Enteral Feeding
Explore Enteral Feeding Products
Choose from a variety of initial placement, replacement and through-the-PEG enteral feeding devices.
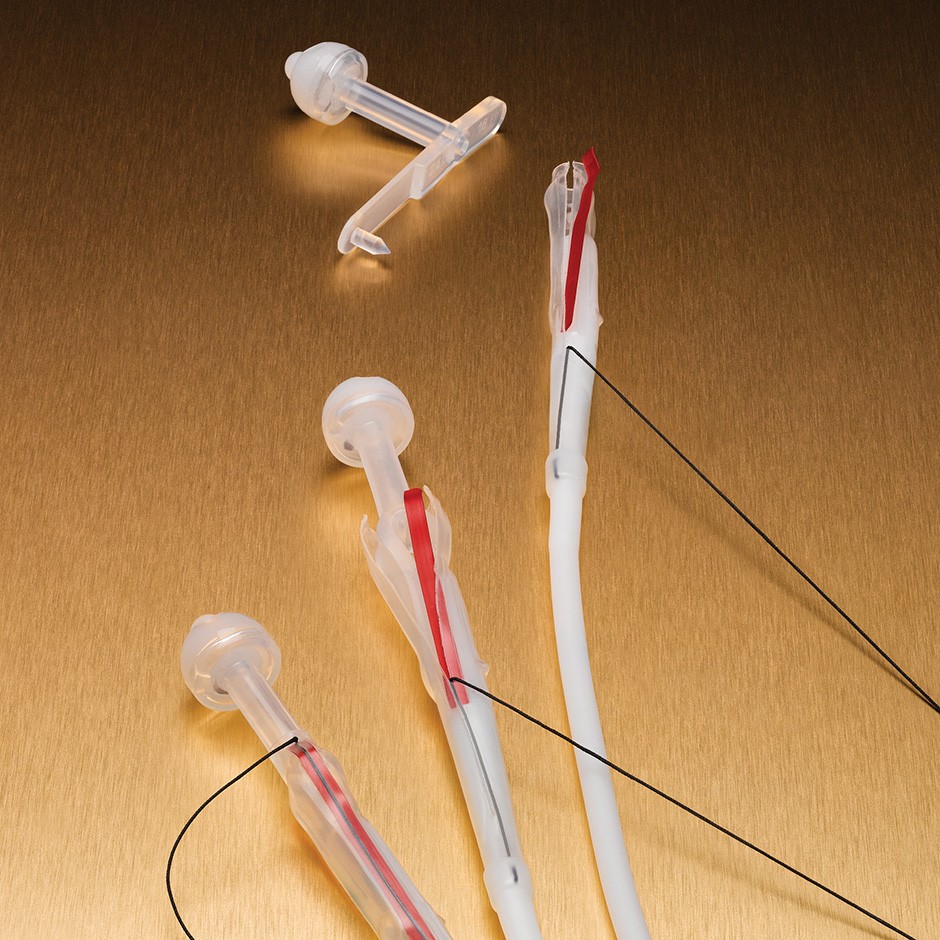
EndoVive™ One-Step Button™ Low Profile Initial Placement Percutaneous Endoscopic
Gastrostomy (PEG) Kit and Accessories
The One-Step Button Low Profile Initial Placement PEG Kit allows for the initial placement of a low profile button.
EndoVive™ Enteral Access Initial Placement: Standard Percutaneous Endoscopic Gastrostomy (PEG) Kit
The Standard Percutaneous Endoscopic Gastrostomy (PEG) Kit's Silicone dome-bolstered PEG tube available in multiple French sizes with two external bolster options to accommodate physician and patient needs: Round and T-bar.
EndoVive™ Enteral Access Initial Placement: Safety Percutaneous Endoscopic Gastrostomy (PEG) Kit
The Safety Percutaneous Endoscopic Gastrostomy (PEG) Kit's Silicone dome-bolstered PEG tube available in multiple French sizes with two external bolster options to accommodate physician and patient needs: Round and T-bar.
EndoVive™ Enteral Access Replacement: Balloon Replacement Gastrostomy Tubes (Straight and Right Angle)
Straight and right angle feeding tubes. Right angle bolster designed to be less obtrusive and may reduce premature pull-out.
EndoVive™ Low Profile Button Replacement Gastrostomy Tube Kit
The EndoVive Low Profile Button design may help reduce the risk of accidental pull-out. The low profile design and flexibility at the skin level may help support patient comfort and discretion.
All trademarks are the property of their respective owners.