Boston Scientific accounts are for healthcare professionals only.






Advantage Fit™ System Transvaginal Mid-Urethral Sling System
Reimbursement
Configure or select a product to continue to order
- Overview
- Clinical data
- Technical specifications
- Training
- Resources
- Ordering information
Over 20 years on the market
Boston Scientific’s clinically supported Advantage mesh has been documented in more than 100 publications to date and has been used in more than 1 million slings.
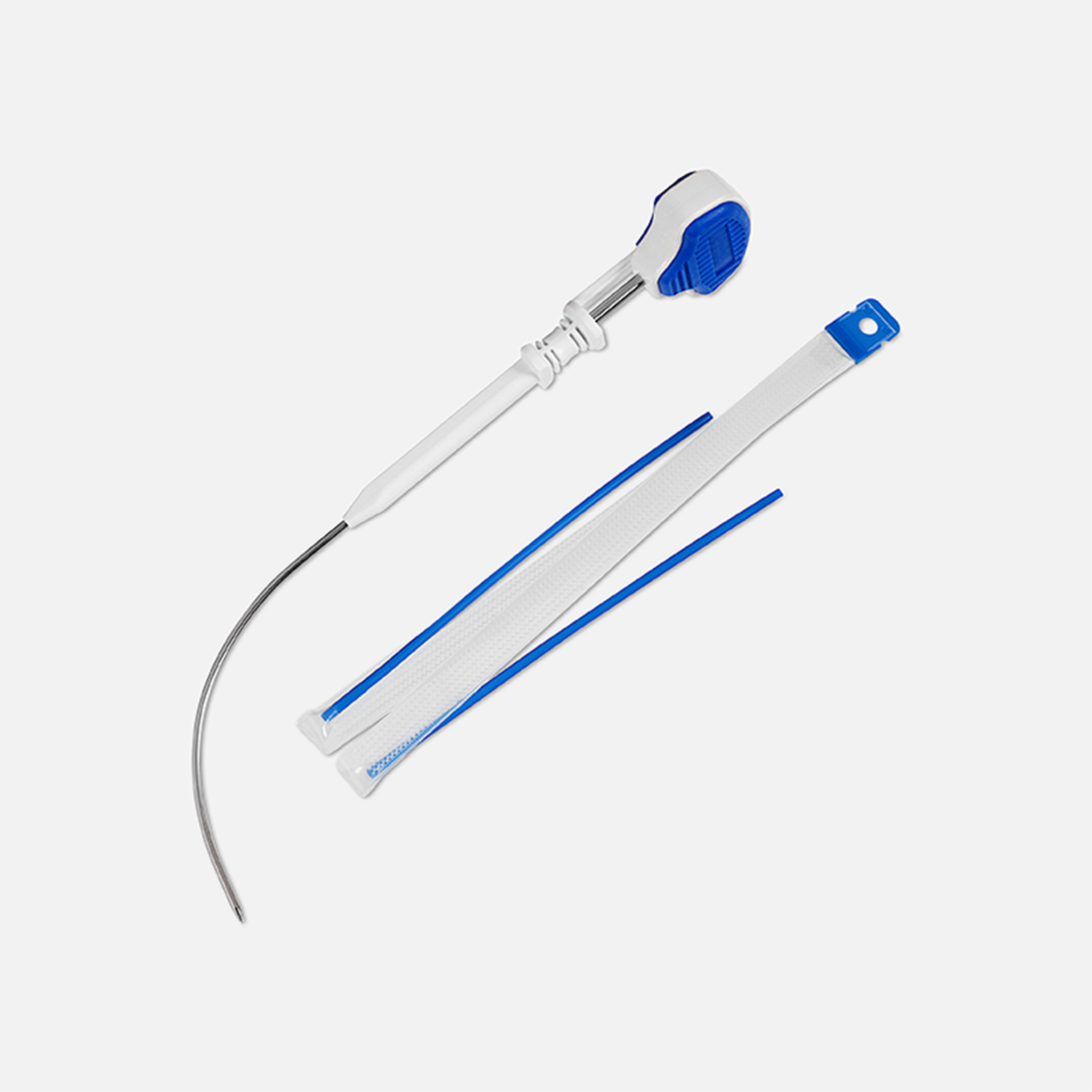
Why choose Advantage Fit
Advantage Fit Mesh assembly
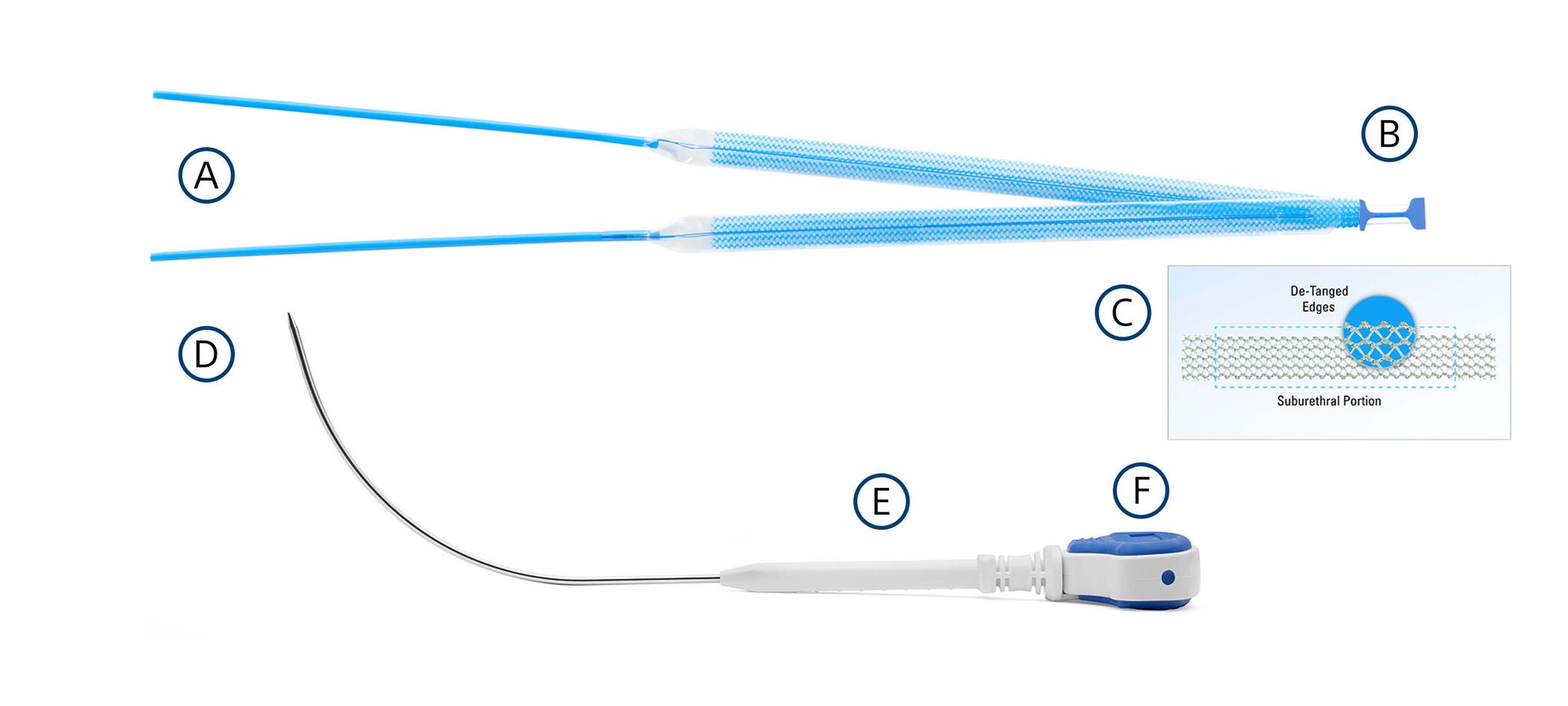
Ergonomic delivery
What’s included
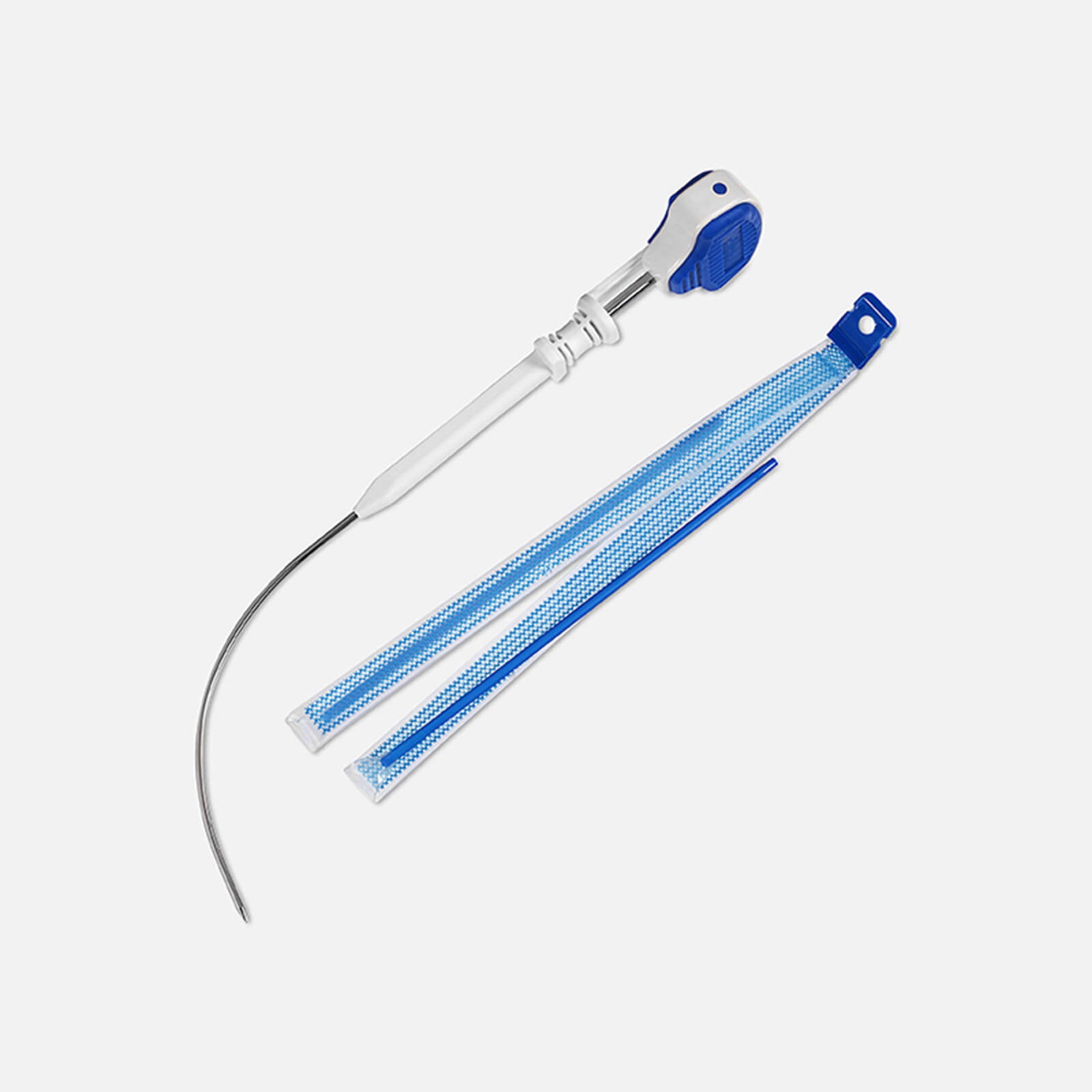
Questions?
We are here to help answer any questions you may have about Advantage Fit.
Clinical highlights
June 2017, Chevrot et al., published in International Urogynecology Journal
May 2010, Lim et al., published in International Urogynecology Journal
Technical specifications
| Mesh | |
| Thickness | 0.66 mm |
| Pore size | 1182 μm |
| Fiber size (diameter) | 0.15 mm |
| Weight | 100 g/m2 |
| Length | 44.5 cm |
| Delivery | |
| Advantage Fit™ needle diameter | 2.7 mm |
| Sleeve length | 22.25 cm per side |
| Dilator length | 17 cm |
| Overall length | 83.5 cm |
Education and training for urology
Online medical training and education courses
The EDUCARE online platform makes medical education and training relevant, comprehensive, personal, and accessible. Register to access procedural videos, case studies, and training resources.
Product brochure
Ordering information
UPN | Description | Quantity |
M0068502120 | Advantage Fit™ Blue System | 1 delivery device and 1 mesh assembly (blue) |
M0068502110 | Advantage Fit System | 1 delivery device and 1 mesh assembly (clear) |
All images are owned by Boston Scientific. All trademarks are the property of their respective owners.
For female Mid-Urethral Slings: CAUTION: Federal (US) law restricts this device to sale by or on the order of a physician trained in the use of surgical mesh for repair of stress urinary incontinence.
Refer to package insert provided with this product for complete Indications for Use, Contraindications, Warnings, Precautions, Adverse Events, and Instructions prior to using this product.
The following adverse events have been reported due to suburethral sling placement, any of which may be ongoing, but are not limited to: As with all implants, local irritation at the wound site and/or a foreign body response may occur, Foreign body reaction may be acute or chronic, Pain (pelvic, vaginal, groin/thigh, suprapubic, dyspareunia) (acute or chronic), Dyspareunia, Tissue responses to the mesh implant could include: erosion into organs (urethra, bladder or other surrounding tissues); exposure/extrusion into the vagina, Mesh contact with urine via erosion/exposure/extrusion may result in stone formation, scarring/scar contracture, Necrosis, fistula formation (acute or chronic), inflammation (acute or chronic), Mesh contracture, Tissue contracture, Vaginal shortening or stenosis that may result in dyspareunia and/or sexual dysfunction, Pain with intercourse that may not resolve, Exposed mesh may cause pain or discomfort to the patient’s partner during intercourse, Sexual dysfunction, including the inability to have intercourse. Like all foreign bodies, the mesh may potentiate an existing infection. Allergic reaction has been reported. Known risks of surgical procedures for the treatment of incontinence include: pain, ongoing pain (pelvic, vaginal, groin/thigh, suprapubic, dyspareunia), Severe, chronic pain, Apareunia, Leg weakness, Infection, De novo detrusor instability, Complete failure of the procedure/failure to resolve a patient’s stress urinary incontinence, Voiding dysfunction (incontinence, temporary or permanent lower urinary tract obstruction, difficulty urinating, pain with urination, overactive bladder, and retention), Bruising, bleeding (vaginal, hematoma formation), Abscess, Vaginal discharge, Dehiscence of vaginal incision, Edema and erythema at the wound site, Perforation or laceration of vessels, nerves, bladder, urethra or bowel may occur during placement. The following additional adverse events have been reported for the Solyx SIS System: Dysuria, Hematuria. The occurrence of these events may require surgical intervention and possible removal of the entire mesh. In some instances, these events may persist as a permanent condition after surgical intervention or other treatment. Removal of mesh or correction of mesh-related complications may involve multiple surgeries. Complete removal of mesh may not be possible and additional surgeries may not always fully correct the complications.