Boston Scientific accounts are for healthcare professionals only.
.png)
EMBLEM™ MRI Subcutaneous Implantable Defibrillator (S-ICD) System
Reimbursement
Configure or select a product to continue to order
- Overview
- Implant procedure
- Clinical data
- mCRM™ System
- Technical specifications
- Ordering information
- Training
- Resources
Overview
Results are in from the PRAETORIAN DFT Trial | Primary endpoint met.
How it works
The EMBLEM MRI S-ICD System uses a pulse generator capable of delivering life-saving defibrillation therapy.
Unlike transvenous ICDs (TV-ICDs), the S-ICD System leaves the heart and vasculature untouched, avoiding the risks and complications associated with transvenous leads.1-3
Why choose the EMBLEM S-ICD System?
The EMBLEM™ S-ICD is proven to lower the risk of serious lead-related complications compared to transvenous ICDs.1
Reduced risk, proven performance
The EMBLEM S-ICD System is the only subcutaneous defibrillator with over two decades of proven performance.1,2,4-12 It remains outside the heart and sternum offering a less-invasive alternative that is clinically proven to reduce risks.2,13,14
The system has provided protection to 175,000 patients worldwide and counting.15
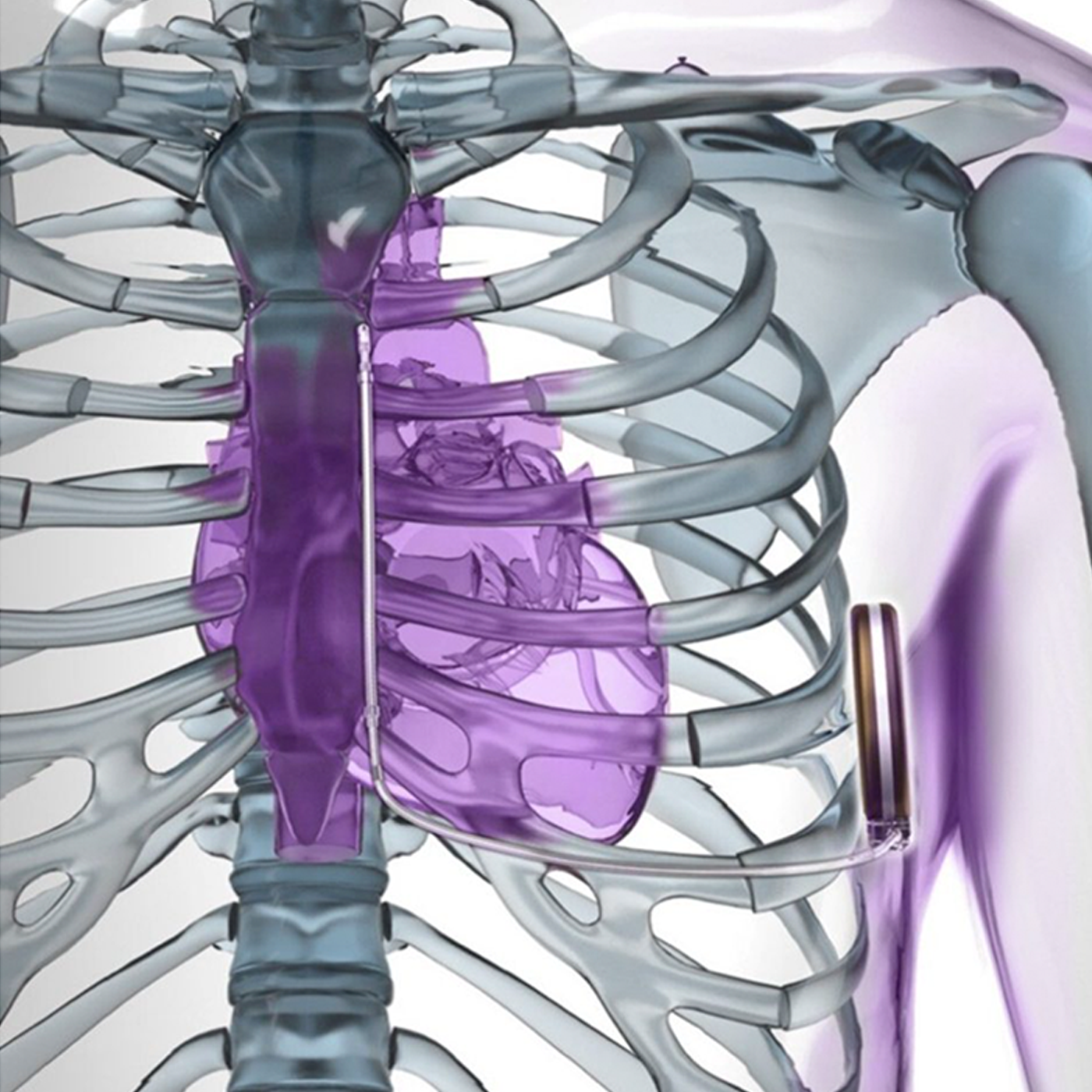
Personalized patient care
Upon FDA approval, the Modular CRM (mCRM™) System* and EMPOWER™ Leadless Pacemaker (LP)* is intended to provide an upgrade pathway, for patients who develop a need for ATP or bradycardia pacing.16,17
*Caution: Investigational Device. Limited by U.S. law to investigational use only. Not available for sale.


Patient selection
The EMBLEM S-ICD is recommended by clinical guidelines and supported by appropriate use criteria for patients at high risk of infection, inadequate venous access and any patient without a pacing indication — the majority of ICD-indicated patients.4,5,12 Technology advancements to the S-ICD over the past 10 years support the device as an option for most Primary Prevention patients.2,8
Find more information on why S-ICD should be considered for all patients without a pacing indication in the Benefits of EMBLEM S-ICD for Primary Prevention Patients (pdf).
Key features and specifications
SMART Pass
The SMART Pass filter is designed to reduce cardiac over-sensing and data has demonstrated that the inappropriate shock rate for S-ICD is now lower than TV-ICDs.8,18
AF Monitor
Designed to assist in the detection of silent, new onset or the progression of atrial fibrillation (AFib).19 The AF Monitor will notify a clinician when at least six minutes of AFib have been detected within a day.
Device longevity
Real-world data clinically shows EMBLEM S-ICD longevity of 8.7 years.20
LATITUDE™ NXT remote patient management system
Remote patient management has been shown to decrease mortality, hospitalizations and in-clinic evaluations.21 The EMBLEM MRI S-ICD System leverages LATITUDE NXT remote monitoring technology to help improve patient outcomes and enhance clinic efficiencies.
Proven performance you can count on
Explore clinical study and real-world data on inappropriate shock rates, complication rates and more.
Implant procedure
S-ICD implant techniques
Designed to be implanted using anatomical landmarks, the EMBLEM MRI S-ICD System reduces the need for fluoroscopy during implant and ensures a predictable and consistent implant procedure.19
Advancements in the implant technique have shown optimal placement and positioning can improve patient outcomes.22-26
Two-incision technique
By eliminating the superior parasternal incision used in the three-incision technique, the two-incision implant technique reduces procedure time and improves cosmetic outcomes.24,27
Intermuscular technique
With the intermuscular technique, a pocket is created between the serratus anterior and latissimus dorsi muscles to place the pulse generator behind the latissimus dorsi.23
Supporting videos
Pain control for S-ICD implants
Dr. Miller discusses the options for anesthesia and peri-operative pain control during S-ICD implant.
S-ICD implantation 2025 versus 2012
Dr. Aasbo offers a historical perspective on implant best practices today compared to 2012.
Training and education
Explore continuing education courses, best practices modules and more resources for EMBLEM S-ICD.
Clinical data
Decades of data: Clinical study summaries
PRAETORIAN DFT: Primary endpoint met
The primary objective of the PRAETORIAN DFT trial is to determine whether omitting ventricular fibrillation (VF) conversion testing during S-ICD implantation is non-inferior to performing VF conversion testing, when post operative position of the system is confirmed by assessment of the PRAETORIAN score.28
The trial met the primary endpoint and demonstrated non-inferiority (p < 0.001) of omitting VF conversion testing in EMBLEM S-ICD implants when guided by the PRAETORIAN score.29,30,**
Both arms experienced very low first failed shock conversion of spontaneous arrhythmias.29,30
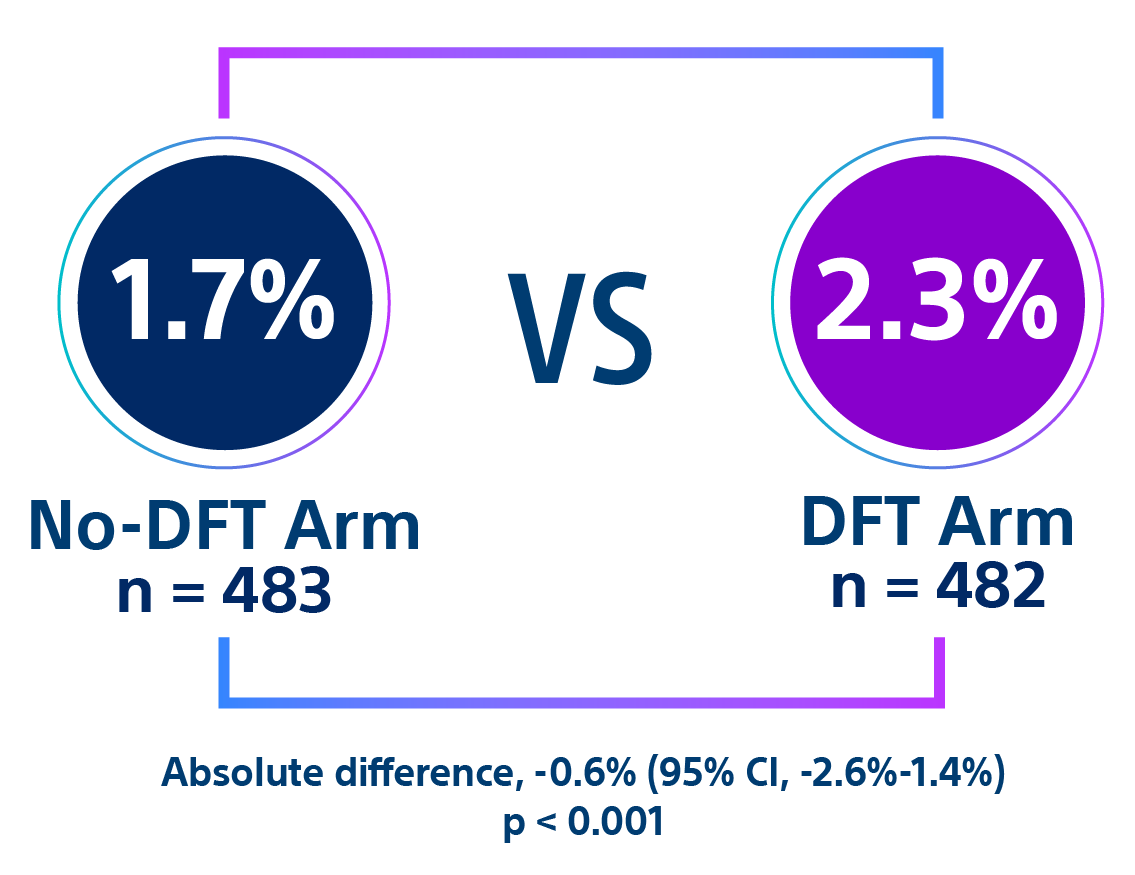
Complication rates were lower in the no-DFT arm compared to the DFT arm.
- After DFT testing, the potentially DFT-related complication rate in the DFT arm was 1.7% compared to zero in the no-DFT arm (p < 0.01).29,30
- Within 30 days of S-ICD implant, the composite complication rate was significantly higher in the DFT arm compared to the no-DFT arm (p < 0.01).29,30
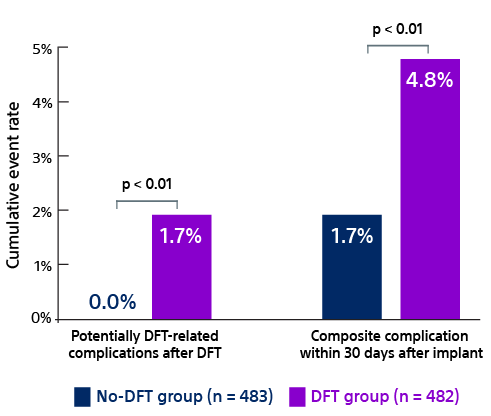
In the no-DFT arm, first and final shock efficacy was 92.1% and 97.9%, respectively.30 In the DFT arm, first and final shock efficacy was 91.5% and 97.5%, respectively.30 First and final shock efficacies in the PRAETORIAN DFT trial are similar to previous large S-ICD trials.7-9,11
| While numerically lower in the no-DFT arm, there was no statistically significant difference in all-cause mortality and arrhythmic death.29,30 | ||||
| No-DFT (n = 483) | DFT (n = 482) | HR (95% CI) | ||
| All-cause mortality | 35 (7.2%) | 39 (8.1%) | 0.9 (0.6-1.4) | |
| Arrhythmic death | 1 (0.2%) | 3 (0.6%) | 0.4 (0.04-3.4) | |
Intraoperative repositions were higher in the no-DFT arm compared to the DFT arm and the differences were not statistically significant.29,30 | ||||
| No-DFT (n = 483) | DFT (n = 482) | HR (95% CI) | ||
| Intraoperative repositioning | 23 (4.8%) | 15 (3.1%) | 1.55 (0.80-3.08) | |
Trial investigators found that omitting VF conversion testing, when the S-ICD system position is confirmed using the PRAETORIAN score, simplified the procedure, reduced complications and found no increase in first failed shocks or S-ICD revisions post implant.29,30
Note: In this trial, DFT testing is synonymous with VF conversion testing during implant.
The PRAETORIAN DFT trial is sponsored by Academic Medical Center (AMC) in Amsterdam. Funding for the trial is provided by Boston Scientific.
The manufacturer recommended VF conversion testing during EMBLEM S-ICD implant and replacement procedures is being evaluated based on the PRAETORIAN DFT trial results and additional available data.
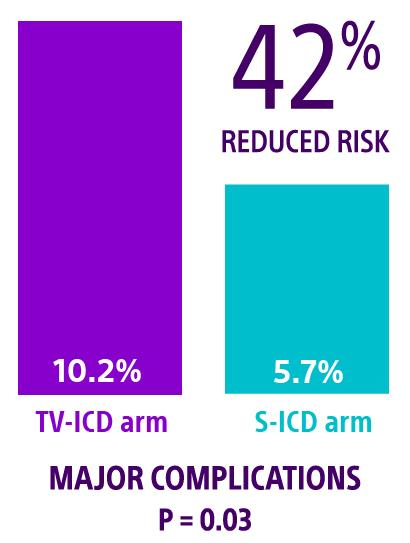
PRAETORIAN XL: Longest randomized trial of ICDs
The PRAETORIAN XL trial reconsented patients for a total follow-up of eight years to evaluate the superiority of the S-ICD to TV-ICD. Patients randomized to the S-ICD arm experienced a 42% lower risk of major complications compared to patients in the TV-ICD arm (HR: 0.58, 95% CI 0.36-0.95, P = 0.03).2
PRAETORIAN: A smart alternative to TV-ICD
PRAETORIAN is the first prospective, randomized, head-to-head, non-inferiority clinical trial that compares S-ICD to TV-ICD. The trial followed 850 patients at 40 centers across Europe and the United States. Full results were published in 2020.9
This content requires a subscription to access the full publication.
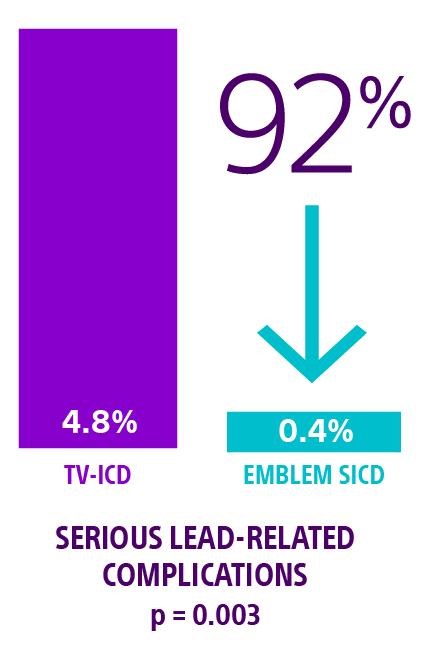
ATLAS trial: S-ICD superior to TV-ICD
The ATLAS trial met its primary superiority endpoint demonstrating a highly significant 92% reduction in serious lead-related complications† at six months for the EMBLEM S-ICD compared to any manufacturers' single chamber TV-ICD devices. p = 0.0031
This content requires a subscription to access the full publication.
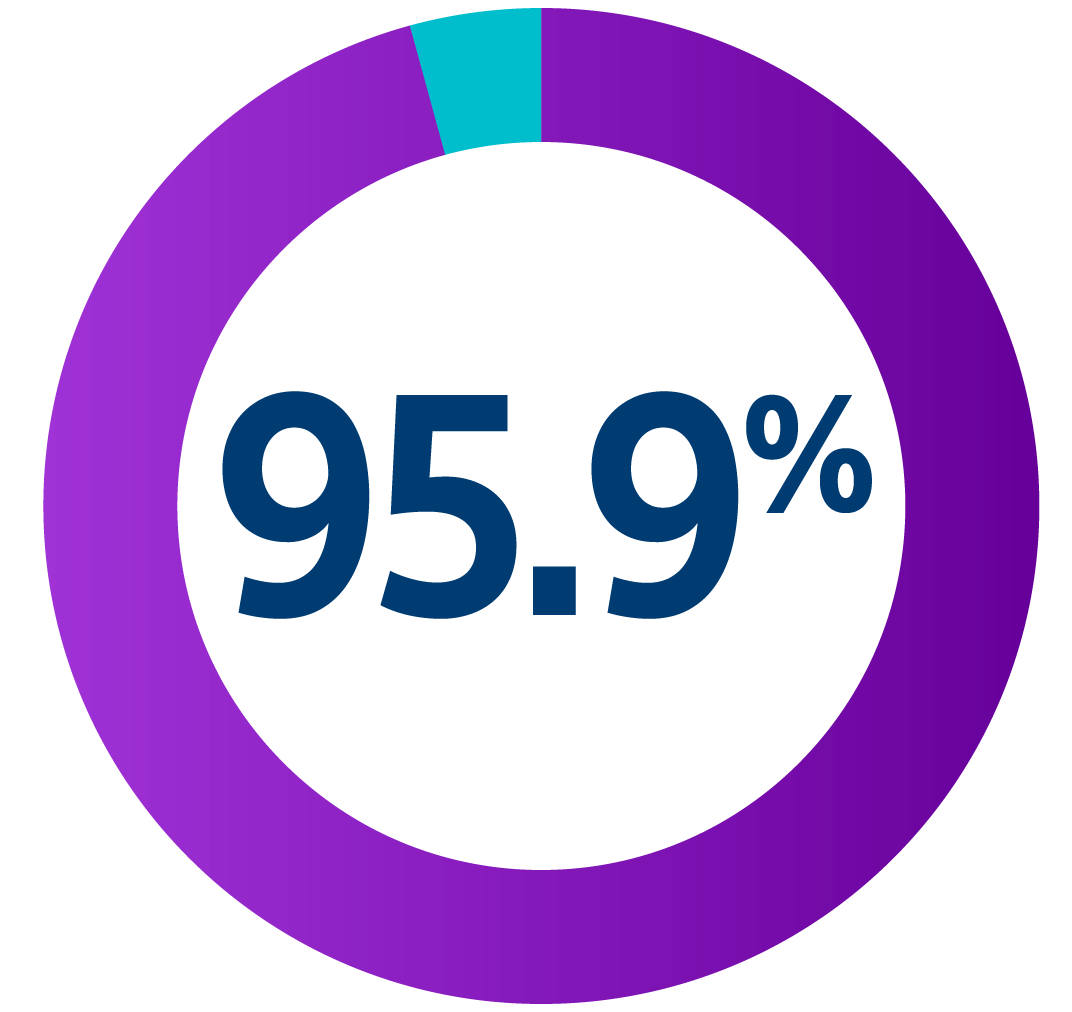
UNTOUCHED study: Inappropriate shocks lower than TV-ICD
The UNTOUCHED study evaluated inappropriate shock (IAS) rates from third-generation S-ICD devices among a conventional patient population. Using enhanced discrimination and standard programming, investigators achieved a lower IAS rate than recorded for most TV-ICDs. The IAS free rate achieved at 18 months was 95.9%.8
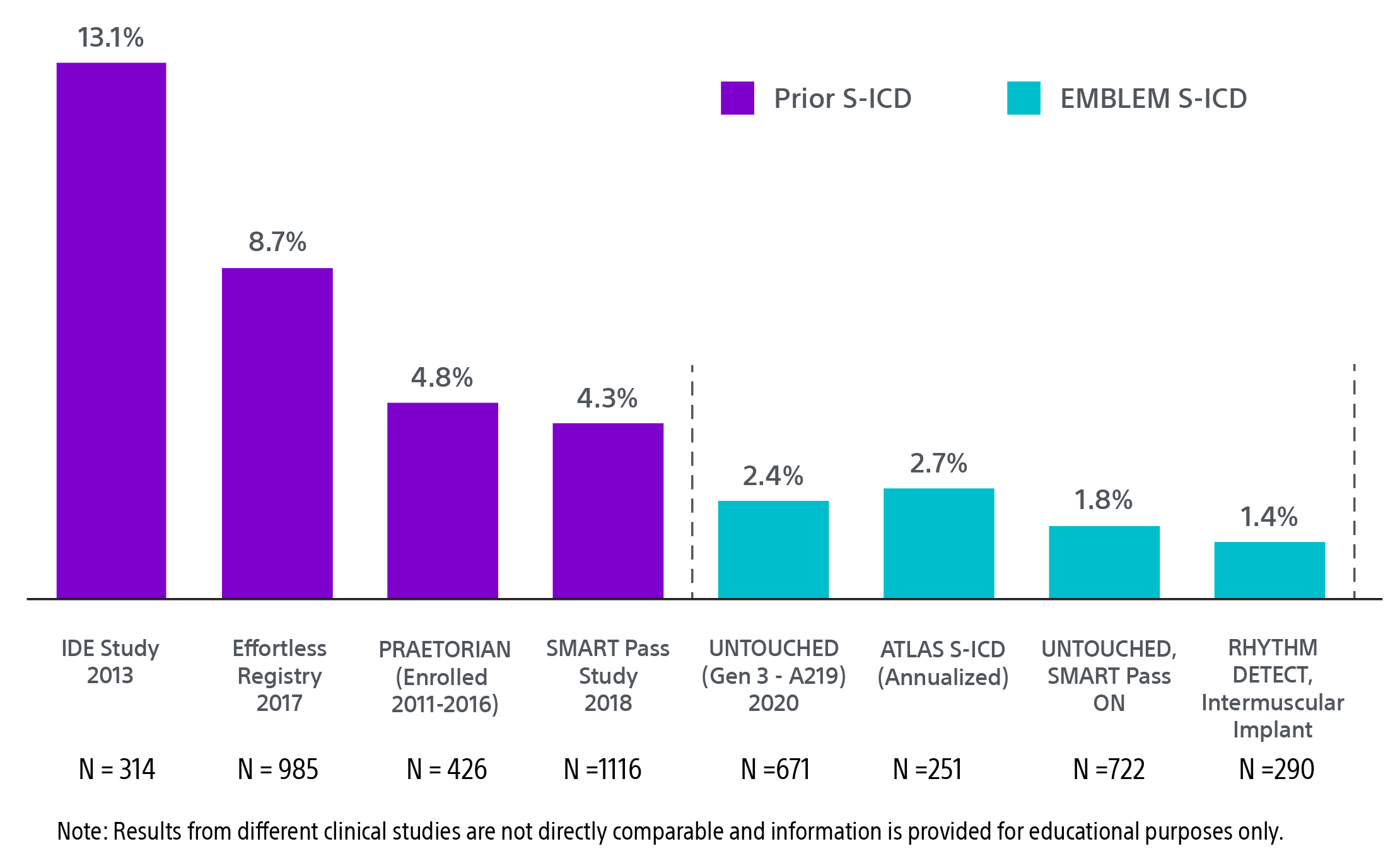
Continued reduction in IAS rates‡
Updates to technology and new implant techniques are credited with a continued reduction of inappropriate shock rates. Recent S-ICD one-year inappropriate shock rates from the UNTOUCHED and RHYTHM DETECT studies were less than 2.0%.18,22 This is five times lower than substernal ICD and comparable to TV-ICD.1,31
Updates to technology and new implant techniques are credited with a continued reduction of inappropriate shock rates. Recent S-ICD one-year inappropriate shock rates from the UNTOUCHED and RHYTHM DETECT studies were less than 2.0%.18,22 This is five times lower than substernal ICD and comparable to TV-ICD.1,31
APPRAISE ATP: Importance of shared decision making
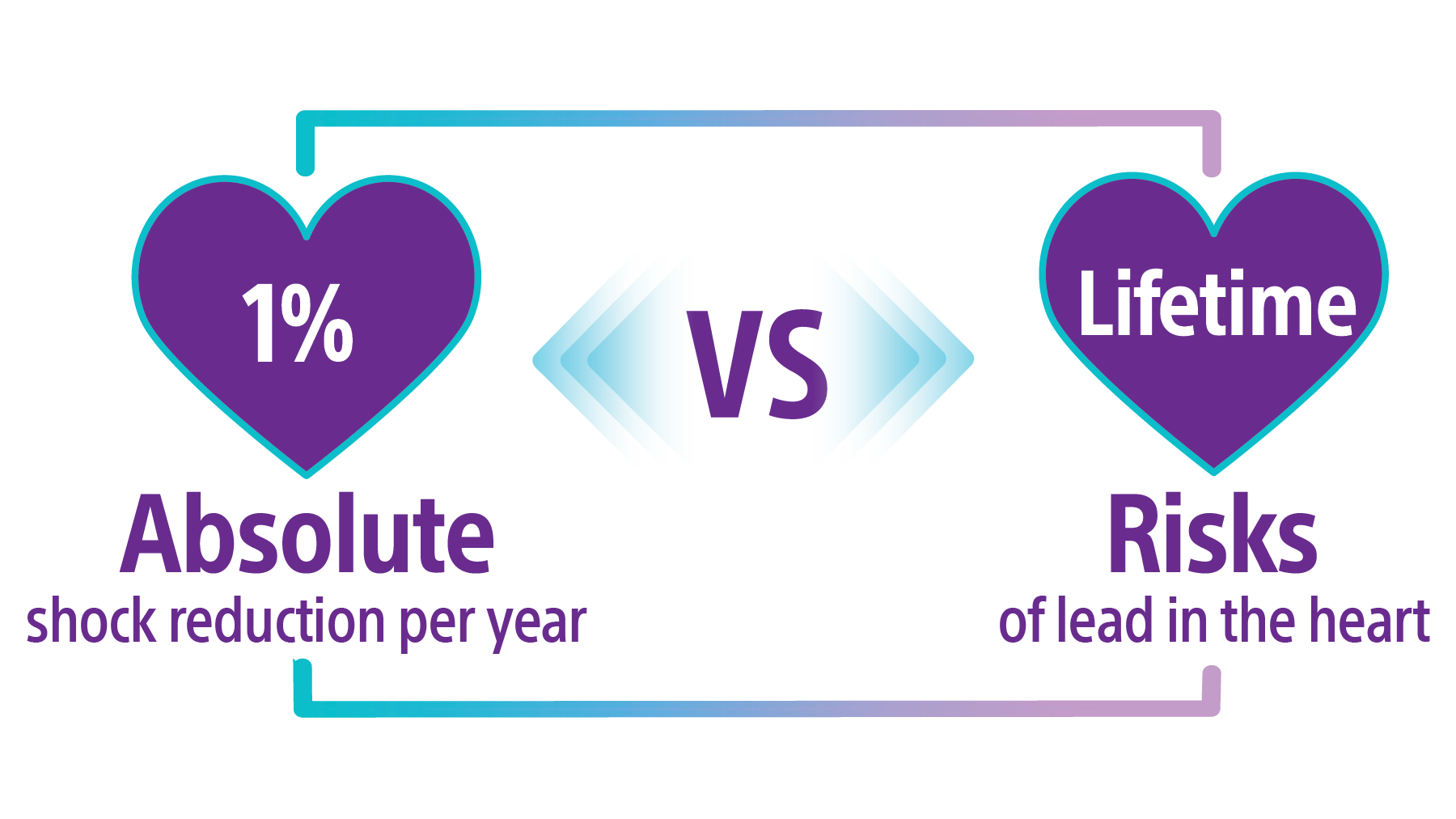
The APPRAISE ATP trial demonstrated superiority with a 28% relative risk reduction in time to first all-cause shock for the ATP ON arm compared to the ATP OFF arm (Log-rank P-value = 0.005). This represents an absolute all-cause shock reduction in 1% of PP ICD indicated patients per year.32
The APPRAISE ATP trial demonstrated superiority with a 28% relative risk reduction in time to first all-cause shock for the ATP ON arm compared to the ATP OFF arm (Log-rank P-value = 0.005). This represents an absolute all-cause shock reduction in 1% of PP ICD indicated patients per year.32
This content requires a subscription to access the full publication.
MODULAR ATP Trial
Explore results from the first modular pacemaker-defibrillator system with wireless communication.
mCRM System
Modular Cardiac Rhythm Management (mCRM) System*
Personalized patient care that pairs leadless ATP and brady pacing with subcutaneous defibrillation.
*Caution: Investigational Device. Limited by U.S. law to investigational use only. Not available for sale.
What is mCRM therapy?
The Modular Cardiac Rhythm Management (mCRM) System* is an evolution in cardiac rhythm management therapy. Upon FDA approval, its components — the EMBLEM S-ICD and the new EMPOWER™ Leadless Pacemaker (LP)* — are designed to operate both independently and in coordination to address evolving clinical needs.16,17
Either device can be implanted first, offering a modular solution that adapts with the patient by providing an upgrade pathway.17 The mCRM System provides patients what they need, when they need it.
How it works
When the EMBLEM S-ICD senses a tachycardia episode, it requests the EMPOWER Leadless Pacemaker* to provide ATP therapy with the intent to stop tachycardia and reset the heart to a normal rhythm.
The EMBLEM S-ICD then monitors the result of the ATP therapy. If the ATP successfully terminates the tachycardia and returns the heart to a normal rhythm, the EMBLEM S-ICD will continue monitoring and no shock will be delivered. If the ATP does not terminate the tachycardia, the EMBLEM S-ICD will deliver shock therapy.
Built on the proven performance of the EMBLEM S-ICD,1,2,4-12 the mCRM System* is designed to offer a modular, leadless approach to cardiac rhythm management.17 This modular configuration is intended to provide flexibility to expand treatment as patient’s needs evolve.
Watch how the S-ICD requests ATP from the EMPOWER LP* in the mCRM System*
*Caution: Investigational Device. Limited by U.S. law to investigational use only. Not available for sale.
mCRM system components

EMBLEM S-ICD
The only extrathoracic implantable defibrillator that provides protection from both sudden cardiac death and the risks and complications associated with transvenous leads.
Remains outside the rib cage, never touching the heart
Reduces risk of systemic infection
Preserves the vasculature

EMPOWER Leadless Pacemaker*
Once FDA approved, the EMPOWER Leadless Pacemaker will pair with the S-ICD to provide pacing or ATP therapy.
EMPOWER is also designed to be a standalone pacemaker
ATP when commanded by a paired S-ICD
Delivery system with inner extendable shaft
20.7F outer diameter delivery catheter
Dedicated retrieval catheter
Rate response via accelerometer

mSYNC™ Communication
Once approved by the FDA, mSYNC will serve as Boston Scientific’s proprietary device-to-device communication technology. In the mCRM System, mSYNC Communication is used by the EMBLEM S-ICD to wirelessly transmit ATP request signals to the EMPOWER Leadless Pacemaker.
*Caution: Investigational Devices. Limited by U.S. law to investigational use only. Not available for sale.
MODULAR ATP trial: Clinical data results
Major Complication-free Rate
97.5%
The EMPOWER Leadless Pacemaker System demonstrated a major complication-free rate of 97.5%.
Communication Testing
98.8%
Communication success rate was 98.8% between the EMBLEM S-ICD and EMPOWER Leadless Pacemaker.
Pacing Capture Threshold (PCT)
97.4%
97.4% of patients experienced PCTs ≤ 2.0 volts (0.4 ms pulse width). (PCTs across patients were 0.56 ± 0.49 V).
Six-month data
The MODULAR ATP trial serves as a key input to FDA submission of the EMPOWER Leadless Pacemaker and the industry's first mCRM system.
This pivotal trial evaluated the safety, effectiveness, and performance of the mCRM System which successfully exceeded all pre-specified safety and effectiveness endpoints at six-month analysis.16
The communication success rate§ of 98% refers to the mCRM wireless communication. This is conducted, one-way communication from the S-ICD to the EMPOWER Leadless Pacemaker to request ATP delivery.
This content requires a subscription to access the full publication.
Twelve-month data33
Twelve-month data33
97.2%
At 12 months, the results showed the EMPOWER Leadless Pacemaker achieved a 97.2% complication-free rate (n = 286).
Additional metrics:
ATP success rate of 67.3% (adjusted rate) resulting in painless ATP and shock-free arrythmia termination.
There was no significant impact to the battery life of the EMPOWER Leadless Pacemaker* or S-ICD with the use of ATP communication or ATP delivery.
Final shock efficacy of 98.2%.
Stable pacing capture thresholds from pre-discharge out to two years, 0.54 ± 0.58 V and 0.57 ± 0.46 V, respectively (mean ± standard deviation).
Technical specifications
EMBLEM MRI S-ICD Pulse Generator
Mechanical specifications
| Model number | A219 |
| Size (W x H x D) | 83.1 x 69.1 x 12.7 mm |
| Mass | 130 g |
| Volume | 59.5 cc (cm³) |
| Projected longevity | 8.7 years20 |
| Battery chemistry | Boston Scientific Li/MnO2 |
| Warranty | 6 years‖ |
| Remote patient monitoring capability | Enabled for LATITUDE™ NXT Remote Patient Management |
Automatic functions
| Sensing configuration | Primary (ring to can), Secondary (tip to can), Alternate (tip to ring) Optimal sensing configuration automatically selected during Auto Setup (manual programming optional) |
| Gain selections | x1, x2 Optimal gain selection automatically selected during Auto Setup (manual programming optional) |
| Rhythm discrimination | INSIGHT™ algorithm automatically activated when the Conditional Shock Zone is programmed |
| Shock polarity | Standard (coil to can), reverse (can to coil) Automatically selects and stores last successful shock polarity |
| Adaptive shock polarity | Shock polarity alters automatically after failed shock |
| SMART charge | Automatically extends initial detection time to allow self-termination of non-sustained tachyarrhythmias |
| SMART pass | High-pass filter designed to reduce oversensing while maintaining an appropriate sensing margin |
| Internal warning system | Audible tone alerts patients to elective replacement indicators, electrode impedance out of range, prolonged charge times, failed device integrity check. NOTE: Exposure to MRI scanning can cause a permanent loss of the Beeper volume. The physician and patient should weigh the benefit of the MR procedure against the risk of losing the Beeper. |
Programmable parameters
| Shock zone | 170 bpm - 250 bpm (steps of 10 bpm) |
| Conditional shock zone | Off, On 170 bpm - 240 bpm (minimum 10 bpm less than shock zone) |
| S-ICD System therapy | Off, On |
| Post-shock pacing | Off, On (50 ppm, max 30 sec, demand-based) |
| Induction capability | 1 - 10 sec (50 Hz/200 mA) |
| Delivered energy | 80J biphasic (only programmable during manual shock and induction test: 10J - 80J, steps of 5J) |
| AF monitor | Off, On (model A219 only) |
| MRI protection | Time-out (hours): 6, 9, 12, 24 |
Diagnostics
| Episode storage | S-ECG storage for over 40 arrhythmic events (treated and untreated) |
| AF episodes | 7 stored (model A219 only) |
| Other data | Electrode impedance System status (remaining battery life, patient alerts, etc.) Date and time stamp |
EMBLEM S-ICD Electrode Model 3501
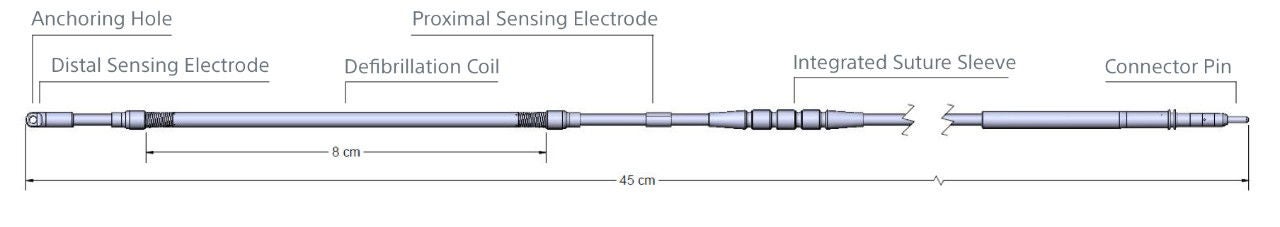
Electrode specifications
| Model number | 3501 |
| Type | Tripolar |
| Length | 45 cm |
| Distal type size (diameter) | 11.5 Fr/3.84 mm |
| Coil size (diameter) | 9 Fr/3 mm |
| Electrode shaft size (diameter) | 7 Fr/2.33 mm |
| Sensing surface area | Distal: 36 mm² Proximal: 46 mm² |
| Sensing location | Distal: At tip Proximal: 120 mm from tip |
| Defibrillation surface area | 750 mm² |
| Defibrillation location | 20 - 100 mm from tip |
| Materials | Insulation: Polycarbonate, Polyurethane Electrodes: MP35N Conductors: MP35N Connector Pin: MP35N Integrated Suture Sleeve: Radiopaque White Silicone Slit Suture Sleeve: Silicone |
Ordering information
| Model number | Description |
| A219 | EMBLEM MRI S-ICD Pulse Generator |
| 3501 | EMBLEM S-ICD Electrode |
| 4712 | EMBLEM S-ICD Electrode Insertion Tool |
| 3300 | LATITUDE™ Programming System |
Training
Education for the EMBLEM S-ICD System
Expand your S-ICD knowledge with these guides, webinars and interactive tools.

A series of eight S-ICD implant best practices videos.

Procedural videos and an interactive PDF present implant workflow and best practices for S-ICD.
Online medical training and education courses
The EDUCARE online platform makes healthcare education and training more relevant, more comprehensive, more personal, and more accessible. Register to access a library of procedural videos, case studies, training resources, and events.
Resources
Product resources
Spec sheet
Patient website
Videos
S-ICD vs. TV-ICD: What’s ideal for your patients?
Dr. Jeanne Poole reviews several case studies and the decision-making process for choosing the right ICD.
Why consider S-ICD for primary patients 50+
Dr. Mehta discusses why the S-ICD should be considered in all patients, including primary prevention patients over 50.
Patient age and S-ICD
Saumya Sharma, MD shares his perspective on how patient age may impact S-ICD selection.
* Caution: Investigational Device. Limited by US law to investigational use only. Not available for sale.
** The manufacturer recommended VF conversion testing during EMBLEM S-ICD implant and replacement procedures is being evaluated based on the PRAETORIAN DFT trial results and additional available data.
† In the ATLAS trial, serious complications were defined as moderate-severe or severe tricuspid regurgitation, hemothorax/pneumothorax, cardiac perforation, tamponade, pericardial effusion or pericarditis, ipsilateral upper extremity deep vein thrombosis and lead dislodgement or loss of sensing or pacing requiring revision.
‡ IAS graph references for S-ICD (see at bottom of Clinical data tab)
IDE study: Weiss R, Knight BP, Gold MR, et al. Safety and efficacy of a totally subcutaneous implantable-cardioverter defibrillator. Circulation. 2013;128(9):944-953. doi:10.1161/circulationaha.113.003042
Effortless Registry: Lambiase PD, Theuns DA, Murgatroyd F, et al. Subcutaneous implantable cardioverter-defibrillators: long-term results of the EFFORTLESS study. Eur Heart J. 2022;43(21):2037-2050. doi:10.1093/eurheartj/ehab921
PRAETORIAN: Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526-536. doi:10.1056/NEJMoa1915932
SMART Pass: Theuns D, Brouwer TF, Jones PW, et al. Prospective blinded evaluation of a novel sensing methodology designed to reduce inappropriate shocks by the subcutaneous implantable cardioverter-defibrillator. Heart Rhythm. 2018;15(10):1515-1522. doi:10.1016/j.hrthm.2018.05.011
UNTOUCHED Gen 3: Gold MR, Lambiase PD, El-Chami MF, et al. Primary results from the Understanding Outcomes With the S-ICD in Primary Prevention Patients With Low Ejection Fraction (UNTOUCHED) trial. Circulation. 2021;143(1):7-17. doi:10.1161/CIRCULATIONAHA.120.048728
ATLAS S-ICD: Healey JS, Krahn AD, Bashir J, et al. Perioperative safety and early patient and device outcomes among subcutaneous versus transvenous implantable cardioverter defibrillator implantations: a randomized, multicenter trial. Ann Intern Med. 2022;175(12):1658-1665. doi:10.7326/M22-1566
UNTOUCHED SMART Pass: Boersma LVA, Aasbo J, Knops RE, et al. The impact of SMART pass algorithm status on inappropriate shock rates in the UNTOUCHED study. Europace. 2022;24(suppl 1):euac053.391. doi:10.1093/europace/euac053.391
RHYTHM DETECT: Botto GL, Ziacchi M, Nigro G, et al. Intermuscular technique for implantation of the subcutaneous implantable defibrillator: a propensity-matched case–control study. Europace. 2023;25(4):1423-1431. doi:10.1093/europace/euad028
§ Communication testing assessed a pass/fail of communication at the programmed S-ICD telemetry setting and was required at the 6-month visit in four body postures. The test had to pass in more than 88% of attempts, across all postures, to be defined as successful.
‖ For full warranty terms and conditions go to bostonscientific.com/en-US/pprc/warranty-info-forms.html
REFERENCES:
- Healey JS, Krahn AD, Bashir J, et al. Perioperative safety and early patient and device outcomes among subcutaneous versus transvenous implantable cardioverter-defibrillator implantations: a randomized, multicenter trial. Ann Intern Med. 2022;175(12):1658-1665. doi:10.7326/M221-566.
- Olde Nordkamp LRA, de Veld JA, Ghani A, et al. Device-related complications in transvenous versus subcutaneous defibrillator therapy during long-term follow-up: the PRAETORIAN-XL trial. Circulation. 2025;0(0). doi:10.1161/CIRCULATIONAHA.125.074576.
- Leong DP, Dokainish H, Mondesert B, et al. Effects of implantable cardioverter-defibrillator leads on the tricuspid valve and right ventricle: a randomized trial. JACC Clin Electrophysiol. 2024;10(9):2088-2096. doi:10.1016/j.jacep.2024.04.034.
- Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary. Circulation. 2018;138(13):e210-e271. doi:10.1161/CIR.0000000000000548.
- Russo AM, Desai MY, Do MM, et al. ACC/AHA/ASE/HFSA/HRS/SCAI/SCCT/SCMR 2025 appropriate use criteria for implantable cardioverter-defibrillators, cardiac resynchronization therapy, and pacing. J Am Coll Cardiol. 2025;85(11):1213-1285. doi:10.1016/j.jacc.2024.11.023.
- Bardy G, Smith W, Hood M, et al. An entirely subcutaneous implantable cardioverter-defibrillator. N Engl J Med. 2010;363(1):36-44.
- Gold MR, El-Chami MF, Burke MC, et al. Postapproval study of a subcutaneous implantable cardioverter-defibrillator system. J Am Coll Cardiol. 2023;82(5):383-397. doi:10.1016/j.jacc.2023.05.034.
- Gold MR, Lambiase PD, El-Chami MF, et al. Primary results from the UNTOUCHED trial. Circulation. 2021;143(1):7-17. doi:10.1161/CIRCULATIONAHA.120.048728.
- Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526-536. doi:10.1056/NEJMoa1915932.
- Knops RE, van der Stuijt W, Delnoy P, et al. Efficacy and safety of appropriate shocks and antitachycardia pacing in transvenous and subcutaneous ICDs: PRAETORIAN analysis. Circulation. 2022;145(5):321-329. doi:10.1161/CIRCULATIONAHA.121.057816.
- Lambiase PD, Theuns DA, Murgatroyd F, et al. Long-term results of the EFFORTLESS study. Eur Heart J. 2022;43(21):2037-2050. doi:10.1093/eurheartj/ehab921.
- Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2022;43(40):3997-4126. doi:10.1093/eurheartj/ehac262.
- Payne JE, Gold MR. A substernal defibrillator lead with pacing capability. JACC Clin Electrophysiol. 2019;5(2):197-198. doi:10.1016/j.jacep.2018.12.006.
- Romers H, van Dijk V, Boersma L. Evolution of extravascular ICD therapy. Heart Rhythm O2. 2023;4(1):59-64. doi:10.1016/j.hroo.2022.09.021.
- Boston Scientific. CRM Product Performance Report. Q3 2024.
- Knops RE, Lloyd MS, Roberts PR, et al. A modular communicative leadless pacing-defibrillator system. N Engl J Med. 2024;391(15):1402-1412. doi:10.1056/NEJMoa2401807.
- Lloyd MS, Brisben AJ, Reddy VY, et al. Design and rationale of the MODULAR ATP global clinical trial. Heart Rhythm O2. 2023;4(7):448-456. doi:10.1016/j.hroo.2023.05.004.
- Boersma LVA, Aasbo J, Knops RE, et al. SMART Pass algorithm status and inappropriate shocks in UNTOUCHED. Europace. 2022;24(Suppl 1):euac053.39. doi:10.1093/europace/euac053.391.
- Boston Scientific. EMBLEM S-ICD and EMBLEM MRI S-ICD User’s Manual. 2021.
- van der Stuijt W, Williams JL, Brisben AJ, et al. Battery longevity and its predictors in subcutaneous implantable cardioverter-defibrillators (S-ICDs): A mathematical analysis derived from a large dataset. Heart Rhythm. Mar 2025;22(3):868-870. doi:10.1016/j.hrthm.2024.09.009.
- Akar J, et al. Use of remote monitoring and outcomes in ICD patients. Heart Rhythm Society Scientific Sessions. 2014;Abstract LB03-03.
- Botto GL, Ziacchi M, Nigro G, et al. Intermuscular S-ICD implantation: propensity-matched study. Europace. 2023;25(4):1423-1431. doi:10.1093/europace/euad028.
- Winter J, Siekiera M, Shin DI, et al. Long-term performance of intermuscular S-ICD implantation. Europace. 2017;19(12):2036-2041. doi:10.1093/europace/euw297.
- Brouwer TF, Miller MA, Quast ABE, et al. Evaluation of four S-ICD implantation techniques. Circ Arrhythm Electrophysiol. 2017;10:e004663. doi:10.1161/CIRCEP.116.004663.
- Droghetti A, Locatelli A, Casiraghi B, et al. Totally submuscular S-ICD implantation in obese patients. Clin Case Rep. 2016;4(10):1009-1011. doi:10.1002/ccr3.652.
- Migliore F, Allocca G, Calzolari V, et al. Intermuscular two-incision S-ICD technique: registry results. Pacing Clin Electrophysiol. 2017;40(3):278-285. doi:10.1111/pace.12987.
- Knops RE, Olde Nordkamp LR, de Groot JR, et al. Two-incision S-ICD implantation technique. Heart Rhythm. 2013;10:1240-1243. doi:10.1016/j.hrthm.2013.05.016.
- Quast ABE, Baalman SWE, Betts TR, et al. Rationale and design of the PRAETORIAN-DFT trial: A prospective randomized CompArative trial of SubcutanEous ImplanTable CardiOverter-DefibrillatoR ImplANtation with and without DeFibrillation testing. Am Heart J. 2019;214:167-174. doi:10.1016/j.ahj.2019.05.002.
- Knops R. A randomized comparison of subcutaneous implantable cardioverter defibrillator implantation with or without defibrillation testing: the primary results of the PRAETORIAN-DFT Trial. HRS 2026. LB-525395.
- Knops RE, Marquie C, Nordbeck P, et al. Subcutaneous Defibrillator Implantation With or Without Defibrillation Test: The Primary Results of the Randomized PRAETORIAN-DFT Trial. Circulation. 2026;0(0)doi:10.1161/CIRCULATIONAHA.126.080638.
- Friedman P, Murgatroyd F, Boersma LVA, et al. Chronic safety and performance of the EV-ICD: pivotal study. Heart Rhythm. 2023;20(7):1091-1092. doi:10.1016/j.hrthm.2023.04.038.
- Schuger C, Joung B, Ando K, et al. APPRAISE ATP randomized clinical trial. JAMA. 2024;332(20):1723-1731. doi:10.1001/jama.2024.16531.
- Lloyd MS, Reddy VY, Roberts P, et al. One-year outcomes of the MODULAR ATP trial. Circ Arrhythm Electrophysiol. 2025. doi:10.1161/circep.125.014395.
