Medical Specialties > Structural Heart > EuroPCR 2021 Clinical Presentation
The Early neo2 Registry is an investigator-initiated and conducted, single arm, multicenter registry that retrospectively assessed the ACURATE neo2™ Valve outcomes in 554 patients across 12 centers.1
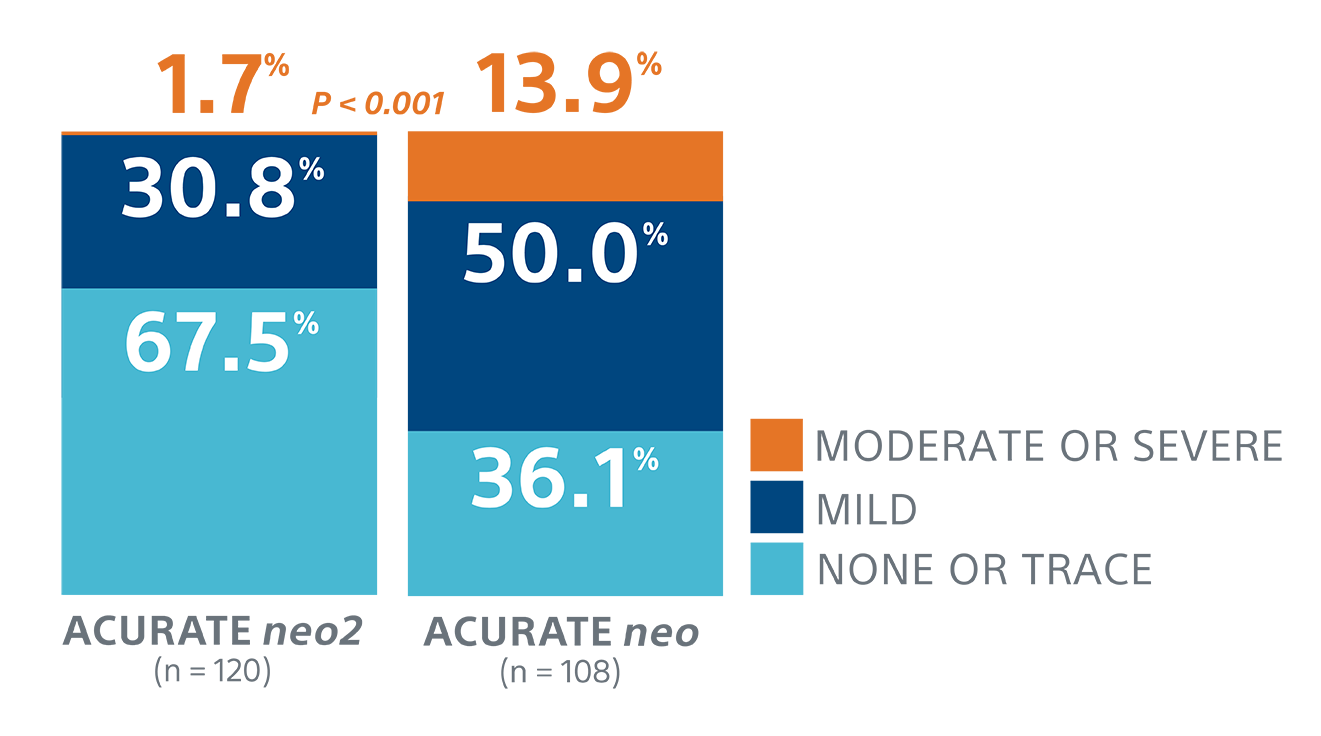
At EuroPCR 2021 data was presented on a separate videodensitometry angiographic core-lab adjudicated analysis comparing patients from the Early neo2 Registry implanted with the ACURATE neo2 Valve against patients implanted with the ACURATE neo™ Valve. In this study, the ACURATE neo2 Valve demonstrated a statistically significant lower rate of angiographically assessed moderate/severe aortic regurgitation (1.7% vs 13.9%, P<0.001).2
In the USA, the ACURATE neo2 valve is an Investigational Device and restricted under federal law to investigational use only, not available for sale. In the EU, the ACURATE neo2 Valve is CE marked.
A separate videodensitometry angiographic core-lab adjudicated analysis compared patients implanted with the ACURATE neo2 and ACURATE neo valves and demonstrated significant aortic regurgitation reduction.1
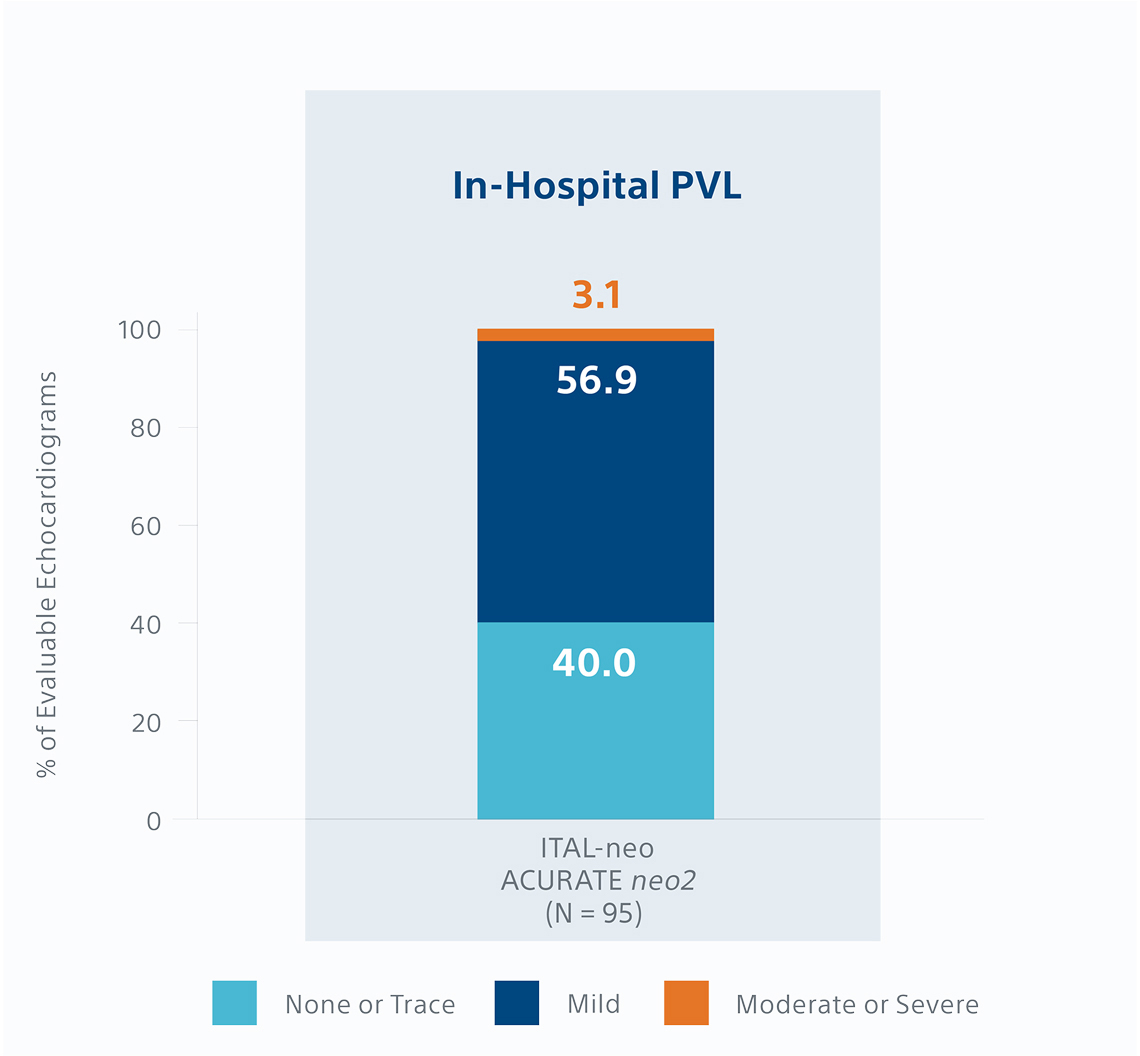
Percent of evaluable echocardiograms from a separate videodensitometry angiographic core-lab analysis of the Early neo2 Registry outcomes.1,*
2. Buono A. Short-term outcomes of a novel self-expanding device: ITAL-neo Registry. Euro PCR 20211. Rück A. Results from the Early neo2 Registry on ACURATE neo2 TAVI Valve. EuroPCR congress. 2021. conference. 2021.
Key Registry outcomes:
- Device success rate; 97.9%
- In-hospital PVL rate; 3.1% moderate/severe*
- In-hospital new permanent pacemaker implantation rate; 11.2%
- Pre-discharge mean gradient; 8.2mmHg*
- In-hospital Stroke rate; 1.1%
3. Buono A. Short-term outcomes of a novel self-expanding device: ITAL-neo Registry. Euro PCR 2021 conference. 2021.
