References:
1. Surapaneni, M., et al. "Designing Paclitaxel drug delivery systems aimed at improved patient outcomes: current status and challenges." International Scholarly Research Notices (2012).
2. The Business Research Company - Drug-Coated Balloons Global Market Report: https://www.thebusinessresearchcompany.com/report/drug-coated-balloons-global-market-report
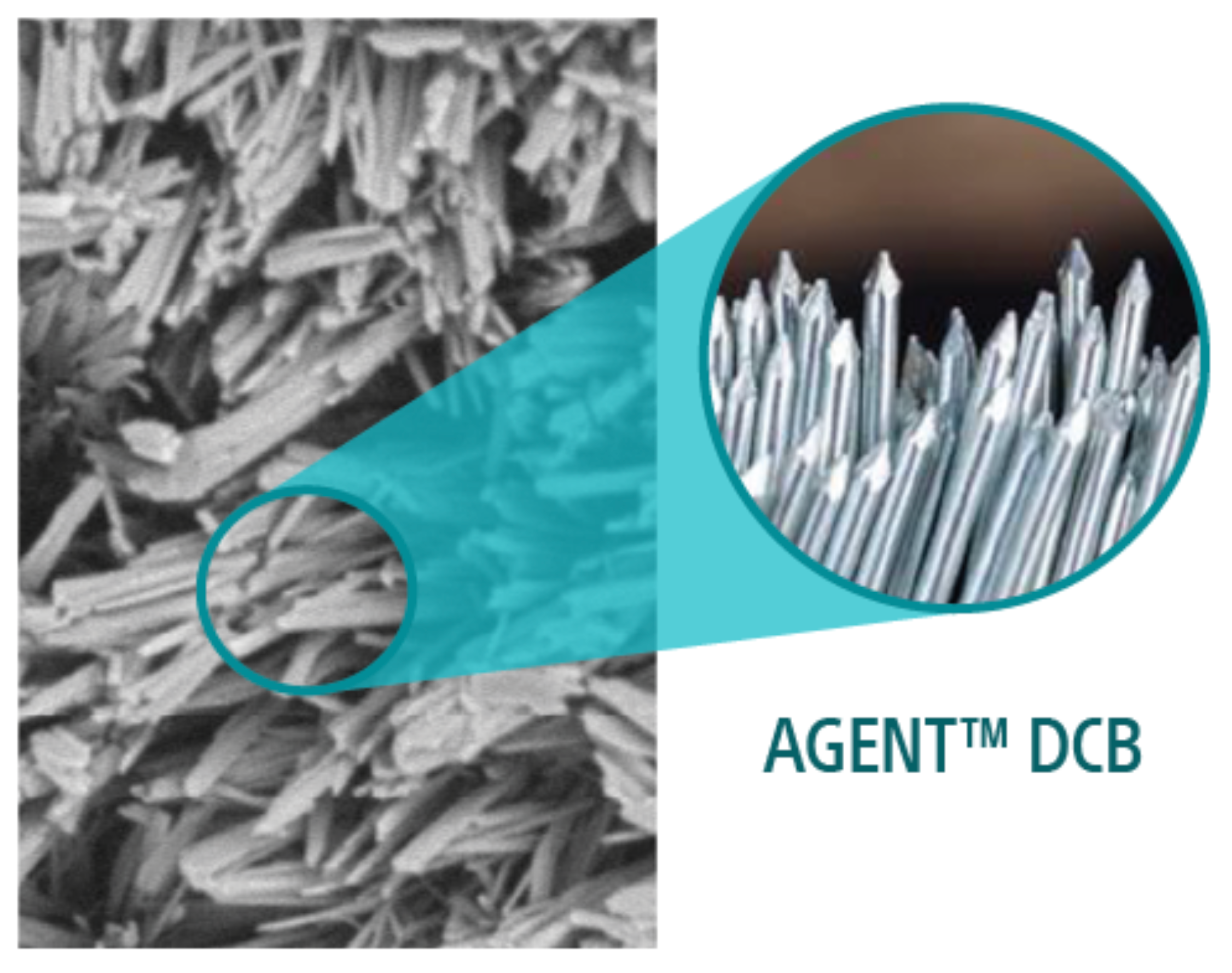
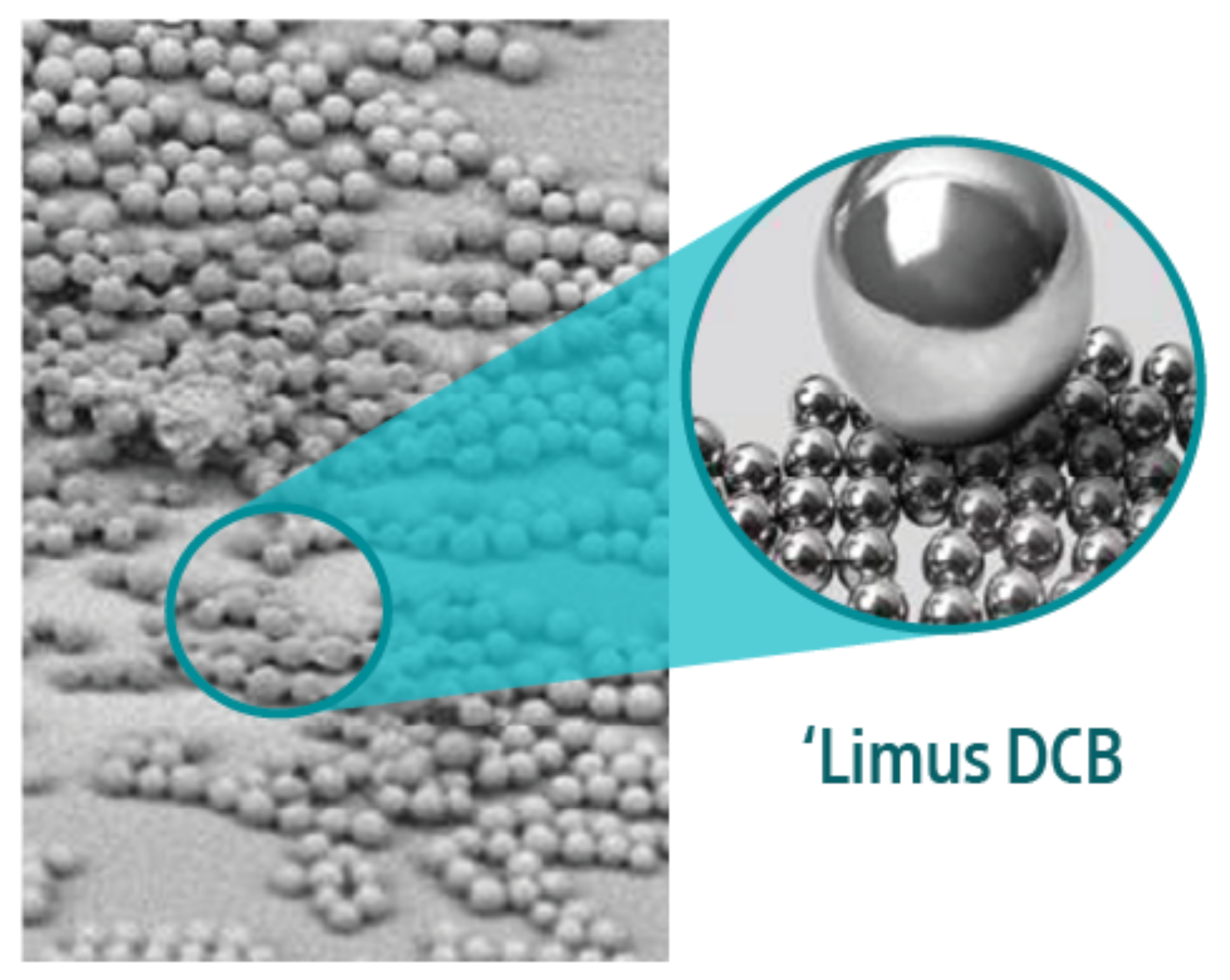
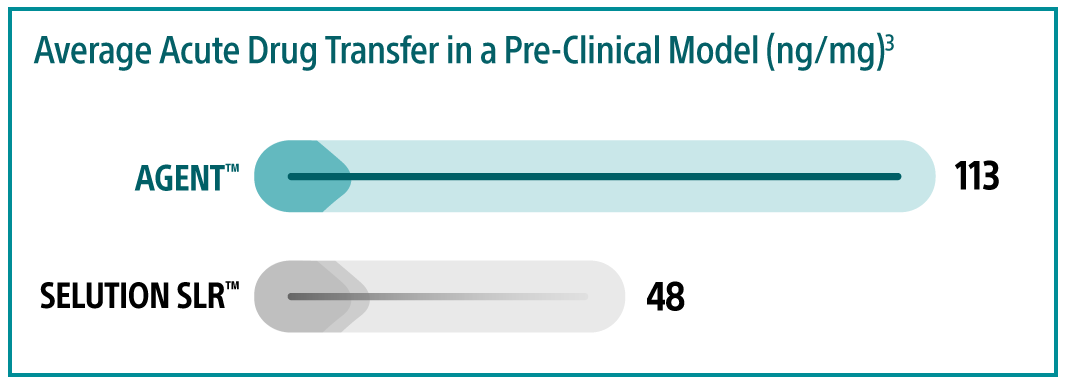
3. DCB pre-clinical testing performed by Boston Scientific Corporation. Data-on-file at BSC. In several independent studies, AGENT and Selution DCB were delivered to different coronary arteries. Treated vessel segments were analyzed at one-hour for drug concentration.
4. Surapaneni, M., et al. "Designing Paclitaxel drug delivery systems aimed at improved patient outcomes: current status and challenges." International Scholarly Research Notices (2012).
5. Data on file.
6. Chang, G., et al. Scientific Reports 9.1 (2019): 6839
7. Böhme, T., Noory, E., Beschorner, U., Macharzina, R., & Zeller, T. (2020). The Selution SLRTM drug-eluting Balloon System for the Treatment of Symptomatic Femoropopliteal Lesions. Future Cardiology, 17(2), 257–267. https://doi.org/10.2217/fca-2020-0085
*Each device was evaluated individually using a downstream particulate bench test designed to replicate physiological blood flow. Pre‑tracking images show coating before use; post‑tracking images show devices after simulated guide‑catheter tracking, prior to inflation. Bench Test results may not necessarily be indicative of clinical performance. The testing was performed by Boston Scientific. Data on file.
Selution SLR™ is an investigational device and is limited by U.S. law to investigational use.