Boston Scientific accounts are for healthcare professionals only.
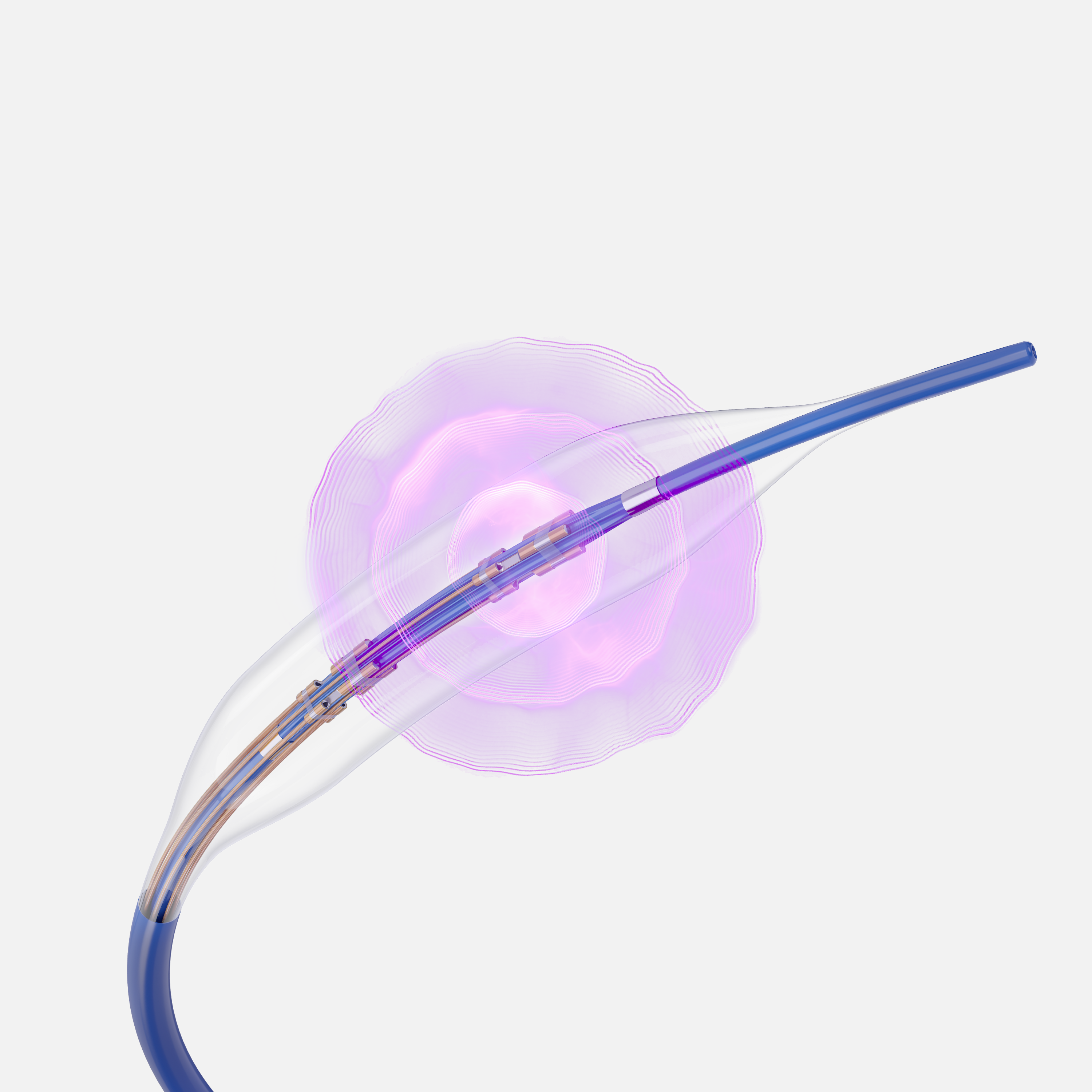
SEISMIQ™ 4CE Coronary Intravascular Lithotripsy Catheter
Configure or select a product to continue to order
- IVL overview
- Product details
- Clinical data
Intravascular lithotripsy (IVL)
Intravascular lithotripsy (IVL) is a catheter‑based therapy designed to treat patients with calcified coronary artery disease. IVL delivers acoustic pressure waves inside a balloon to fracture hardened calcium, restore vessel compliance, and enable effective lesion preparation prior to definitive therapy, such as treating with a stent.
Laser‑based IVL takes traditional IVL a step further. By using focused laser energy and a quad-emitter design to generate acoustic pressure waves within the balloon, SEISMIQ 4CE is designed to deliver precise, consistent calcium modification—helping physicians prepare complex lesions with greater control while minimizing the need for high-pressure dilation.
IVL in calcium morphologies:
Severe Concentric Calcium
Eccentric Calcium
Nodular Calcium
Looking for information on our SEISMIQ Peripheral IVL Catheter?

Discover the Modern PCI approach
Subscribe for updates on Interventional Cardiology topics
Receive timely updates on significant announcements, exclusive opportunities to engage with peers through educational events, and access valuable tools to enhance your ability to assist more patients.
Product details
SEISMIQ is a new coronary Intravascular Lithotripsy (IVL) System - an advanced laser-based platform that is designed to deliver consistent acoustic output to treat a broad range of calcified lesions.
A SEISMIQ shift in calcium modification

SEISMIQ 4CE is designed for:
2026
FRACTURE IDE Trial1
The FRACTURE IDE Trial is a prospective, multicenter, single-arm study using SEISMIQ 4CE to treat 420 patients with calcified coronary artery disease (CAD) with primary endpoints assessed at 30 days. Secondary endpoints including long-term safety and effectiveness will be measured through 24 months. This trial was conducted at 46 sites across the US, UK, and EU.
The FRACTURE IDE Trial studied a highly calcified subset of patients and lesions, including 100% site-reported severe angiographic calcium and OCT measurements of 27.4% nodular and arc of 271.9 ± 77.4 at the site of max calcium. The severely calcified de novo coronary lesions were ≥ 2.5 mm and ≤ 4.0 mm; with a lesion length ≤ 40 mm.
PRIMARY ENDPOINTS MET
Primary safety endpoint met:
93.3%
MACE-free at 30 days*
Primary effectiveness endpoint met:
93.7%
Procedural success**
Trial outcomes:
- Safety endpoint excluding peri-procedural MI: 99.3%
- Effectiveness endpoint excluding peri-procedural MI: 99.8%
- Final average MSA of 6.52 mm2
- Average stent expansion of 94.2%†
- Ventricular capture rate of 16.4%‡
2026
RESTORE First-in-Human Study2
RESTORE was a prospective, multicenter feasibility study of 41 PCI patients with a single de novo, severely calcified coronary lesion (RVD 2.5-4.0 mm; length ≤ 60 mm) treated with SEISMIQ 4CE and followed for 30 days.
PRIMARY ENDPOINTS
Primary safety endpoint:
90.2%
MACE-free at 30 days*
Primary effectiveness endpoint:
89.5%
Procedural success**
Study outcomes:
- Final average MSA of 8.0 mm2
- Average stent expansion of 100%†
- Ventricular capture rate of 14.6%‡
*MACE defined as CV death, MI, TVR.
**Procedural success defined as DES delivery with a final residual stenosis < 50% and freedom from in-hospital MACE.
† MSA at site of max calcium relative to proximal and distal references.
‡ Ventricular capture is per patient per case.
References
1. McEntegart M. et al. Safety and efficacy of novel intravascular lithotripsy system for coronary calcium: FRACTURE study. EuroPCR 2026 LBCT May 19.
2. Price M, Budrys P, Baranauskas A, et al. Laser- and optics-based intravascular lithotripsy for the treatment of calcified coronary stenoses: the RESTORE first-in-human study. Circ Cardiovasc Interv, 2026.
CAUTION: Investigational device. Limited by US law to investigational use only. Not available for sale.


