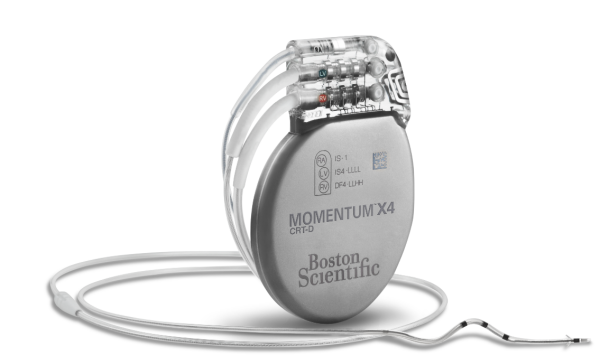
MOMENTUM™ X4 CRT-D and MOMENTUM CRT-D
Powered by EnduraLife™ Battery Technology
Models G138, G124, G125, G126
The MOMENTUMTM X4 CRT-D features SmartCRTTM Technology, an intelligent way to optimize where, when and how to pace. This, coupled with the HeartLogicTM Heart Failure Diagnostic, ImageReadyTM MR-Conditional and EnduraLifeTM Battery Technology means MOMENTUM offers an extraordinary, holistic approach to cardiac care.
Key Resources
Explore
Product Details


EnduraLife Battery Technology offers up to 13.3 years projected longevity with MultiSite Pacing turned on.**

Personalize CRT therapy with smart solutions that allow you to optimize where, when and how to pace.
*Boehmer JP, Hariharan R, Devecchi FG, et al. A Multisensor algorithm predicts heart failure events in patients with implanted devices: resultsfrom the MultiSENSE study. JACC Heart Fail. 2017 Mar;5(3):216-25.
** Assumes: 2.0V RA, LV-only, 2.0V LV, 700Ω, 15% A pacing, 100% LV pacing, No LATITUDE, No Respiratory Rate Sensor, No Heart Failure Sensor Suite.
†When conditions of use are met.
Mechanical Specifications
| Model | Type | Size (cm) (W x H x D) | Mass (g) | Volume (cc) | Connector Type (RA RV LV) | C-Codes |
|---|---|---|---|---|---|---|
| G138 | X4 CRT-D | 5.37 x 8.08 x 0.99 | 73.4 | 32.0 | RA: IS-1; RV: IS-1/DF-1; LV: IS4 | C1882 |
| G124 | CRT-D | 5.37 x 8.18 x 0.99 | 73.6 | 32.5 | RA: IS-1 ; RV: DF4; LV: IS-1 | C1882 |
| G125 | CRT-D | 5.37 x 8.08 x 0.99 | 72.8 | 32.0 | RA: IS-1; RV: IS-1/DF-1; LV: IS-1 | C1882 |
| G126 | CRT-D | 5.37 x 8.08 x 0.99 | 72.9 | 32.0 | RA: IS-1; RV: IS-1/DF-1; LV: LV-1 | C1882 |
Longevity Information
| Projected longevitya | Ventricular Chambers |
RA/RV | LV | LVbd | 500Ω with LATITUDE™b |
700Ω with LATITUDE™b |
700Ω no LATITUDE™, RS, or HFSSc |
|---|---|---|---|---|---|---|---|
| MultiSite Pacing Off | |||||||
| Typical programmed setting | BiV | 2.5 V | 3.0 V | Off | 9.7 | 10.5 | 11.3 |
| Maximum labeled longevity | LV-Only | 2.0 V / Off | 2.0 V | Off | 12.9 | 13.2 | 14.7 |
| MultiSite Pacing On | |||||||
| Typical programmed setting | BiV MSP | 2.5 V | 3.0 V | 3.0 V | 8.2 | 9.1 | 9.7 |
| Maximum labeled longevity | LV-Only MSP | 2.0 V / Off | 2.0 V | 2.0 V | 11.5 | 12.1 | 13.3 |
- Assumes 70 PPM LRL; DDDR mode; 0.4 ms Pulse Width (RA, RV, LV); sensors On, Heart Failure Sensor Suite On.
- Projected longevity is calculated assuming 2 maximum energy charging cycles per year, including automatic capacitor re-forms and therapeutic shocks. These calculations also assume 3-channel EGM Onset is on and that the pulse generator spends 3 months in Storage mode during shipping and storage.
a. Assumes ZIP telemetry use for 2 hours at implant and for 40 minutes annually for in-clinic follow-up checks.
b. Assumes standard use of the LATITUDE™ Communicator as follows: Daily Device Check on, quarterly scheduled remote follow ups, and other typical interrogations.
c. Assumes LATITUDE™ Communicator is not used, Respiratory Sensor is Off, and Heart Failure Sensor Suite is Off.
d. Applies to models with MultiSite Pacing (MSP).
Additional Longevity Information
- Boston Scientific devices have corporate warranties at 6 years in available geographies. Warranty information available at www.bostonscientific.com/warranty.
- Devices use Li/MnO2 chemistry.
- The Usable Battery Capacity is 1.9 Amp-hours (typical implant to battery capacity depleted).
- Shelf life is 2 years (before use by date).