314 patients. 5-13
CRT-D survival data
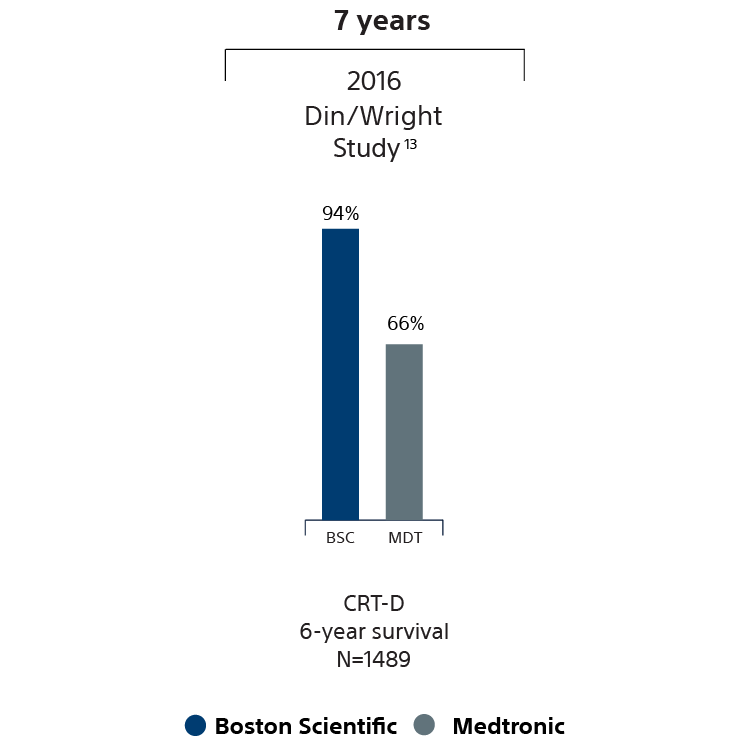
Clinically proven over time
Since 2008, Boston Scientific EnduraLife batteries continue to show impressive longevity.
Products that feature EnduraLife batteries
EnduraLife resources
References
*Assumes 70 PPM LRL; DDDR mode; 0.4 ms Pulse Width (RA, RV, LV); sensors On, Heart Failure Sensor Suite On.
† 2020 data on file at Boston Scientific.
1. Boston Scientific ICDs and CRT-Ds with contemporary battery tehcnology have 1.9 Ah. Medtronic ICDs and CRT-Ds have 1.1 Ah. PHYSICIAN'S TECHNICAL MANUAL RESONATE™ HF ICD, RESONATE™ EL ICD, PERCIVA™ HF ICD, PERCIVA™ ICD, VIGILANT™ EL ICD, MOMENTUM™ EL ICD. Part Number: 51114084-001 EN US 2021-10-27. PHYSICIAN'S TECHNICAL MANUAL RESONATE™ HF CRT-D, RESONATE™ CRT-D, RESONATE™ X4 CRT-D, VIGILANT™ CRT-D, VIGILANT™ X4 CRT-D, MOMENTUM™ CRT-D, MOMENTUM™ X4 CRT-D. Part Number: 51113879-001 EN US 2021-10-27. Medtronic Cobalt™ XT DR ICD MRI SureScan™ Model DDPA2D4 device manual. Medtronic Cobalt™ XT VR ICD MRI SureScan™ Model DVPA2D4 device manual. Medtronic Cobalt™ XT HF Quad CRT-D MRI SureScan™ DTPA2QQ 2019-04-23 device manual.
2. Boston Scientific Implantable Cardioverter Defibrillator Physician’s Technical Manual 359401-002 page 30 and 359403-002 page 27. St. Jude Medical High Voltage Devices based on battery discharge testing performed by BSC, data on file. 2/10/2010. Medtronic Evera™ XT VR DVBB2D4 Device Manual page 25.
3 Williams JL, Harley B, Williams G, “First Demonstration of Cardiac Resynchronization Therapy Defibrillator Service Life Exceeding Patient Survival in a Heart Failure with Reduced Ejection Fraction Cohort,” J Innov Cardiac Rhythm Manage. 2020; 11(12): 4325–4332.
4 Williams J, et al. Modeling Long-term Effect of Biventricular Defibrillator Battery Capacity on Major Complications and Costs Associated with Replacement Procedures. Poster presented at: 2021 Heart Rhythm Society; July 2021; Boston, MA.
5 Haarbo J, Hjortshoj S, Johansen J, Jorgensen O, Nielsen J, Petersen H. Device Longevity in Cardiac Resynchronization Therapy Implantable Cardioverter Defibrillators Differs Between Manufacturers: Data from the Danish ICD Registry. Presented at HRS 2014. http://ondemand.hrsonline.org/common/presentation-detail.aspx/15/35/1241/9000. Boston Scientific = 136 patients, Medtronic = 6 51 patients, St. Jude Medical = 1,587 patients, Bitronik = 369 patients. Time to exchange of the device because of battery depletion or device failure recorded in the Danish ICD Registry was the endpoint. The four-year survival rate for devices in the Danish Registry study was 81.1% for Medtronic and 95.7% for Boston Scientific (P<0.01).
6 J. Williams, R. Stevenson. Contemporary cardiac resynchronization implantable cardioverter defibrillator battery longevity in a community hospital heart failure cohort. Presented at HFSA 2014. http://www.onlinejcf.com/article/. S1071-9164(14)00389-3/fulltext. Boston Scientific = 53 patients, Medtronic = 28 patients, St. Jude Medical = 10 patients. Four-year survival rate calculated using device replacements for battery depletion as indicated by ERI.
7 Ellis CR, Dickerman DI, Orton JM, Hassan S, Good EG, Okabe T, Andruilli JA, Quan KJ, Greenspon AJ. Ampere Hour as a Predictor of Cardiac Resynchronization Defibrillator Pulse Generator Battery Longevity: A Multicenter Study. PACE 2016 doi: 10.1111/pace.12831 first published online 11-MAR-2016. The five major institutions performing the study include, at Vanderbilt University, Henry Ford Hospital, University of Michigan, Thomas Jefferson University, Cooper Health System, North Ohio Heart Center. Boston Scientific = 322 patients, Medtronic = 794 patients, St. Jude Medical = 186 patients. Five-year survival rate calculated using device replacements for battery depletion as indicated by ERI.
8 Landolina M, Curnis A, Morani G, Vado A, Ammendola E, D’onofrio A, Stabile G, Crosato M, Petracci B, Ceriotti C, Bontempi L, Morosato M, Ballari GP, Gasparini M. Longevity of implant Cardioverter-defibrillators for cardiac resynchronization therapy in current clinical practice: an analysis according to influencing factors, device generation, and manufacturer. Europace2015;17:1251-58. doi:10.1093/eurospace/euv109. First published online: May 14, 2015. Medtronic = 532 patients, Boston Scientific = 291 patients, St. Jude Medical = 106 patients, Biotronik = 20 patients, Sorin = 69. Five-year survival rate of latest marketed devices (between 2006 to 2010) calculated using device replacements for battery depletion as indicated by ERI.
9 Zanon F, Martignani C, Ammendola E, Menardi E, Narducci ML, De Filippo P, Santamaria M, Campana A, Stabile G, Potenza DR, Pastore G, Iori M, La Rosa C, and Biffi M. Device Longevity in a Contemporary Cohort of ICD/CRT-D Patients Undergoing Device Replacement. Doi:10.1111/jce.12990, First published online 20-APR-2016. Comparison of device longevity by Kaplan-Meier curves of CRT-D systems extracted between March 2013 and May 2015. Medtronic = 195 patients, Boston Scientific = 157 patients, St. Jude = 72, Biotronik = 9.
10 Provided by Dr. Ernest Lau on 04/29/15 in support of Lau E, Wilson C, Ashfield K, McNair W, McEneany D, Roberts M, Large Capacity LiMnO2 Batteries Extended CRTD Longevity in Clinical Use Compared to Smaller Capacity LiSVO Batteries Over 6 Years. Presented at HRS 2015. Medtronic = 62 patients, Boston Scientific = 27 patients, St. Jude = 66 patients. Five-year survival rate calculated using device replacements for battery depletion as indicated by ERI.
11 von Gunten S, Schaer BA, Yap SC, Szili-Torok T, Kühne M, Sticherling C, Osswald S, Theuns DA. Longevity of implantable cardioverter defibrillators: a comparison among manufacturers and over time. Europace. 2015 Nov 25; . Epub 2015 Nov 25. Total patients = 3436.
12 Alam MB, Munir MB, Rattan R, Adelstein E, Jain S, Saba S. Battery longevity from cardiac resynchronization therapy defibrillators: differences between manufacturers and discrepancies with published product performance reports. Europace 016;doi:10.1093/europace/euw044. First published online: 22-MAR-2016. Kaplan Meier curves depicting survival of CRT devices free from battery depletion by device manufacturer. Battery Longevity in Cardiac Medtronic = 416 patients, Boston Scientific = 173 patients, St. Jude Medical = 57patients. Previously evaluated these patients at a four-year survival rate calculated using device replacements for battery depletion as indicated by ERI. 2014; Europace (2014) 16,246-51.
13 Shabanna Din, Shabanna, Mcgee, Rao, Archana, Wright, Jay D. Longevity of implantable cardioverter defibrillators: The impact of device manufacturer and device type on device longevity were assessed. Europace. 2015 Nov 25; . Epub 2015 Nov 25. Total patients = 3436. Cardiostim Abstract 2016. Total patients = 1489
14 Boehmer JP, Hariharan R, Devecchi FG, et al. A Multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. JACC Heart Fail. 2017 Mar;5(3):216-25.