Boston Scientific accounts are for healthcare professionals only.
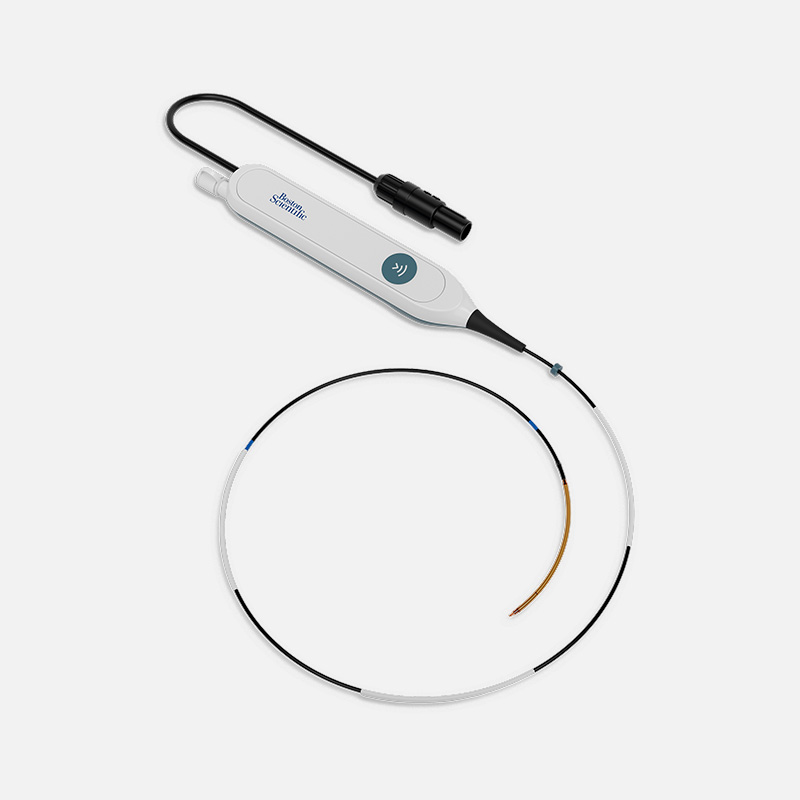
CEDAR™ Endovenous Radiofrequency System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical data
- Technical specifications
- Ordering information
- Training
Why choose CEDAR RFA System
Stay up to date
Sign up to get technology updates, news about major conferences, symposium invitations, and more.
1. Shang T, Zhu YF, Jiang JS, He J, Yang J, Lu T, et al. A multicenter randomized controlled trial evaluating a new radiofrequency ablation system in the treatment of primary great saphenous vein incompetence: Six-month results of the ACOART RF CLOSURE study. Phlebology. 2026. doi:10.1177/02683555261426651
Clinical data
CEDAR RFA clinical trial outcomes
A multicenter, randomized controlled trial evaluating a new radiofrequency ablation system in the treatment of primary great saphenous vein incompetence: Six-month results of the ACOART RF CLOSURE study1
Study Overview
- Multicenter, randomized controlled trial evaluating the CEDAR RFA system versus ClosureFast™
- 188 patients with primary great saphenous vein (GSV) incompetence enrolled across 5 vascular centers
- Primary endpoint: total GSV occlusion at 6 months
- Secondary endpoints: device success, technical success, patient‑reported outcomes (AVVQ, VCSS), and safety
Key Results
Effectiveness
- Complete GSV occlusion at 6 months:
- CEDAR: 97.78%
- ClosureFast: 98.86%
- Device success: 100% for both systems
- Immediate technical success: 100% for both systems
- Patient outcomes: Significant and comparable improvement in AVVQ and VCSS scores in both groups
- Large‑diameter vein performance:
- Patients with GSV ≥12 mm: 100% occlusion at 6 months for both devices
Safety
- Overall adverse event rates comparable between systems
- No device‑related adverse events or serious adverse events (SAEs) recorded
1. Shang T, Zhu YF, Jiang JS, He J, Yang J, Lu T, et al. A multicenter randomized controlled trial evaluating a new radiofrequency ablation system in the treatment of primary great saphenous vein incompetence: Six-month results of the ACOART RF CLOSURE study. Phlebology. 2026. doi:10.1177/02683555261426651
Technical information
CEDAR Endovenous RFA Catheter specifications
Model | ERA-C70-US | ERA-C30-US |
Introducer sheath (min. ID size) | 7F (2.3 mm) | 7F (2.3 mm) |
Insertable length | 60 cm | 60 cm |
Heating element diameter | 2.15 mm | 2.15 mm |
Heating element length | 7 cm | 3 cm |
Max power setting | 40W | 18W |
Default target temperature setting | 120°C | 120°C |
Compatible guidewire | 0.025 in | 0.025 in |
CEDAR Endovenous RFA Generator specifications
| Model | ERA-G5-US |
| Input | 110–230 VAC, 50/60 Hz, 120 VA |
| Height | 11.0 in |
| Width | 14.2 in |
| Depth | 7.6 in |
| Weight | 12.1 lbs |
Ordering information
| Product Name | UPN |
| CEDAR™ Endovenous Radiofrequency Ablation Catheter (3 cm) | ERA-C30-US |
| CEDAR Endovenous Radiofrequency Ablation Catheter (7 cm) | ERA-C70-US |
| Endovenous Radiofrequency Generator | ERA-G5-US |
Online medical training and education courses for CEDAR Endovenous Radiofrequency System
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
*CEDAR™ Endovenous Radiofrequency Ablation System includes the CEDAR Endovenous Radiofrequency Catheter and Endovenous Radiofrequency Generator
CEDAR RFA System manufactured by Acotec Scientific Co., Ltd. and distributed exclusively in the U.S. by Boston Scientific.
