Boston Scientific accounts are for healthcare professionals only.

FARAWAVE™ NAV Pulsed Field Ablation Catheter
Configure or select a product to continue to order
- Overview
- Clinical data
- Technical specifications
- Ordering information
- Training
- Resources
How it works
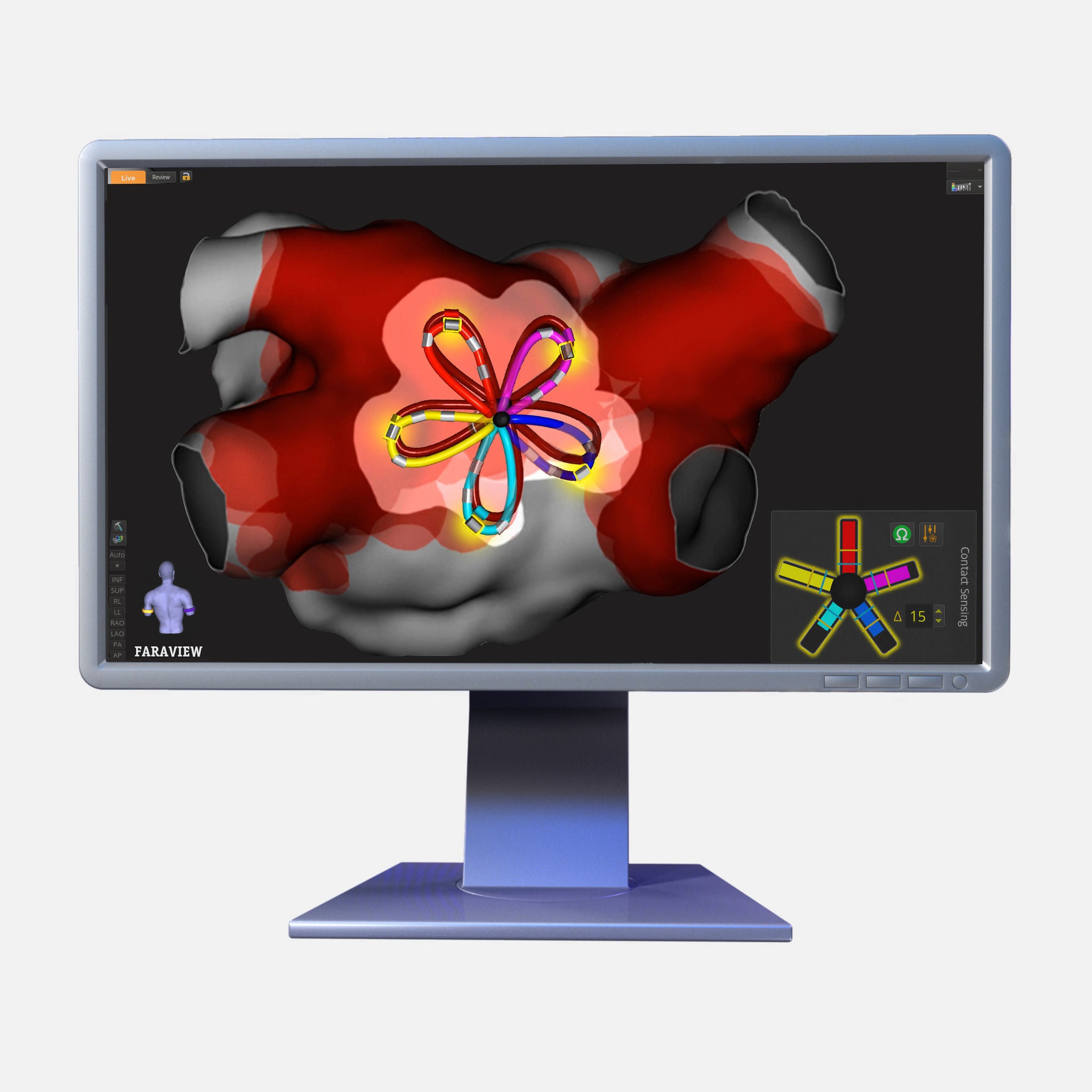
The FARAWAVE NAV PFA Catheter, paired with the FARAVIEW Software Module, enables a single-catheter solution to map and ablate supporting a more predictable, consistent workflow. Experience dynamic visualization of catheter placement, shape, and rotation while minimizing the need for exchanges.
Why choose the FARAWAVE NAV PFA Catheter
- Nav-enabled single-catheter solution to map and ablate supporting a more consistent workflow, while minimizing exchanges
- Dynamic visualization of catheter shape, position, and rotation to support potential fluoroscopy reduction
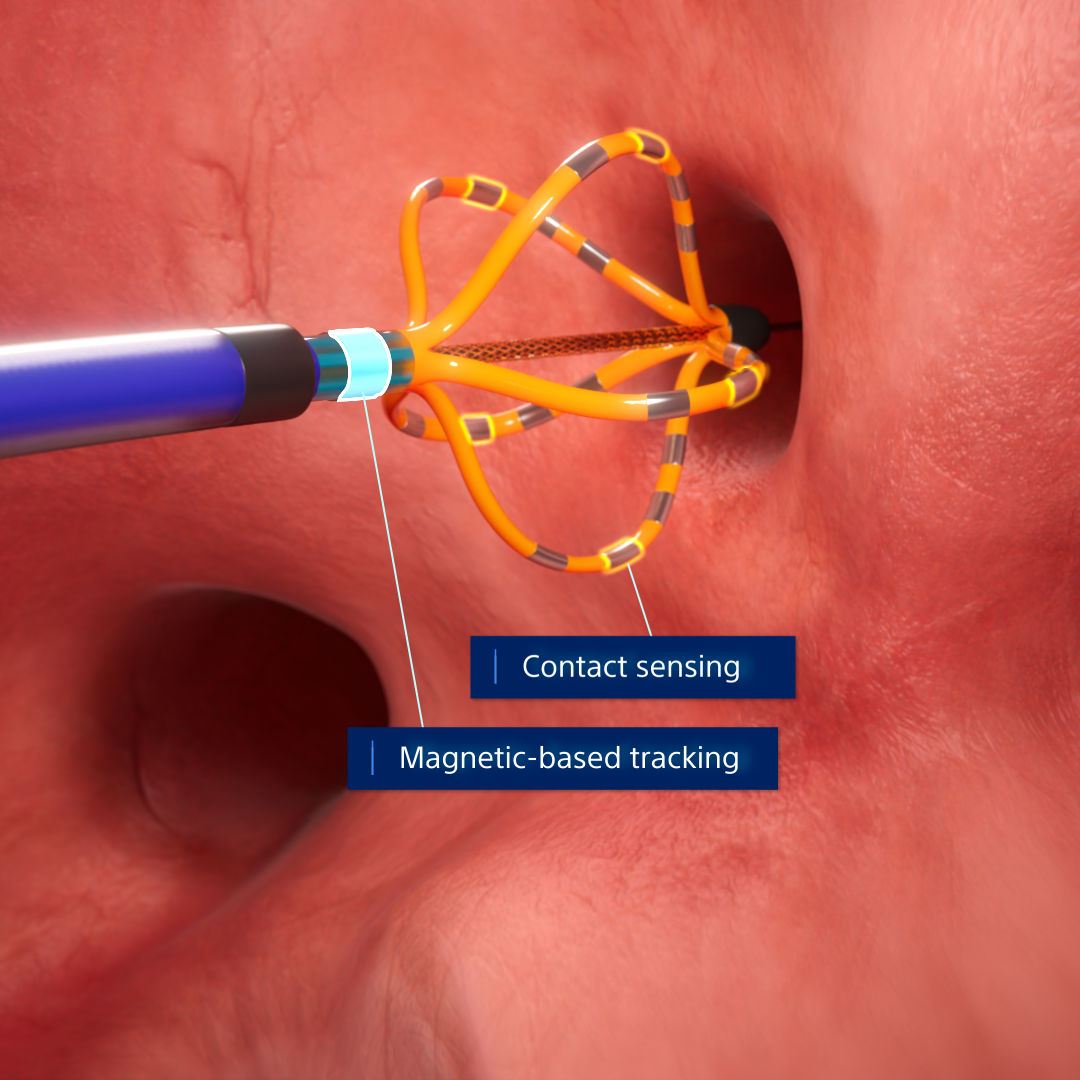
- Shaft electrode enables sheath detection and renders all shapes of the FARAWAVE NAV Catheter
- Contact sensing visualized on the FARAVIEW Software Module provides real-time contact information for the FARAWAVE NAV PFA Catheter
Reduce catheter exchanges and streamline PFA workflows
With the magnetically-tracked FARAWAVE NAV PFA Catheter integrated with the FARAVIEW Software Module, you can collect geometry, ablate and confirm workflows—limiting the need for additional catheter exchanges.
FIELDTAG™ Technology and contact sensing provide deeper insights into estimated electric field volume, tissue proximity, and ablation overlap.
Enhanced localized mapping
Software advancements enable enhanced map quality for improved mapping visualization, including pre- and post-ablation assessment, while maintaining the same proven single-catheter workflow.
OPAL 7.0 Software: Enhance map quality and planning and assessment of therapy delivery during FARAPULSE™ PFA workflows on the OPAL HDx™ Mapping System.

FARAPULSE
The most proven PFA platform
Discover what peers are saying about FARAVIEW
Hear firsthand from physicians as they share their excitement about the transformative FARAVIEW mapping experience.
Required products
Clinical data
DISRUPT-AF real-world registry1
Safe, efficient single-catheter map and ablate workflow with the FARAWAVE™ NAV Pulsed Field Ablation (PFA) Catheter, visualized with FARAVIEW™ Software Module on the OPAL HDx™ Mapping System. The DISRUPT-AF real-world registry (n=477) showed a 0.2% catheter-related complication event rate, predictable procedures and an average fluoroscopy time of 3.8 minutes.
NAVIGATE-PF trial2
The NAVIGATE-PF clinical trial was a multi-center, first-in-human feasibility study that assessed the acute and chronic accuracy of FIELDTAG™ Technology on the FARAVIEW™ Software Module in visualizing pulsed field energy delivery by the FARAWAVE™ NAV catheter. The study included two phases:
Phase I (acute assessment):
- Phase 1 included 30 patients that underwent PV isolation, with 30% also receiving posterior wall ablation.
- PFA markers were placed during PFA application and high-quality, post-ablation HD maps with INTELLAMAP ORION™ Mapping Catheter were completed in 15 pts. PFA markers were overlaid on the HD map and contours were drawn at the outer border of the low voltage areas (defined as ≤0.5mV) where at least 2 overlapping PFA markers were dropped.
Phase II (chronic assessment):
- Included 20 patients at 1 center to evaluate chronic lesion durability and alignment between acute PFA tag locations and chronic low voltage borders.
NAVIGATE-PF3: 60 day remap
60-day remapping after FARAWAVE NAV PFA Catheter ablation showed 95% (63/66) PV durability and 86% (12/14) PW durability.
FARAWAVE NAV PFA Catheter technical specifications
| Feature | Specifications |
| Catheter type | Over-the-wire |
| Shaft diameter | 12F |
| Distal diameter | 31 mm; 35 mm |
| Usable length | 115 cm |
| Number of electrodes | 20 |
| Guidewire compatibility | 0.035” diameter, 180 cm minimum length |
| Sheath compatibility | FARADRIVE Steerable Sheath, 74 cm length REF M004PF21M402 (USA) |
FARAWAVE NAV PFA Catheter ordering information
| Product description | Reference number |
| FARAWAVE NAV PFA Catheter 31 mm (distal diameter) | M004PF41M411 |
| FARAWAVE NAV PFA Catheter 35 mm (distal diameter) | M004PF41M412 |
Accessories
| Product description | Reference number |
| PFA Rx Gen 2 Connection Cable for FARAWAVE NAV | M004PF41M444 |
| Merit inqWire | IQ35F180J1O5RS |
Online education
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Brochures
Additional FARAPULSE PFA Platform resources
Find product brochures, spec sheets, clinical data compilations and more on our FARAPULSE resources page.
*Baylis Medical Company Radiofrequency Puncture Generator RFP-100A. Baylis Medical Company is a wholly owned subsidiary of Boston Scientific Corporation.
References:
1. Nair, D., et al. (2026) Clinical experience with map-and-ablate workflows enabled by the second-generation catheter: findings from the DISRUPT-AF Registry. Presented at AF Symposium 2026.
2. Reddy, V. et al. (2025). Visualization and chronic assessment of electrical field tags for pulsed field ablation of pulmonary veins with the second-generation pentaspline catheter. Presented at HRS 2025.
3. Reddy, V. Y., et al. (2026). Chronic lesion prediction of pulsed field ablation using dynamic shape visualization and tags with the second-generation pentaspline catheter. Presented at AF Symposium 2026.