Safe, Effective, Durable
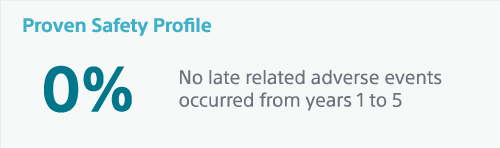
Rezūm™ Water Vapor Therapy is now the only in‐office BPH therapy with proven prostate volume reduction and long-term durability out to 5 years while preserving sexual function.1
The 5-year clinical trial, conducted by Kevin McVary, MD, and Claus Roehrborn, MD, was published in the Journal of Urology in September 2021.

rate through 5 years1

rate through 5 years1

procedure time1
“Rezūm Therapy has been embraced by urology practices, health technology assessment bodies, and urologic societies throughout the US and Europe. This adoption is attributed to the evidence of its clinical advantages, including sustained relief of LUTS, enhanced quality of life, and durability of treatment response.”1
Lasting Relief for Patients

Convective steam therapy is proven to reduce prostate volume, without thermal lesions occurring outside of the treatment zone.7 Unlike other MISTs, Rezūm™ Therapy is recommended by the AUA for treating middle lobe obstruction (MLO).8 And the 5-year study affirms a best-in-class surgical retreatment rate of just 4.4%.1
That’s why patients are satisfied with Rezūm Therapy. According to a 4-year single-center study of 255 men, 97% would recommend this treatment to a friend.9
The Results





Study Details
Overview
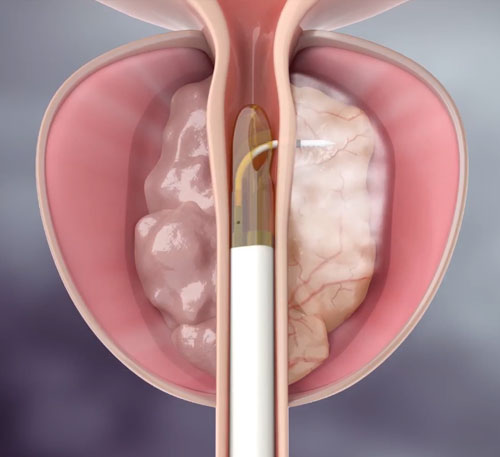
This was a multicenter (15 sites), randomized, sham-controlled trial of Rezūm Water Vapor Therapy in men with moderate-to-severe LUTS due to benign prostatic hyperplasia (BPH), with the inclusion of the final surgical and BPH medication retreatment rates. Thermal therapy involved injection of water vapor, using the Rezūm System, into obstructive tissue, including the middle lobe and/or enlarged central zone.
Patient Profile
197 subjects ≥ 50 years old
International Prostate Symptom Score (IPSS) | Maximum flow rate (Qmax) | Prostate volume |
| ≥13 | ≤ 15 ml/s | 30-80 cc |
(Thermal therapy with the Rezūm System: sham control rigid cystoscopy)
All Rezūm Therapies were completed in the office or ambulatory-surgery center.
Preserve Sexual Function with Rezūm Therapy
Sexual function can be important to patients. Rezūm Therapy patients experience durable LUTS relief while preserving sexual function.1
Results from a post-hoc analysis of the 5-year Rezūm II prospective, multi-center, randomized, blinded controlled trial suggests Rezūm Therapy does not cause clinically meaningful* declines in erectile function or ejaculatory function out to 5 years.10 This analysis was performed on all treated subjects who were sexually active at baseline with no other surgical or medical management for BPH during the 5-year study period.
* None of the patients in the study changed sexual function category (no dysfunction, mild, moderate, severe).
Leading Urologist Kevin McVary, MD, FACS, presents the final outcomes of the Rezūm Water Vapor 5-year pivotal study.
Request More Information
Get in touch with our team: Whether you’re looking to get started with Rezūm or have general questions about the product, procedure or clinical data, our team is here to help.
About Rezūm Therapy
Rezūm Water Vapor Therapy uses convective energy to remove obstructive prostate tissue, treating the cause of BPH. This treatment delivers targeted, controlled doses of the stored thermal energy in water vapor directly to the region of the prostate gland with the obstructive tissue causing the lower urinary tract symptoms (LUTS) secondary to benign prostatic hyperplasia (BPH).11
Related Content
Read the latest editorial from principal investigator Kevin T. McVary, MD, FACS, in the Physician’s Perspectives series.
Watch the full presentation from Kevin McVary, MD, FACS, on the final outcomes of the Rezūm Water Vapor 5-year pivotal study.
Read the September 2021 Urology Times interview with Kevin McVary, MD, FACS, on the ancillary 5-year study focused on erectile and ejaculatory outcomes.
Indications, Safety and Warnings
References
- McVary KT, Gittelman MC, Goldberg KA, et al. Final 5-year outcomes of the multicenter randomized sham-controlled trial of Rezūm water vapor thermal therapy for treatment of moderate-to-severe lower urinary tract symptoms secondary to benign prostatic hyperplasia. J Urol. 2021 Sep;206(3):715-24.
- Roehrborn CG, Barkin J, Gange SN, et al. Five-year results of the prospective randomized controlled prostatic urethral L.I.F.T. study. Can J Urol. 2017 Jun;24(3):8802-13.
- Zlotta AR, Giannakopoulos X, Maehlum O, et al. Long-term evaluation of transurethral needle ablation of the prostate (TUNA) for treatment of symptomatic benign prostatic hyperplasia: clinical outcome up to five years from three centers. Eur Urol. 2003 Jul;44(1):89-93.
- Rosario DJ, Phillips JT, Chapple CR. Chapple, Durability and cost-effectiveness of transurethral needle ablation of the prostate as an alternative to transurethral resection of the prostate when alpha-adrenergic antagonist therapy fails. J Urol. 2007 Mar;177(3):1047-51; discussion 1051.
- Hill B, Belville W, Bruskewitz R, et al. Transurethral needle ablation versus transurethral resection of the prostate for the treatment of symptomatic benign prostatic hyperplasia: 5-year results of a prospective, randomized, multicenter clinical trial. J Urol. 2004 Jun;171(6 Pt 1):2336-40.
- Miller PD, Kastner C, Ramsey EW, et al. Cooled thermotherapy for the treatment of benign prostatic hyperplasia: durability of results obtained with the Targis system. Urology. 2003 Jun;61(6):1160-4.
- Mynderse LA, Hanson D, Robb RA, et al. Rezūm System water vapor treatment for lower urinary tract symptoms/benign prostatic hyperplasia: validation of convective thermal energy transfer and characterization with magnetic resonance imaging and 3-dimensional renderings. Urology. 2015 Jul;86(1):122-7.
- Lerner LB, McVary KT, Barry MJ, et al. Management of lower urinary tract symptoms attributed to benign prostatic hyperplasia: AUA Guideline 2021. J Urol. 2021 Oct;206:806-26.
- Mooney R, Goldberg K, Wong D, et al. Convective radio frequency thermal therapy for treatment of benign prostatic hyperplasia: Singe office experience with 255 patients over 4 years. Urol Pract. 2020 Jan;7(1):28-33.
- McVary KT, El-Arabi A, Roehrborn C. Preservation of Sexual Function 5 Years After Water Vapor Thermal Therapy for Benign Prostatic Hyperplasia. Sex Med. 2021 Dec;9(6):100454.
- Data on File with Boston Scientific.
Caution: U.S. Federal law restricts this device to sale by or on the order of a physician.
IMPORTANT INFORMATION: These materials are intended to describe common clinical considerations and procedural steps for the use of referenced technologies but may not be appropriate for every patient or case. Decisions surrounding patient care depend on the physician’s professional judgment in consideration of all available information for the individual case. Boston Scientific (BSC) does not promote or encourage the use of its devices outside their approved labeling. Case studies are not necessarily representative of clinical outcomes in all cases as individual results may vary.
All images are the property of Boston Scientific. All trademarks are the property of their respective owners.