Boston Scientific accounts are for healthcare professionals only.





Vercise Genus™ Deep Brain Stimulation (DBS) System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical data
- Technical specifications
- Ordering Information
- Training
- Resources
Redefining Deep Brain Stimulation with innovation
Easily customize therapy to match each patient's needs with the Vercise Genus DBS system.
How it works
Choose from five small, contoured rechargeable and non-rechargeable MR-Conditional IPGs designed for patient comfort and convenience, with the most therapy options** available.
When Boston Scientific IPGs are combined with Image Guided Programming using Illumina 3D™ technology and Vercise™ Cartesia™ X and Vercise™ Cartesia™ HX Directional Leads, clinicians can quickly personalize therapy in each patient’s specific anatomy for optimal patient outcomes.1,2
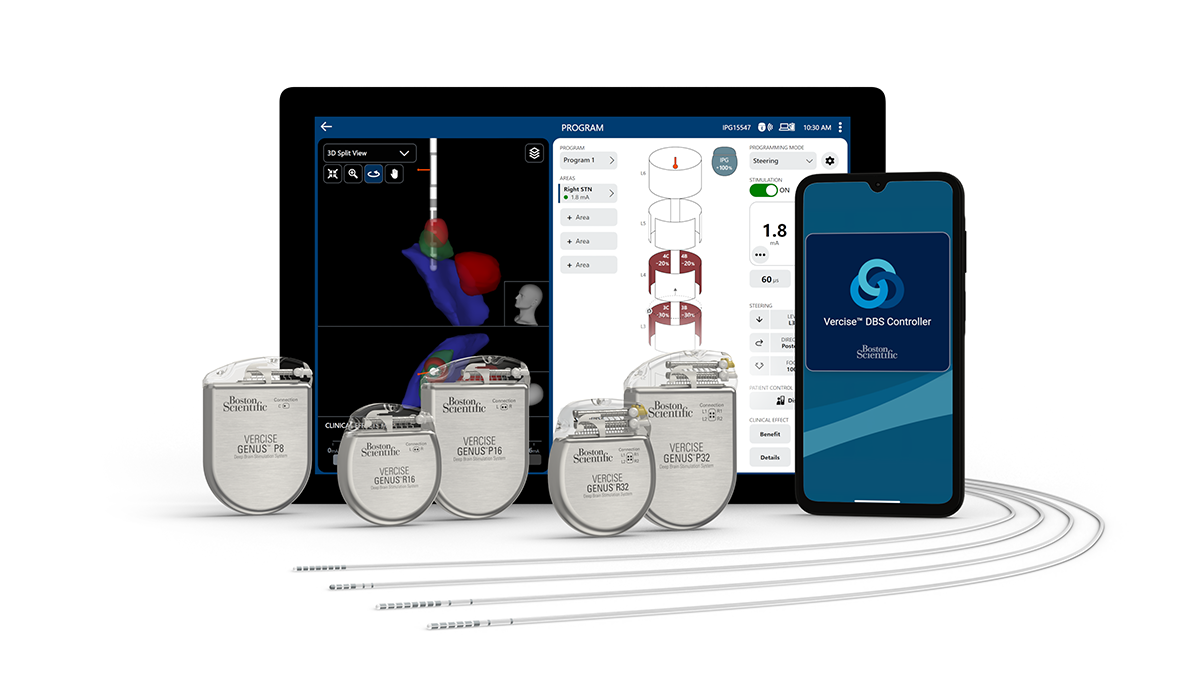
Discover the Vercise Genus DBS System

DBS batteries designed for patient comfort and convenience
With more than 250,000 patients implanted worldwide and 10 years of proven performance, Boston Scientific has a DBS battery you can depend on.3
Rechargeable options may provide more than 6 weeks of stimulation between recharging cycles.4 Non-rechargeable options include the Vercise™ P8 Battery, the only directional single channel primary cell in the market.
The most therapy options for DBS**
Boston Scientific’s DBS provides the most therapy options** to personalize patients’ DBS journeys today and in the future.

Monopolar

Bipolar

Bipolar Directional
DBS therapy options only available from Boston Scientific
Demonstrated to improve side-effect thresholds and improve efficacy5,6
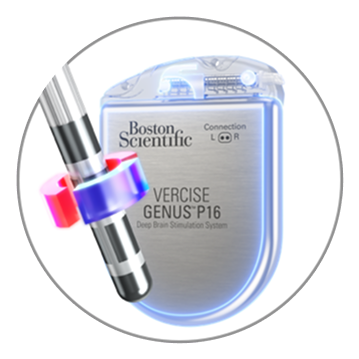
Semi-Bipolar
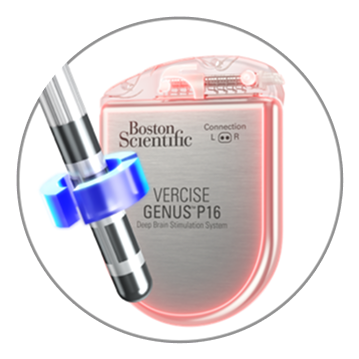
Anodic
Intelligent Image Guided Programming for seamless, personalized therapy
Years of science, minutes to program.1 Quickly generating a patient-specific stimulation plan, Image Guided Programming with Illumina 3D technology provides patients with precise therapy to achieve proven clinical outcomes.2
Image Guided Programming clinical data
Image Guided Programming has been proven to improve patient outcomes, preserve therapy stability and save programming time.1,2,7
Vercise Cartesia X and Vercise Cartesia HX Directional Leads
Precisely shape and steer stimulation where it’s needed most and leverage more programming flexibility by combining the power of Multiple Independent Current Control (MICC) with the Vercise Cartesia X and Vercise Cartesia HX Directional Leads.

68%
of patients see an improvement in symptoms or reduction in side effects by switching to a Boston Scientific DBS device (n=22).9
Why choose the Vercise Genus DBS System
The Vercise DBS System is supported by dozens of Parkinson’s disease studies, including the first multi-center, prospective, double-blind, randomized with sham control trial.8
Featured clinical publications
June 2020
The Boston Scientific DBS System demonstrated improved on-time and decrease in UPDRS III scores.8
Boston Scientific’s DBS therapy is supported by the first multi-center, prospective, double-blind, randomized with sham control trial and comprehensive Real World Outcomes studies in Parkinson’s disease.
November 2021
Boston Scientific’s Image Guided Programming makes it easier and more efficient to personalize every aspect of your patient’s DBS journey.
Image Guided Programming makes delivering directional stimulation easier and more efficient, with a 56% reduction in programming time.1
More clinical data
The Vercise™ Genus DBS System is supported by dozens of studies with hundreds of patients, showing clinical benefits to patients globally.
Technical specifications
| Vercise Genus R16 | Vercise Genus P16 | Vercise Genus R32 | Vercise Genus P32 | Vercise Genus P8 | |
|---|---|---|---|---|---|
| Volume | 20.1 cm3 | 34.9 cm3 | 21.6 cm3 | 36.6 cm3 | 34.9 cm3 |
| Thickness | 10.7 mm | 11.6 mm | 10.7 mm | 11.6 mm | 11.6 mm |
| Multiple Independent Current Control (MICC) | Yes | Yes | Yes | Yes | Yes |
| Directional Bipolar Stimulation | Yes | Yes | Yes | Yes | Yes |
| Anodic Stimulation | Yes | Yes | Yes | Yes | Yes |
| Communication | Bluetooth® | Bluetooth® | Bluetooth® | Bluetooth® | Bluetooth® |
| MR Conditional | Yes | Yes | Yes | Yes | Yes |
| Number of Ports | 2 | 2 | 4 | 4 | 1 |
| RemoteExpert Programming Support | Yes | Yes | Yes | Yes | Yes |
| Number of Contacts | 16 | 16 | 32 | 32 | 8 |
Ordering information
| MODEL NUMBER | DESCRIPTION |
|---|---|
| M365DB12160 | Vercise Genus™ R16 Implantable Pulse Generator Kit, 16 Contact |
| M365DB12320 | Vercise Genus™ R32 Implantable Pulse Generator Kit, 32 Contact |
| M365DB14160 | Vercise Genus™ P16 Implantable Pulse Generator Kit, 16 Contact |
| M365DB14320 | Vercise Genus™ P32 Implantable Pulse Generator Kit, 32 Contact |
| M365DB2202450 | Vercise™ Cartesia™ Directional Lead |
| M365DB14080 | Vercise GENUS P8 IPG Kit |
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Resources
To explore our comprehensive portfolio, visit BostonScientific.com/DBS
To get answers to your questions and additional information, contact a DBS sales representative
Find Vercise Genus DBS System procedural coding and reimbursement information for inpatient, outpatient, office and ASC codes
If you are a patient looking for more information on DBS, visit DBSandMe.com
References:
The Vercise Genus™ DBS System, Vercise Genus Mixed System with Vercise™ M8 Adapter, Vercise Genus Mixed System with Vercise™ Adapter S8, Vercise Gevia™ DBS System, and Vercise™ DBS Lead-only system (before Stimulator is implanted) provide safe access to full-body MRI scans when used with specific components and the patient is exposed to the MRI environment under specific conditions defined in the supplemental manual ImageReady™ MRI Guidelines for Boston Scientific DBS Systems.
** Information for competitive devices excerpted from the literature published by Medtronic (M982261A015 Rev A, M939241A051 Rev A, M013074C001 Rev B, M982097A013 Rev A, M13075C001 Rev B, M019192C002 Rev A) and Abbott (ARTEN600150429 - B, ARTEN600102238 - A, ARTEN600266398 -A, ARTEN600308953 -A, ARTEN600308947 -A), and
Schüpbach, Michael & Chabardes, Stephan & Matthies, Cordula & Pollo, Claudio & Steigerwald, Frank & Timmermann, Lars & Vandewalle, Veerle & Volkmann, Jens & Schuurman, P.. (2017). Directional leads for deep brain stimulation: Opportunities and challenges. Movement Disorders. 32. 10.1002/mds.27096.
Steffen, J. K., Reker, P., Mennicken, F. K., Dembek, T. A., Dafsari, H. S., Fink, G. R., Visser-Vandewalle, V., & Barbe, M. T. (2020). Bipolar Directional Deep Brain Stimulation in Essential and Parkinsonian Tremor. Neuromodulation: Technology at the Neural Interface, 23(4), 543–549. DOI: 10.1111/ner.13109
Reker, P., Dembek, T. A., Becker, J., Visser-Vandewalle, V., & Timmermann, L. (2016). Directional deep brain stimulation: A case of avoiding dysarthria with bipolar directional current steering. Parkinsonism & Related Disorders, 31, 156-158. https://doi.org/10.1016/j.parkreldis.2016.08.007
Kirsch, A. D., Hassin-Baer, S., Matthies, C., Volkmann, J., & Steigerwald, F. (2018). Anodic versus cathodic neurostimulation of the subthalamic nucleus: A randomized-controlled study of acute clinical effects. Parkinsonism & Related Disorders, 55, 61-67. https://doi.org/10.1016/j.parkreldis.2018.05.015.
Results from different clinical investigations are not directly comparable. Information provided for educational purposes only.
- Lange F, et al. (2021). Reduced Programming Time and Strong Symptom Control Even in Chronic Course Through Imaging-Based DBS Programming. Front. Neurol. 12:785529. doi: 10.3389/fneur.2021.785529
- Torres V, et al. Image-guided programming deep brain stimulation improves clinical outcomes in patients with Parkinson’s disease. NPJ Parkinsons Dis. 2024 Jan 27;10(1):29.
- Boston Scientific data on file.
- Niketeghad S et al. (2026) Characterizing Time to Depletion in Multiple Independent Current Control (MICC) Rechargeable Implantable Pulse Generators (IPGs) for Deep Brain Stimulation. Poster presented at: North American Neuromodulation Society (NANS); Jan 2026 Las Vegas, NV.
- Reker P, Dembek TA, Becker J, Visser-Vandewalle V, Timmermann L. Directional deep brain stimulation: A case of avoiding dysarthria with bipolar directional current steering. Parkinsonism Relat Disord. 2016 Oct:31:156–158.
- Kirsch AD, Hassin-Baer S, Matthies C, Volkmann J, Steigerwald F. Anodic versus cathodic neurostimulation of the subthalamic nucleus: A randomized-controlled study of acute clinical effects. Parkinsonism Relat Disord. 2018 Oct:55:61-67.
- Aldred JL et al. Sustained Therapeutic Benefits Using Image-Guided Programming at Activation of Deep Brain Stimulation for Parkinson's Disease. Mov Disord Clin Pract. 2025 Nov;12(11):1821-1830. doi: 10.1002/mdc3.70154. Epub 2025 Jun 5. PMID: 40474569; PMCID: PMC12625106.
- Vitek, Jerrold L et al. Subthalamic nucleus deep brain stimulation with a multiple independent constant current-controlled device in Parkinson's disease (INTREPID): a multicentre, double-blind, randomised, sham-controlled study. The Lancet Neurology, Volume 19, Issue 6, 491 - 50
- Ojukwu DI, Wang AR, Hornbeck TS, Lim EA, Sharrard J, Dhall R, Buch VP, Halpern CH. Conversion to Hybrid Deep Brain Stimulation System to Enable Multi-Contact Fractionation Can be Therapeutic. Mov Disord. 2022 Apr 7. doi: 10.1002/mds.29007. Epub ahead of print. PMID: 35393689.