Trial Rationale
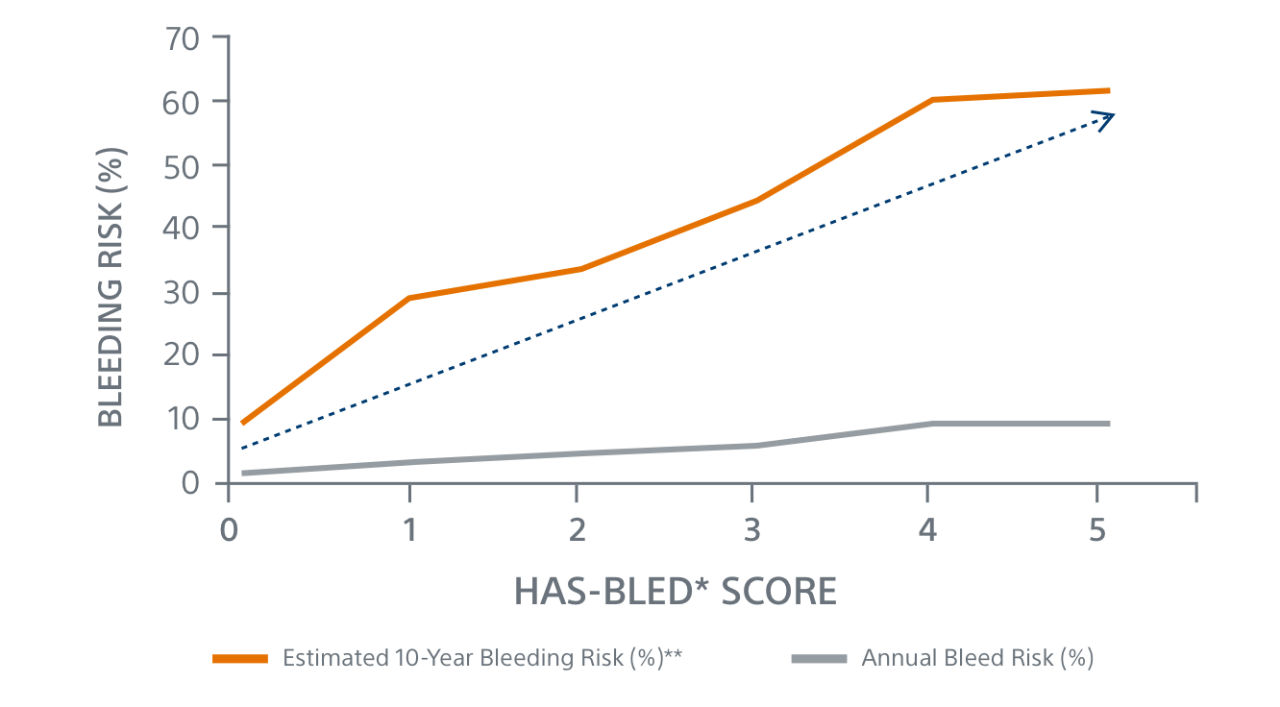
**Assumes constant risk despite increasing age, and bleeding risk is independent from bleeding risk in previous years.
The CHAMPION‑AF clinical trial seeks to demonstrate WATCHMAN FLXTM is a first‑line stroke risk reduction therapy for most NVAF patients.
With this trial, Boston Scientific expands its LAAC leadership with unmatched clinical evidence intended to demonstrate WATCHMAN FLX is the optimal choice versus NOAC for lifelong stroke risk reduction.
Why LAA Closure?
With ~40% of oral anticoagulant (OAC) indicated patients left unprotected from stroke due to dissatisfaction and non-adherence with OACs, CHAMPION‑AF seeks to show WATCHMAN FLX as a first‑line option for stroke risk-reduction in most NVAF patients.7
Over 500,000 patients have already been successfully treated with WATCHMANTM LAAC technology.
Why CHAMPION-AF?


Find a Trial Site
Boston Scientific has selected US sites to be CHAMPION-AF investigational centers. If you have a candidate in mind, view the active trial list provided by clinicaltrials.gov, and reach out to a study physician near you to discuss next steps for referral.
1. Colilla S, Crow A, Petkun W, Singer DE, Simon T, Liu X. Estimates of current and future incidence and prevalence of atrial fibrillation in the u.S. Adult population. The American journal of cardiology. 2013;112:1142-1147
2. Fuster V, Ryden LE, Cannom DS, Crijns HJ, Curtis AB, Ellenbogen KA, et al. Acc/aha/esc 2006 guidelines for the management of patients with atrial fibrillation—executive summary: A report of the american college of cardiology/american heart association task force on practice guidelines and the european society of cardiology committee for practice guidelines (writing committee to revise the 2001 guidelines for the management of patients with atrial fibrillation). J Am Coll Cardiol. 2006;48:854-906
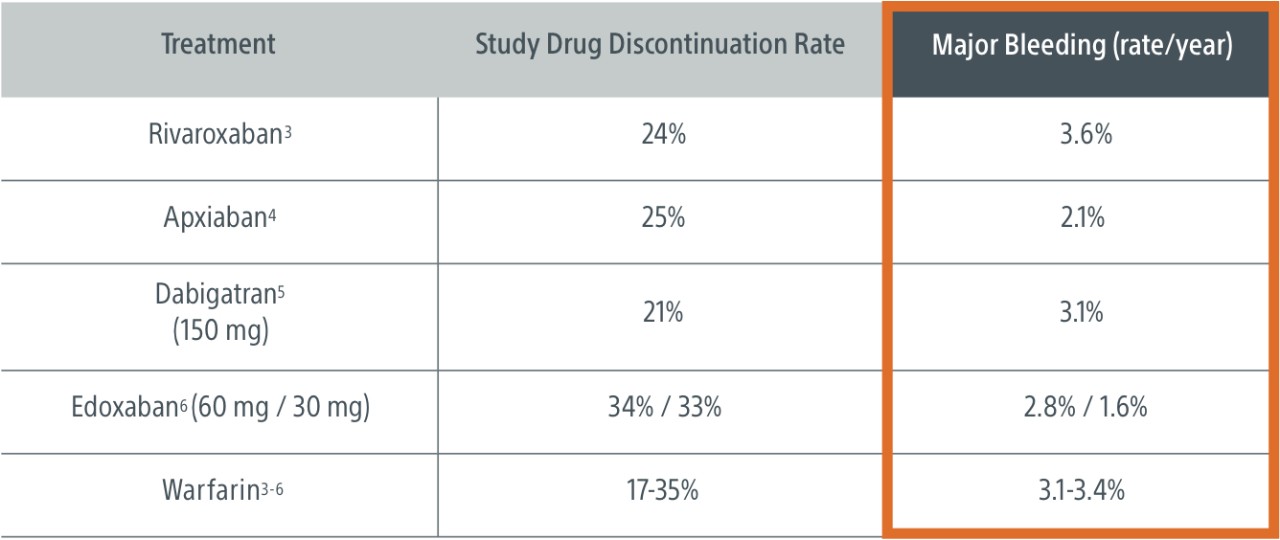
3. Patel, M. NEJM 2011; 365(10): 883-891.
4. Granger, C. NEJM 2011; 365(11): 981-992.
5. Connolly, S. NEJM 2009; 361(12): 1139-1151.
6. Giugliano, R. NEJM 2013; 369(22): 2093-2104.
7. Marzec, et al. JACC 2017: 69(20): 2475-2484.

