Boston Scientific accounts are for healthcare professionals only.

Eluvia™ Drug-Eluting Vascular Stent System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical Data
- Technical specifications
- Ordering information
- Training
- Resources
Eluvia is the #1 most-implanted SFA stent.1
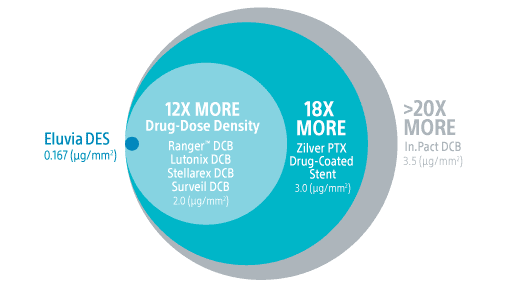
Eluvia DES provides consistent, durable outcomes in challenging superficial femoral artery (SFA) disease and features a polymer design for controlled drug release.

How it works
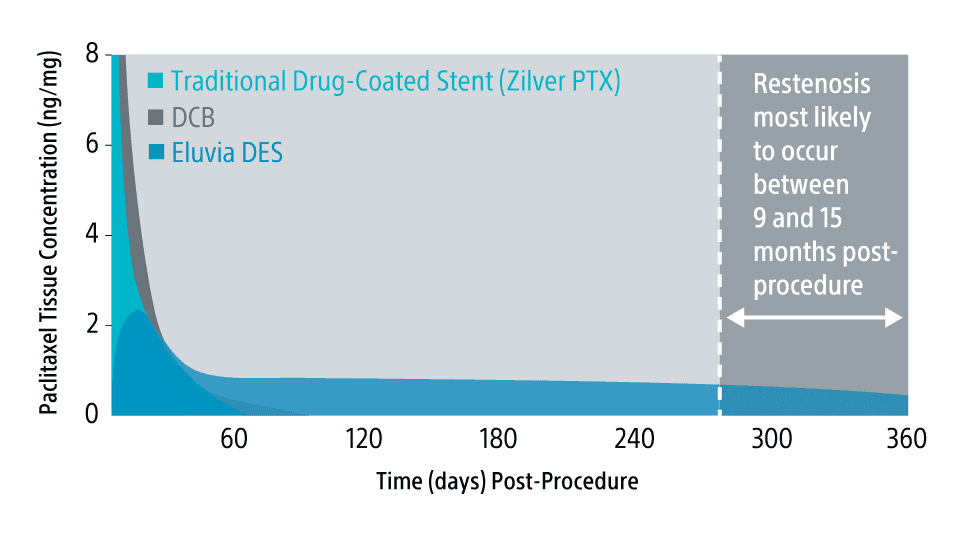
Engineered for the challenges of the SFA, Eluvia DES is the only drug-eluting stent designed to efficiently deliver drug to the target lesion for over a year, when restenosis is most likely to occur in the SFA.
Choose Eluvia
References:
*These investigator-sponsored studies are supported by grant funding from Boston Scientific. Boston Scientific is not responsible for the collection, analysis or reporting of these studies, which remain the sole responsibility of the investigators. Information for the use in countries with applicable product registrations.
**The CAPSICUM study was supported by the Research Association for Lower Limb Artery Revascularization (LIBERAL) sponsored by the following companies: Boston Scientific Japan K.K., OrbusNeich Foundation, Terumo Corp., and Kaneka Medix Corporation. The funding companies played no role in the design of the study; selection of the enrolled patients; treatment strategy; revascularization procedures or equipment; or collection, analysis, or interpretation of the data.
*** No BSC funding
1. Data on file at Boston Scientific. DRG US hospital market data, June 2024
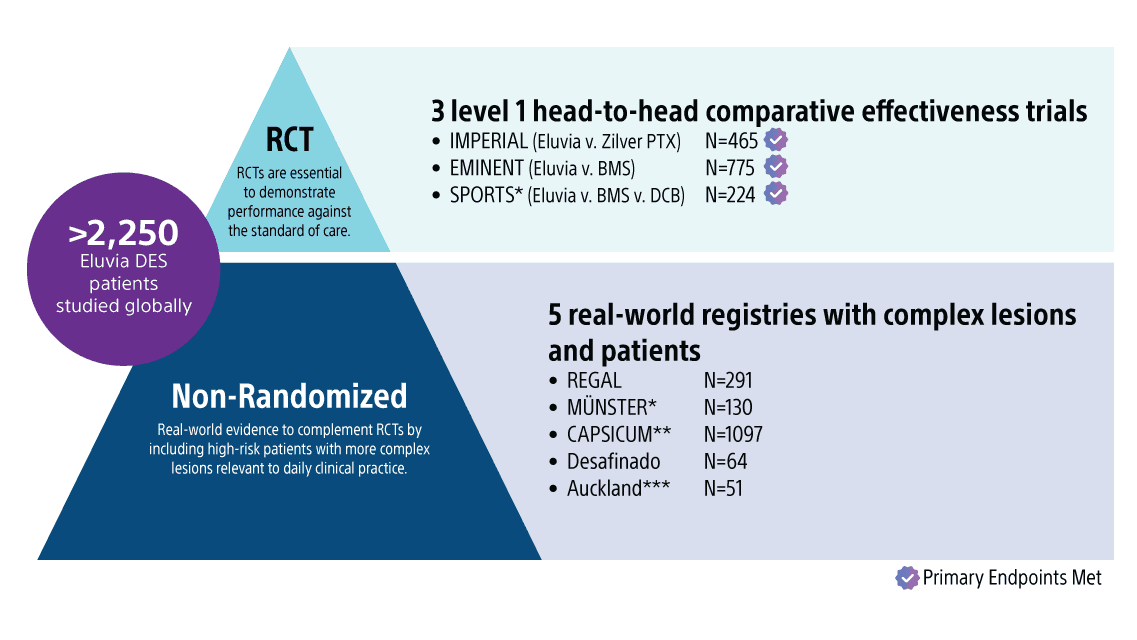
Featured clinical publications
October 2023
SPORTS RCT
SPORTS is the first investigator-sponsored, core-lab adjudicated, prospective, multi-center, three-arm RCT comparing outcomes of Eluvia DES vs. BMS (investigator choice) vs. Drug-Coated Balloon (DCB) (SeQuent Please), 1:1:1, in TASC C/D lesions.5
The Eluvia arm in the SPORTS RCT had an average lesion length of 235mm and is one of the most complex cohorts studied for the stent to date.
August 2018
IMPERIAL RCT
IMPERIAL is the first head-to-head Randomized Control (2:1) comparing a polymer-coated, paclitaxel-eluting stent (Eluvia DES) versus polymer-free, paclitaxel-coated stent (Zilver PTX) for endovascular femoropopliteal lesions up to 140mm in length; multi-center, single-blind; core lab adjudicated.
October 2022
EMINENT RCT
EMINENT1 is the largest Randomized Controlled Trial (RCT) (2:1) comparing Eluvia™ Drug-Eluting Vascular Stent System to self-expanding bare metal stents (BMS) for superficial femoral artery/proximal popliteal artery (SFA/PPA); EU multi-center; superiority trial; core lab adjudicated.
References:
1. EMINENT RCT 1-Year results presented by Yann Gouëffic, MD. VIVA 2021
2. EMINENT Trial: A global randomized controlled multi-center trial with 2:1 randomization of the Eluvia™ Drug-Eluting Stent against commercially-available Self-Expanding Bare Nitinol Stents, single-blind, superiority design; independent core lab adjudication. Primary Endpoint: 1-Year Binary Primary Patency rate of 83.2% in the Eluvia arm vs. 74.3% in the Bare-Metal Stenting arm (p-value = 0.0077).
3. In EMINENT, primary sustained clinical improvement was defined as an improvement (decrease) by at least 1 Rutherford category, without TLR.
4. Gray W. 2-year Outcomes from the IMPERIAL Randomized Head to Head Study of Eluvia DES and Zilver PTX; LINC 2020, Leipzig Jan 28,2020.
5. Tepe G. SPORTS. TCT 2023, October 24, 2023, San Francisco, CA.
| Stent length | ||||||
|---|---|---|---|---|---|---|
| 40 mm | 60 mm | 80 mm | 100 mm | 120 mm | 150 mm | |
| 6 mm | 6F | 6F | 6F | 6F | 6F | 6F |
| 7 mm | 6F | 6F | 6F | 6F | 6F | 6F |
Ordering information
| Description | UPN | GTIN |
|---|---|---|
| Eluvia 6 mm x 40 mm x 130 cm | H74939294600410 | 08714729876571 |
| Eluvia 6 mm x 60 mm x 130 cm | H74939294600610 | 08714729876588 |
| Eluvia 6 mm x 80 mm x 130 cm | H74939294600810 | 08714729876595 |
| Eluvia 6 mm x 100 mm x 130 cm | H74939294601010 | 08714729876601 |
| Eluvia 6 mm x 120 mm x 130 cm | H74939294601210 | 08714729876618 |
| Eluvia 6 mm x 150 mm x 130 cm | H74939294601510 | 08714729876625 |
| Eluvia 7 mm x 40 mm x 130 cm | H74939294700410 | 08714729876694 |
| Eluvia 7 mm x 60 mm x 130 cm | H74939294700610 | 08714729876700 |
| Eluvia 7 mm x 80 mm x 130 cm | H74939294700810 | 08714729876717 |
| Eluvia 7 mm x 100 mm x 130 cm | H74939294701010 | 08714729876724 |
| Eluvia 7 mm x 120 mm x 130 cm | H74939294701210 | 08714729876731 |
| Eluvia 7 mm x 150 mm x 130 cm | H74939294701510 | 08714729876748 |
Available now in 150mm length. Ask your sales representative about Eluvia 150.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.