WATCHMAN FLX™
Left Atrial Appendage Closure Device
WATCHMAN FLX is FDA APPROVED for use in nonvalvular atrial fibrillation patients who are eligible for anticoagulation therapy.
Built on the most studied and implanted LAAC device in the world, WATCHMAN FLX is designed to advance procedural performance and safety while expanding the treatable patient population.
Key Resources
Explore
PINNACLE FLX Clinical Study
Study Design
- 400 patient, 29 US site, single arm, non-randomized trial evaluating WATCHMAN FLX for non-inferiority to safety and efficacy performance goals based on the WATCHMAN™ device.
- Follow-up: 45 days (+TEE), 6 months, 12 months (+TEE), 18 months, and 24 months
- Patient Characteristics: Average CHA2DS2-VASc of 4.2±1.5, Average HAS-BLED of 2.0±1.0
- Post Implant Drug Regimen: NOAC/ASA for 45 days, Clopidogrel/ASA to 6 months, ASA post 6 months
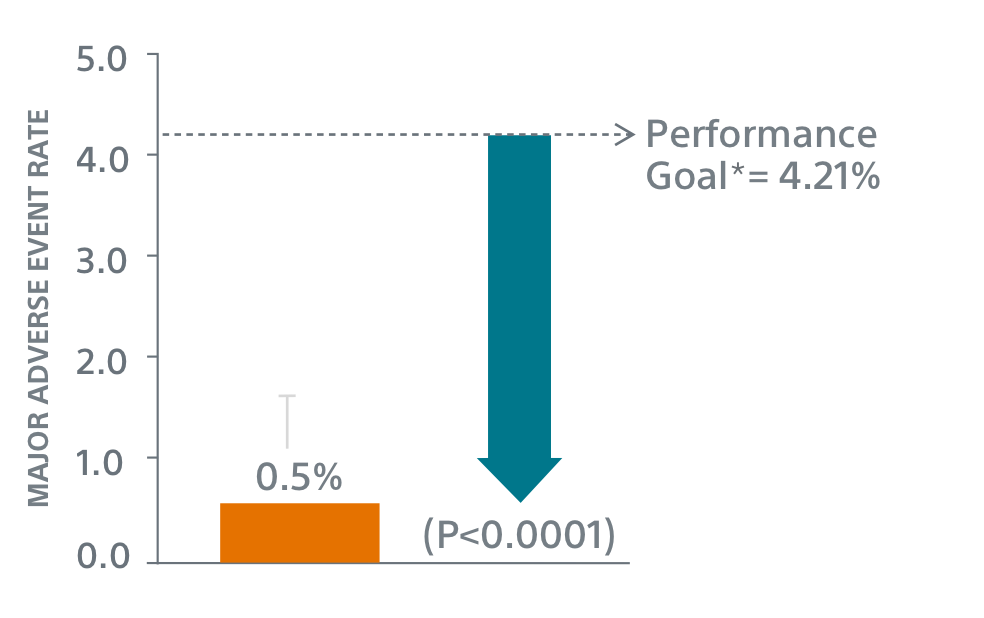
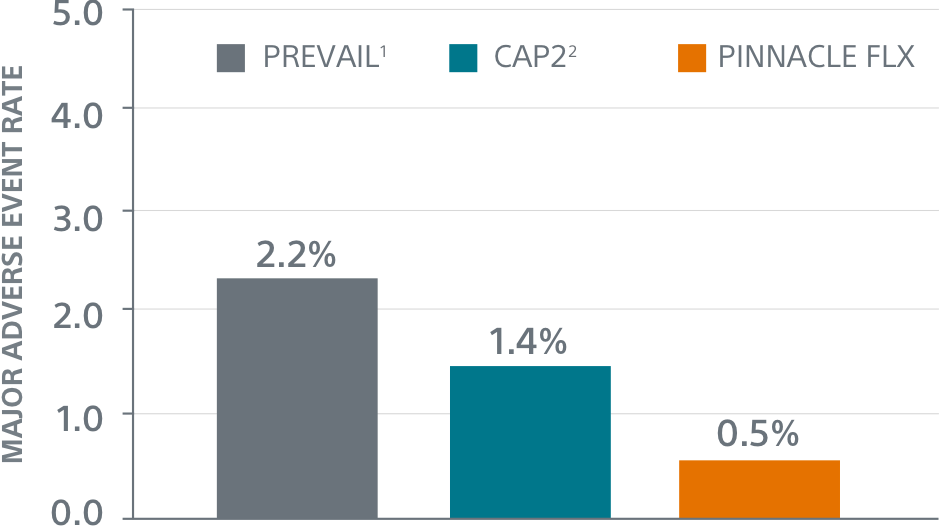
- Primary Safety Endpoint: All-cause death, ischemic stroke, systemic embolism, or device- or procedure-related adverse events requiring surgery or major endovascular intervention within 7 days following the procedure or by hospital discharge, whichever is later.
- Primary Efficacy Endpoint: The rate of effective LAA closure defined as any peri-device flow ≤5mm demonstrated by TEE at 12 months
- Secondary Efficacy Endpoint: The occurrence of ischemic stroke or systemic embolism at 24 months from the time of enrollment
- Inclusion/exclusion criteria is consistent with WATCHMAN clinical study inclusion/exclusion criteria. Patients must be eligible for short-term NOAC vs warfarin in previous clinical studies.
Primary Safety Endpoints1

Primary Efficacy Endpoints1
The rate of effective LAA closure defined as any peri-device flow ≤5mm demonstrated by TEE at 12 months.
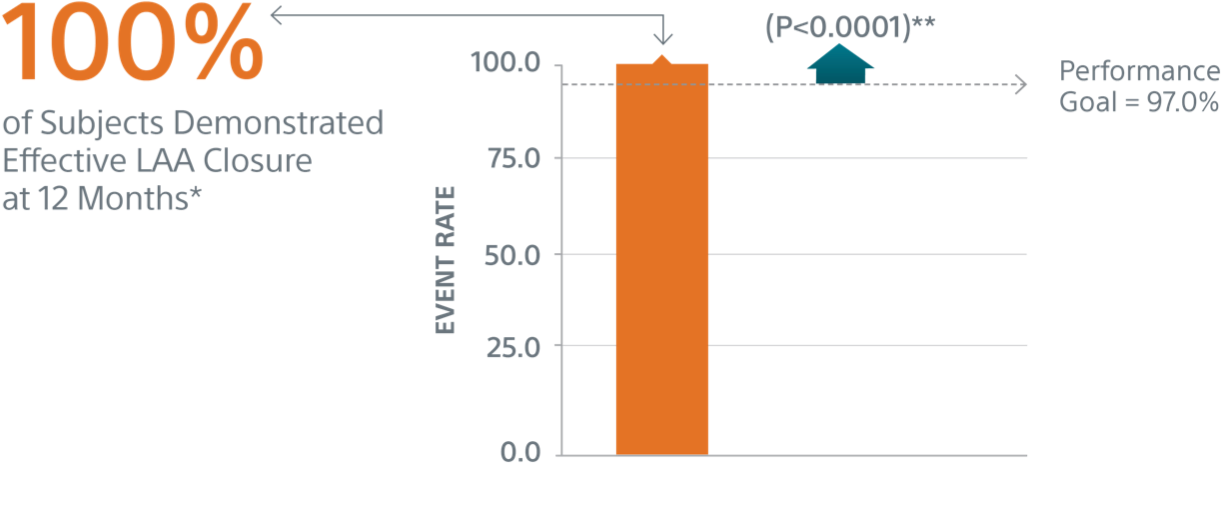
** Performance goal pased on the rates observed in PREVAIL(1) and CAP2(2), minus a clinically relevant delta
Procedure Performance1

NOAC Discontinuation1
2 Holmes, DR., et al, JACC 2019.
Designed to Treat Widest Range of Patient Anatomies
WATCHMAN FLX sizes range from (20mm - 35mm)