Boston Scientific accounts are for healthcare professionals only.

Epic™ Vascular Self-Expanding Stent System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical Data
- Technical Specifications
- Ordering information
- Training
- Resources
Ideal for the Iliac
Designed for an optimal balance between flexibility, radial force, deployment accuracy and fracture resistance. The size matrix provides versaility without compromise.

Epic Stent System is designed for flexibility, deliverability and visibility
Featured clinical data
ORION US IDE Clinical Trial
A Boston Scientific Study of the Epic Self-Expanding Nitonol Stent System in the treatment of Atherosclerotic lesions in Iliac Arteries. To demonstrate similar patency across all TASC classifications.
3 year results
- 0 amputations at 3 years (106 subjects)
- 89.9% freedom from TLR at 3 years (143 Iesions)
Composite Success Rates
- 100% TechnicaI Success
(Per lesion) (166/166) - 99.2% Procedural Success
(Per subject) (124/125) - 95.3% Late Clinical Success at 12 months
(Per subject) (101/106)
12-month Primary Patency by TASC Classification
| Lesions at risk | All | 163.5 | 155 | 153 | 152.5 | 132 |
| A/B | 132.5 | 124 | 123 | 122.5 | 104 | |
| C/D | 27 | 27 | 26 | 26 | 25 |
12-month data
- 0% Ml at 30 days (0/111)
- 3.4% MAE at 9-months (4/117)
- 0% MI in-hospital (0/111)
- 5.4% TVR at 12-months (6/111)
- 0% Amputation of index limb at 12-months (0/111)
Epic Self-Expanding Nitonol Stent System
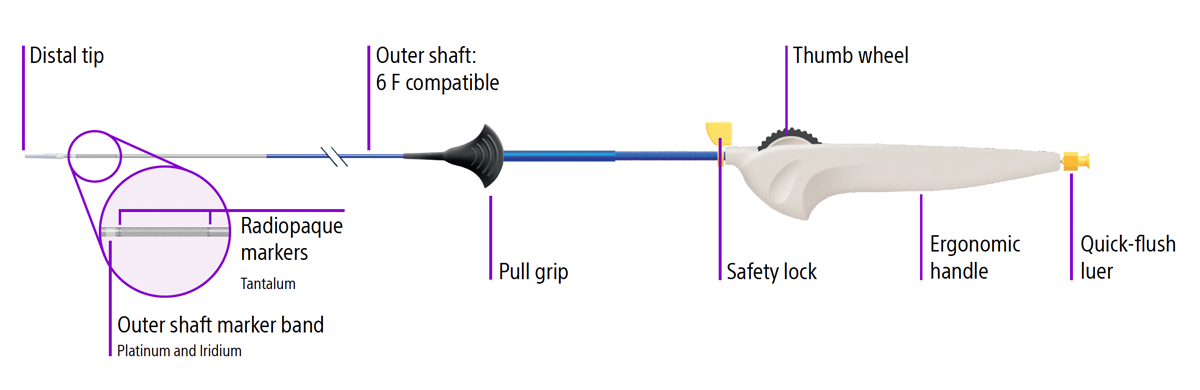
The Epic Stent System design features:
- Robust catheter
- 6F compatibile outer shaft
- Ergonomic handle
- Quick-flush luer
Ordering information
75 cm catheter working length
| UPN/Order Code | Catalog No. | GTIN | Unconstrained Stent Diameter (mm) | Unconstrained Stent Length (mm) | Sheath Compatibility (F) | Catheter Total Length (cm) | Indications | C-Code |
| H74939200062070 | 39200-06207 | 8714729805250 | 6 | 20 | 6 | 75 | 4 | C1876 |
| H74939200063070 | 39200-06307 | 8714729805274 | 6 | 30 | 6 | 75 | 4 | C1876 |
| H74939200064070 | 39200-06407 | 8714729805298 | 6 | 40 | 6 | 75 | 4 | C1876 |
| H74939200066070 | 39200-06607 | 8714729805335 | 6 | 60 | 6 | 75 | 4 | C1876 |
| H74939200068070 | 39200-06807 | 8714729805373 | 6 | 80 | 6 | 75 | 4 | C1876 |
| H74939200061070 | 39200-06107 | 8714729805212 | 6 | 100 | 6 | 75 | 4 | C1876 |
| H74939200061270 | 39200-06127 | 8714729805236 | 6 | 120 | 6 | 75 | 4 | C1876 |
| H74939200072070 | 39200-07207 | 8714729805410 | 7 | 20 | 6 | 75 | 4 | C1876 |
| H74939200073070 | 39200-07307 | 8714729805434 | 7 | 30 | 6 | 75 | 4 | C1876 |
| H74939200074070 | 39200-07407 | 8714729805458 | 7 | 40 | 6 | 75 | 4 | C1876 |
| H74939200076070 | 39200-07607 | 8714729805496 | 7 | 60 | 6 | 75 | 4 | C1876 |
| H74939200078070 | 39200-07807 | 8714729805533 | 7 | 80 | 6 | 75 | 4 | C1876 |
| H74939200071070 | 39200-07107 | 8714729805557 | 7 | 100 | 6 | 75 | 4 | C1876 |
| H74939200071270 | 39200-07127 | 8714729805397 | 7 | 120 | 6 | 75 | 4 | C1876 |
| H74939200082070 | 39200-08207 | 8714729805618 | 8 | 20 | 6 | 75 | 4 | C1876 |
| H74939200083070 | 39200-08307 | 8714729805632 | 8 | 30 | 6 | 75 | 4 | C1876 |
| H74939200084070 | 39200-08407 | 8714729805656 | 8 | 40 | 6 | 75 | 4 | C1876 |
| H74939200086070 | 39200-08607 | 8714729805694 | 8 | 60 | 6 | 75 | 4 | C1876 |
| H74939200088070 | 39200-08807 | 8714729805731 | 8 | 80 | 6 | 75 | 4 | C1876 |
| H74939200081070 | 39200-08107 | 8714729805571 | 8 | 100 | 6 | 75 | 4 | C1876 |
| H74939200081270 | 39200-08127 | 8714729805595 | 8 | 120 | 6 | 75 | 4 | C1876 |
| H74939200092070 | 39200-09207 | 8714729805755 | 9 | 20 | 6 | 75 | 4 | C1876 |
| H74939200093070 | 39200-09307 | 8714729805779 | 9 | 30 | 6 | 75 | 4 | C1876 |
| H74939200094070 | 39200-09407 | 8714729805793 | 9 | 40 | 6 | 75 | 4 | C1876 |
| H74939200096070 | 39200-09607 | 8714729805830 | 9 | 60 | 6 | 75 | 4 | C1876 |
| H74939200098070 | 39200-09807 | 8714729805878 | 9 | 80 | 6 | 75 | 4 | C1876 |
| H74939200091070 | 39200-09107 | 8714729805892 | 9 | 100 | 6 | 75 | 4 | C1876 |
| H74939200102070 | 39200-10207 | 8714729804970 | 10 | 20 | 6 | 75 | 4 | C1876 |
| H74939200103070 | 39200-10307 | 8714729804994 | 10 | 30 | 6 | 75 | 4 | C1876 |
| H74939200104070 | 39200-10407 | 8714729805014 | 10 | 40 | 6 | 75 | 4 | C1876 |
| H74939200106070 | 39200-10607 | 8714729805052 | 10 | 60 | 6 | 75 | 4 | C1876 |
| H74939200108070 | 39200-10807 | 8714729805090 | 10 | 80 | 6 | 75 | 4 | C1876 |
| H74939200101070 | 39200-10107 | 8714729805113 | 10 | 100 | 6 | 75 | 4 | C1876 |
| H74939200123070 | 39200-12307 | 8714729805137 | 12 | 30 | 6 | 75 | 4 | C1876 |
| H74939200124070 | 39200-12407 | 8714729805151 | 12 | 40 | 6 | 75 | 4 | C1876 |
| H74939200126070 | 39200-12607 | 8714729805199 | 12 | 60 | 6 | 75 | 4 | C1876 |
C-Code
The C-code used for this product is C1876, Stent, non-coated/non-covered with delivery system. C-codes are used for hospital outpatient device reporting for Medicare and some private payers.
Note: Boston Scientific is not responsible for the correct use of codes on submitted claims; this information does not constitute reimbursement or legal advice.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.




