SYNERGY™
Everolimus-Eluting Platinum Chromium Coronary Stent System
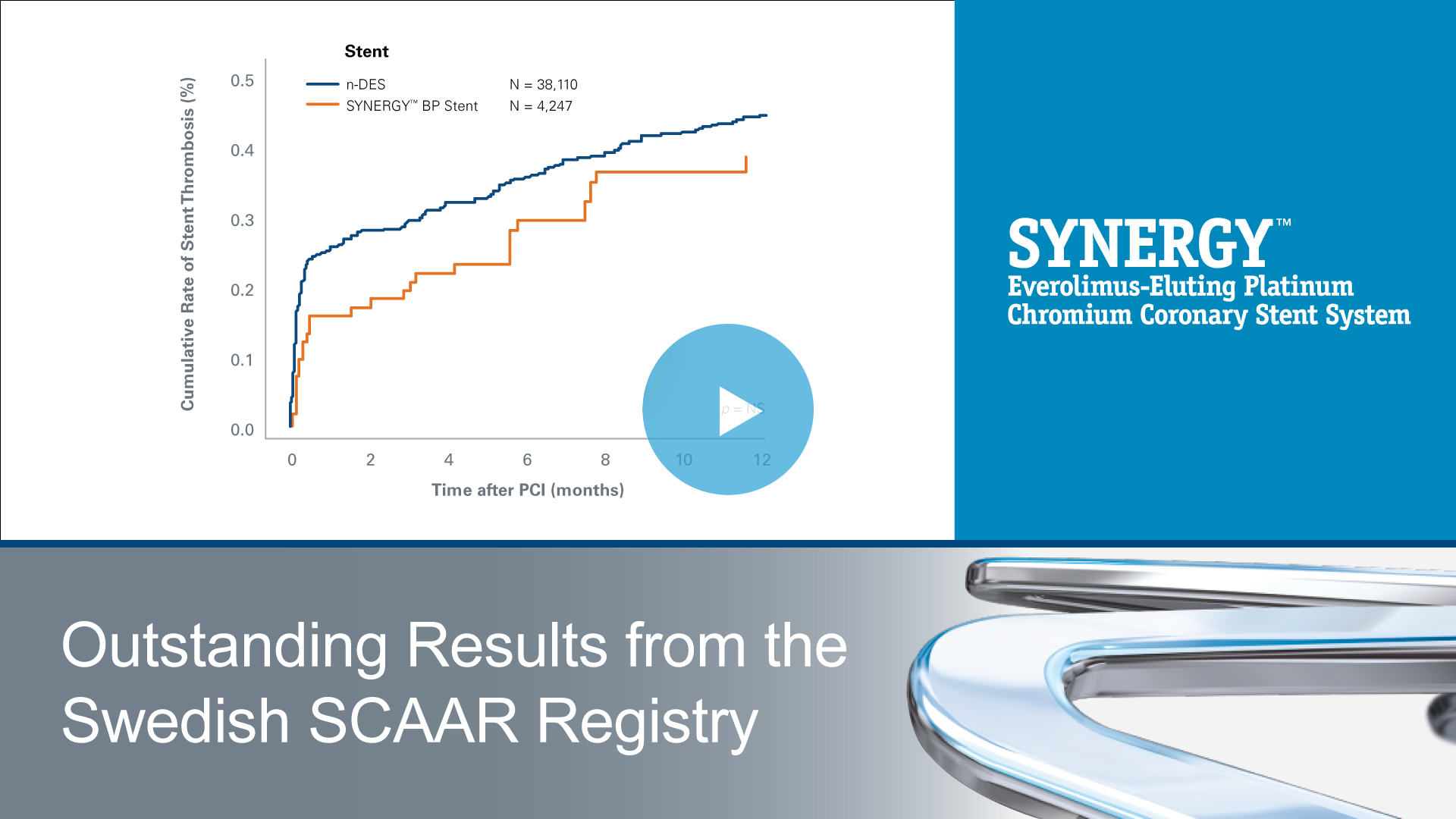
Watch Video: Outstanding Results from the Swedish SCAAR Registry
Boston Scientific accounts are for healthcare professionals only.
Create an account to access online training and education on EDUCARE, manage your customer profile, and connect with customer support and service teams.
My Boston Scientific account
Access your online applications and manage your customer profile.
Quick Links
Call customer care
Everolimus-Eluting Platinum Chromium Coronary Stent System
