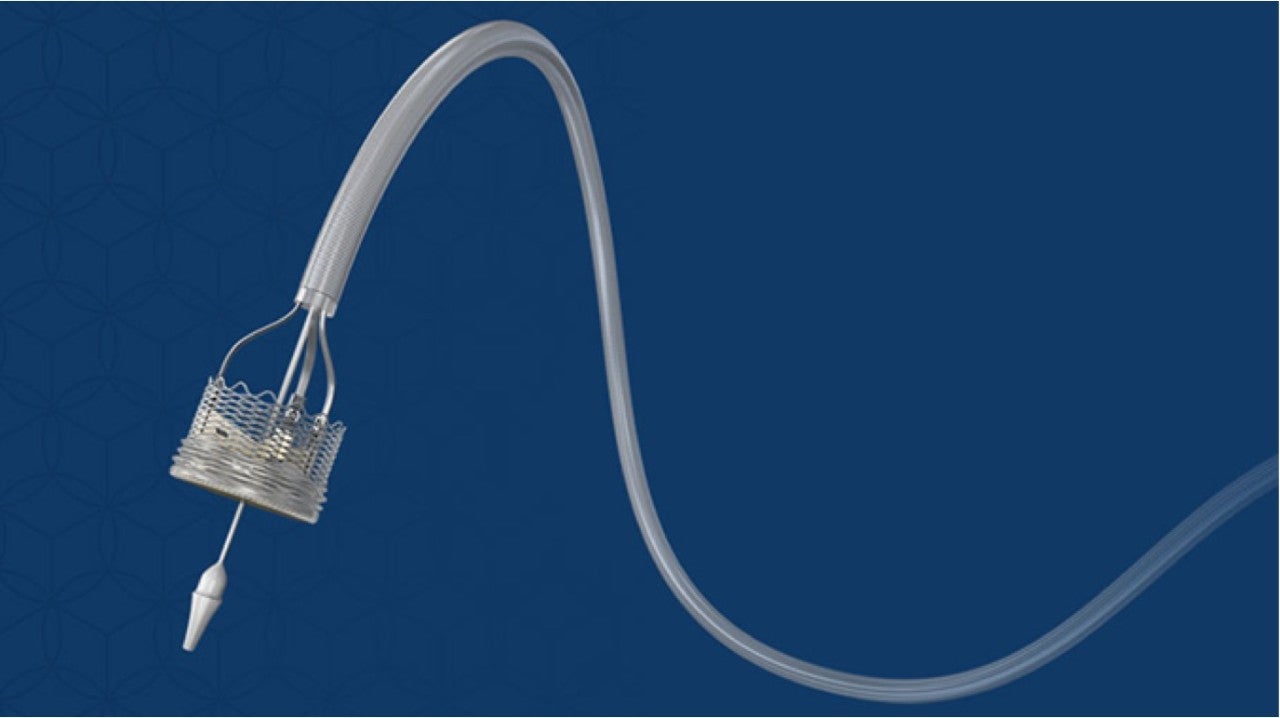
Boston Scientific announced a global, voluntary recall of the LOTUS Edge™ Aortic Valve. This voluntary recall is related solely to the complexities of the delivery system and is not related to the implanted valve. At this time, there are no known safety concerns for patients who have the LOTUS Edge valve implanted. The valve continues to show clinically effective performance post-implant.
For more, read a letter from Dr. Ian Meredith, Boston Scientific’s Global Chief Medical Officer. Read Now.