Boston Scientific accounts are for healthcare professionals only.

WallFlex™ Biliary PLUS RX Fully Covered Stent System
Configure or select a product to continue to order
- Overview
- Clinical
- Ordering information
- WallFlex Biliary portfolio
- Training
- Resources
WallFlex Biliary PLUS: anchored in flexibility
The most flexible fully covered biliary metal stent currently available from Boston Scientific,* the WallFlex Biliary PLUS RX Fully Covered Stent System delivers an array of re-engineered features designed to help reduce the risk of migration. It features a decreased axial force* that’s intended to increase conformability1 while maintaining high radial force.2

Why choose the WallFlex Biliary PLUS RX Stent System
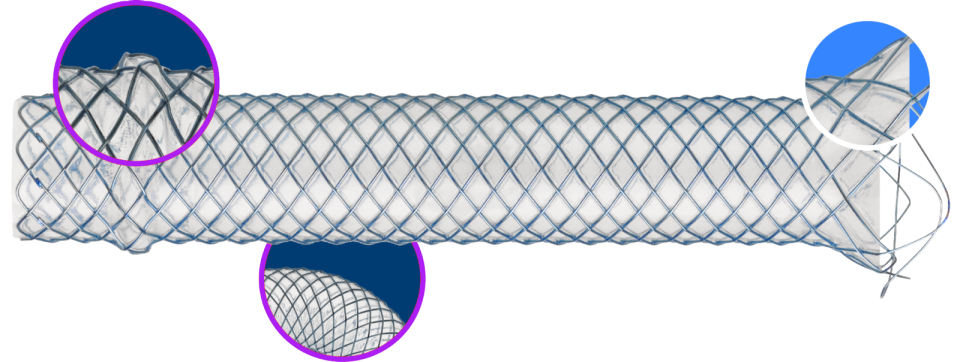
Advanced features for confident placement
An evolution of the WallFlex™ Biliary RX legacy, the WallFlex™ Biliary PLUS RX Fully Covered Metal Stent represents the next generation in the treatment of malignant biliary strictures6. Its reengineered, AXIOS™-inspired proximal bump and enlarged distal flare are designed to help aid against both proximal and distal migration as well as increase conformability due to a decrease in axial force.7
Additional features include reimagined radial forces to help maintain ductal patency9, along with a familiar delivery system that enables deployment control and repositioning.
Considerations for selecting fully covered metal stents
Dr. Jessica Widmer, Associate Professor of Medicine at NYU, shares her clinical perspective on how fully covered stents have influenced treatment approaches, observing that many endoscopists have made the transition away from plastic and uncovered devices to fully covered stents. She notes that when considering cystic duct occlusion, “[Due to] the risks of having the stent occlude with tissue hyperplasia or tumor ingrowth, and difficulty with reinterventions… I prefer fully covered [stents].” Watch the video below to hear Dr. Widmer’s first-hand perspective.
Dr. Widmer is a paid consultant of Boston Scientific.
Common uses of fully covered biliary stents
Fully covered self-expanding metal stents (FCSEMS) are generally used to relieve obstructions and provide biliary drainage.6 For malignant biliary strictures, fully covered metal stents typically provide palliative drainage of bile duct obstructions caused by inoperable cancers. For benign strictures, WallFlex Biliary RX Fully Covered RMV currently has the only removability indication in the US** and can be used for temporary management where sustained patency and removability are required.
Clinical Experiences with WallFlex Biliary PLUS
Watch the video below to hear clinical perspectives and early insights on WallFlex Biliary PLUS from Dr. Carr-Locke and Dr. Widmer.
Dr. Carr-Locke and Dr. Widmer are paid consultants of Boston Scientific.
Ordering information
Product codes
| Material (UPN)/Catalog / REF # | Product description | Stent diameter X length | Box Qty |
| M00547500 | WallFlex Biliary PLUS RX Fully Covered Stent System | 8mm x 60mm | 1 |
| M00547510 | WallFlex Biliary PLUS RX Fully Covered Stent System | 8mm x 80mm | 1 |
| M00547520 | WallFlex Biliary PLUS RX Fully Covered Stent System | 8mm x 100mm | 1 |
| M00547530 | WallFlex Biliary PLUS RX Fully Covered Stent System | 10mm x 40mm | 1 |
| M00547540 | WallFlex Biliary PLUS RX Fully Covered Stent System | 10mm x 60mm | 1 |
| M00547550 | WallFlex Biliary PLUS RX Fully Covered Stent System | 10mm x 80mm | 1 |
| M00547560 | WallFlex Biliary PLUS RX Fully Covered Stent System | 10mm x 100mm | 1 |
| CPT® Code | Code description | ||||||
| 43274 | Endoscopic retrograde cholangiopancreatography (ERCP); with placement of endoscopic stent into biliary or pancreatic duct, including pre- and post-dilation and guide wire passage, when performed, including sphincterotomy, when performed, each stent. | ||||||
| 43275 | Endoscopic retrograde cholangiopancreatography (ERCP); with removal of foreign body(s) or stent(s) from biliary/pancreatic duct(s) | ||||||
| 43276 | Endoscopic retrograde cholangiopancreatography (ERCP); with removal and exchange of stent(s), biliary or pancreatic duct, including pre- and post-dilation and guide wire passage, when performed, including sphincterotomy, when performed, each stent exchanged | ||||||
A proven biliary metal stent portfolio. PLUS so much more.
Choose the biliary metal stent that is right for the patient and their unique stricture with the comprehensive Boston Scientific portfolio. WallFlex Biliary RX Stents are intended to give you the predictable placement and patency you need as you strive to minimize unplanned patient interventions.

See the full WallFlex Biliary portfolio
With Boston Scientific’s comprehensive portfolio, clinicians are able to choose the biliary metal stent that’s appropriate for patient’s clinical presentation and anatomy. The addition of the WallFlex Biliary PLUS RX Stent System provides clinicians with an additional tool to meet individual patient needs.
WallFlex Biliary RX Stents
A comprehensive portfolio of biliary metal stents
Fully covered, partially covered and uncovered WallFlex Biliary RX Stents are available in multiple sizes to accommodate different anatomical and clinical requirements.

WallFlex Biliary PLUS RX Fully Covered Stent System
Stent body re-engineered with anti-migration features including:
- A proximal bump inspired by the AXIOS™ Stent
- An enlarged distal flare* designed to help aid against stent migration
- Designed with balanced axial and radial forces to help conform to the ductal anatomy while addressing patency within the stricture1,2

WallFlex Biliary RX Fully Covered Stent System
A recent addition to the WallFlex Biliary suite, these fully covered stents feature a long length, designed to help clinicians reach strictures that were previously difficult to access.

WallFlex Biliary RX Fully Covered Stent System RMV
The only biliary metal stent in the U.S. with a removability indication.**
Used for treatment of benign biliary strictures secondary to chronic pancreatitis:
• May eliminate the need for repeat procedures3
• May reduce the number of endoscopic retrograde cholangiopancreatography (ERCPs) per patient compared to treatment using multiple plastic stents3

WallFlex Biliary RX Partially Covered Stent System
- High patency rates with a low migration rate6
- Demonstrated migration rate as low as 0%6

WallFlex Biliary RX Uncovered Stent System
A well-trusted tool for biliary malignancy treatment
| WallFlex Biliary PLUS RX Fully Covered Stent System | WallFlex Biliary RX Fully Covered Stent System | WallFlex Biliary RX Fully Covered Stent System RMV | WallFlex Biliary RX Partially Covered Stent System | WallFlex Biliary RX Uncovered Stent System | ||||
|---|---|---|---|---|---|---|---|---|
| Diameters (mm) | 8 | 10 | 8,10 | 8 | 10 | 8 | 10 | 8, 10 |
| Lengths (mm) | 60, 80, 100 | 40, 60, 80, 100 | 100 | 60, 80 | 40, 60, 80 | 60, 80, 100 | 40, 60, 80, 100 | 40, 60, 80, 100 |
| Malignant biliary neoplasm | ||||||||
| Bridge to surgery | ||||||||
| Benign biliary strictures secondary to chronic pancreatitis, with indwell up to 12 months | ||||||||
Considerations for stent selection in malignant strictures***
Dr. Marya is a paid consultant of Boston Scientific.
Evaluating and choosing fully covered biliary metal stents***
Dr. Widmer is a paid consultant of Boston Scientific.
Considerations in choosing uncovered or fully covered biliary metal stents***
Dr. Widmer, Dr. Gromski, Dr. Pineda and Dr. Adler are paid consultants of Boston Scientific.
WallFlex Biliary Stent training modules
Duration: 21:43
Duration: 14:06
Duration: 33:27
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training relevant, comprehensive, personal, and accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Resources
Ready for WallFlex PLUS?
Talk to your Boston Scientific Representative or request a representative reach out to you.
*Compared to WallFlex Biliary RX Fully Covered Stents.
**As of January 2026; WallFlex Biliary RX Fully Covered RMV is indicated for benign biliary strictures secondary to chronic pancreatitis, with indwell up to 12 months
***Results from case studies are not necessarily predictive of results in other cases. Results in other cases may vary.
Indications for use:
WallFlex Biliary RX Stent System, WallFlex Biliary PLUS RX Stent System: indicated for use in the palliative treatment of biliary strictures produced by malignant neoplasms and relief of malignant biliary obstruction prior to surgery.
WallFlex Biliary RX Stent System RMV: indicated for use in the palliative treatment of biliary strictures produced by malignant neoplasms, relief of malignant biliary obstruction prior to surgery, and for indwell up to 12 months in the treatment of benign biliary strictures secondary to chronic pancreatitis.
Warning: The safety and effectiveness of this device for use in vascular system has not been established.
Sources:
1. Devière J, et al. "Plastic and metal stents for benign biliary strictures after orthotopic liver transplantation: a randomized controlled trial." Gastroenterology. 2014;146(3):562–569.
2. Sandha GS, et al. "Endoscopic therapy for bile leak based on a new classification: results in 207 patients." Gastrointest Endosc. 2004;60(4):567–574.
3. Baron TH. "Covered self-expandable metal stents for benign biliary tract diseases." Curr Opin Gastroenterol. 2011;27(3):262–267.
4. The conformability of the Biliary PLUS RX Fully Covered Stent System was assessed through axial force measurements in DV bench tests conducted by Boston Scientific, demonstrating that its axial force is lower than that of other self-expandable metal stents, as referenced in the literature by Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49. Data on file. Bench testing is not indicative of clinical performance.
5. The radial force of the WallFlex Biliary PLUS RX Fully Covered Stent System was assessed through radial force measurements in DV bench tests conducted by Boston Scientific, demonstrating that its radial force exceeds the threshold set by other self-expandable metal stents, as defined in the literature by Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49. Data on file. Bench testing is not indicative of clinical performance.
6. Isayama H, et al. "A prospective randomized study of covered versus uncovered metallic stents for distal malignant biliary obstruction." Gut. 2004;53(5):729–734.
7. Soderlund C, Linder S. Covered metal versus plastic stents for malignant common bile duct stenosis: a prospective, randomized, controlled trial. Gastrointest Endosc. 2006;63(7):986-995.
8. Moss AC, Morris E, Mac Mathuna P. Palliative biliary stents for obstructing pancreatic carcinoma. Cochrane Database Syst Rev. 2006;(1):CD004200. Published 2006 Jan 25.
9. Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49.
10. Yokota Y, Fukasawa M, Takano S, et al. Partially covered metal stents have longer patency than uncovered and fully covered metal stents in the management of distal malignant biliary obstruction: A retrospective study. BMC Gastroenterol. 2017;17:105.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.





