See more. Know more. Do more.
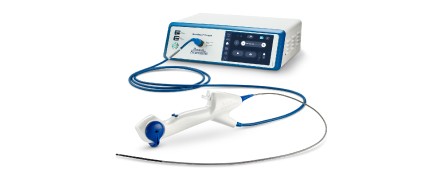
The LithoVue™ Elite Single-Use Digital Flexible Ureteroscope System is the first ureteroscope with intrarenal pressure (IRP) monitoring, giving you the power to make informed, real-time clinical decisions in kidney stone management.
Find out how the next-gen LithoVue Elite System can help you:
Sign up for up-to-date news and information about the next-gen LithoVue Elite System.
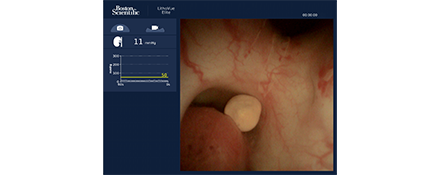
See more with enhanced image quality
Know more by monitoring IRP in real-time.
Why intrarenal pressure matters
Routine measurement of IRP is not currently performed during ureteroscopy procedures. Complications of elevated IRP may include:
- Pain1,2
- Renal damage and pathological changes1,3,4
- Systemic inflammatory response syndrome3,5
- Fluid absorption1,3,6-8
- Fever3,5
- Infection1,3,5,9
- Sepsis1,3,5,9
- Pyelovenous backflow1,3,6-8
Understanding IRP as a predictor of complications has been neglected;3 however, the LithoVue Elite System unlocks the potential to improve our understanding of the impact of elevated IRP on patient outcomes.
Do more with direct control and OR integration.
Take control of image and video capture
The LithoVue Elite System puts direct control of image and video capture from the sterile field in your hands. Programmable buttons on the scope handle enable you to easily record and save data without the need to coordinate with staff.
Seamlessly integrate into your OR
The StoneSmart™ Connect Console, LithoVue Elite’s compact processing unit, integrates into your operating room or existing mobile visualization tower and is designed to reduce clutter, minimize capital footprint, and simplify switching between cystoscope and ureteroscope. The endoscopic image is displayed on your existing OR monitors to help to minimize distraction and operating room logistics.
The LithoVue Elite System is the first device that has been designed on Boston Scientific’s next-generation StoneSmart™ technology platform. The system has built-in support for potential interoperability with future Boston Scientific devices.

Peer-to-peer education on StoneSmart.com
Bio-based plastic handles
LithoVue Elite Ureteroscope handles are made primarily with bio-based plastic material. Up to 85% of the material used in the handles comes from renewably sourced raw material.10* Using plastic made from renewably sourced materials instead of conventional fossil-based plastics offers a more renewable alternative without compromising scope performance.
To learn more about bio-based plastics, view the FAQ.
Transitional Pass-Through (TPT) Payment
Effective January 1, 2023, U.S. Centers for Medicare & Medicaid Services (CMS) has created a Transitional Pass-Through (TPT) payment category which describes single-use endoscopes, including the LithoVue™ Elite Single-Use Digital Flexible Ureteroscope, under the Medicare hospital outpatient prospective payment system (OPPS).19 Hospital Outpatient Departments and Ambulatory Surgery Centers may obtain additional, separate Medicare reimbursement for devices with TPT status, in addition to reimbursement for the procedure(s) furnished during the patient encounter.20 The intent of TPT payment is to facilitate Medicare beneficiary access to the advantages of innovative devices by allowing for adequate payment.21
A new device billing code is effective January 1, 2023, through December 31, 2025.
| HCPCS Code | Descriptor |
|---|---|
| C1747 | Endoscope, single-use (i.e., disposable), urinary tract, imaging/illumination device (insertable)22 |
Boston Scientific recommends the new C-Code be considered for use in charging and billing for single-use ureteroscopes, including the LithoVue Elite Single-Use Digital Flexible Ureteroscope.23 Payments will vary by facility based on hospital reported charges.
If you have additional questions, review the reimbursement coding reference guide or reach out to your local sales representative, or contact us.
Tools and resources
Stay informed
Sign up to receive up-to-date news and information about the next-gen LithoVue Elite System.