Medical Specialties > Interventional Cardiology > Coronary Interventions > PCI Product Portfolio > AGENT IDE Clinical Trial > Understanding ISR
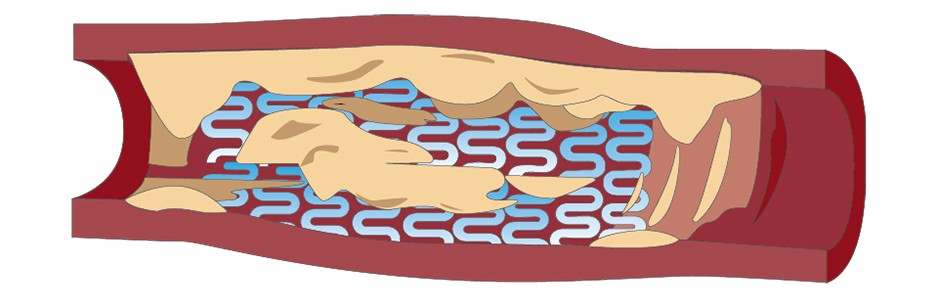
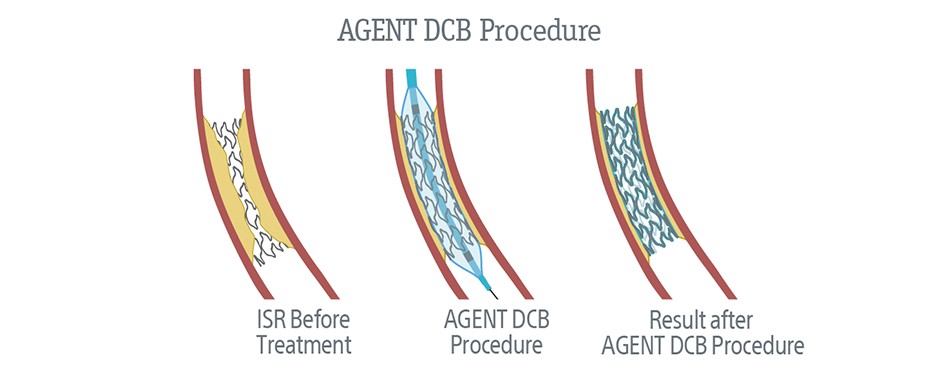

If you have been diagnosed with ISR and you are interested in learning more about the AGENT IDE clinical trial, discuss with your doctor.
