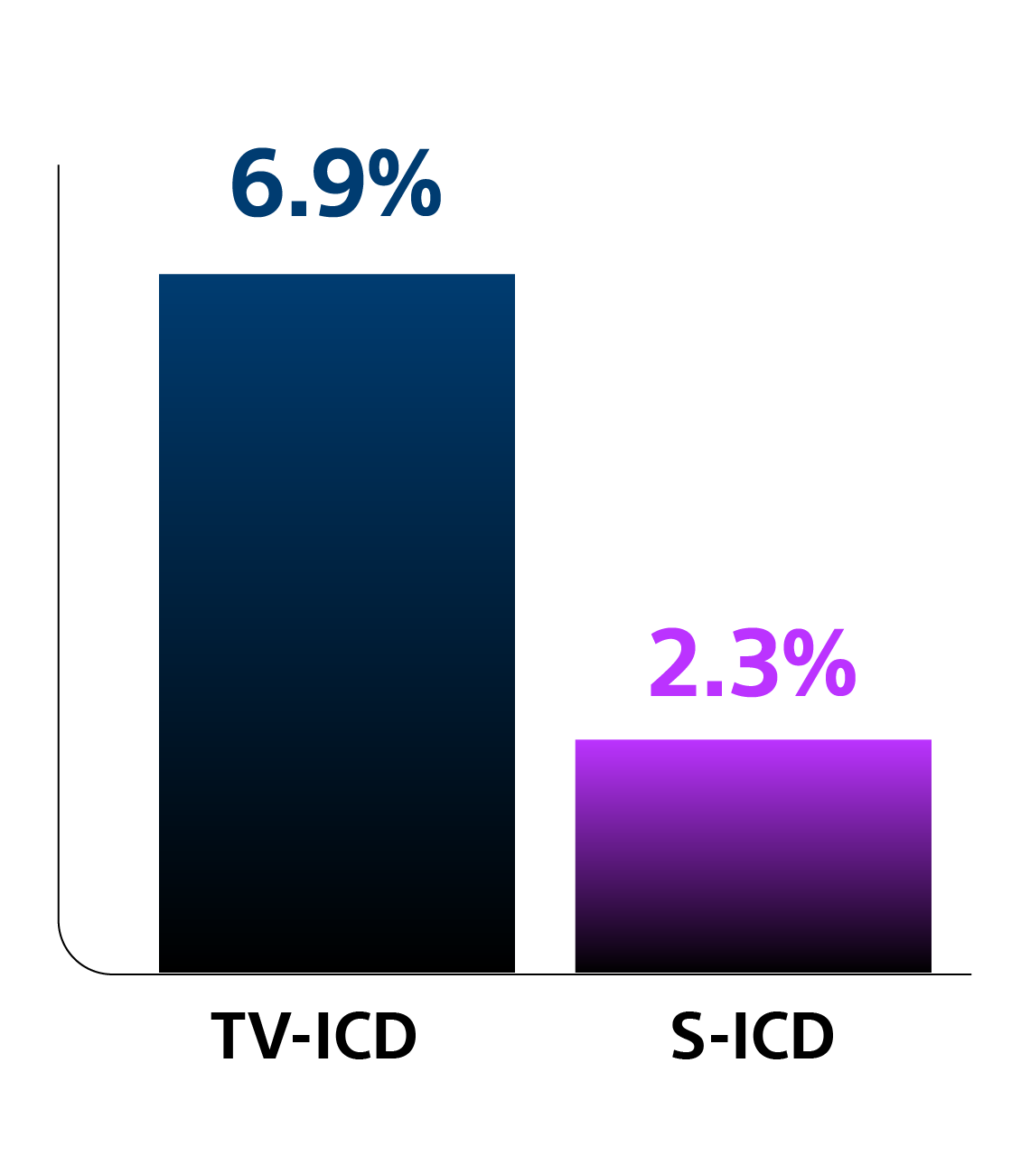ATLAS Trial1
Head-to-head Trial: Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) vs the Transvenous ICD (TV-ICD)
A prospective, randomised, controlled trial comparing lead-related complication rates between the S-ICD and single-chamber TV-ICDs at six months post-implant.
Patient Population
•Total: N=503
•S-ICD group: n=251
•TV-ICD group: n=252
Key Results
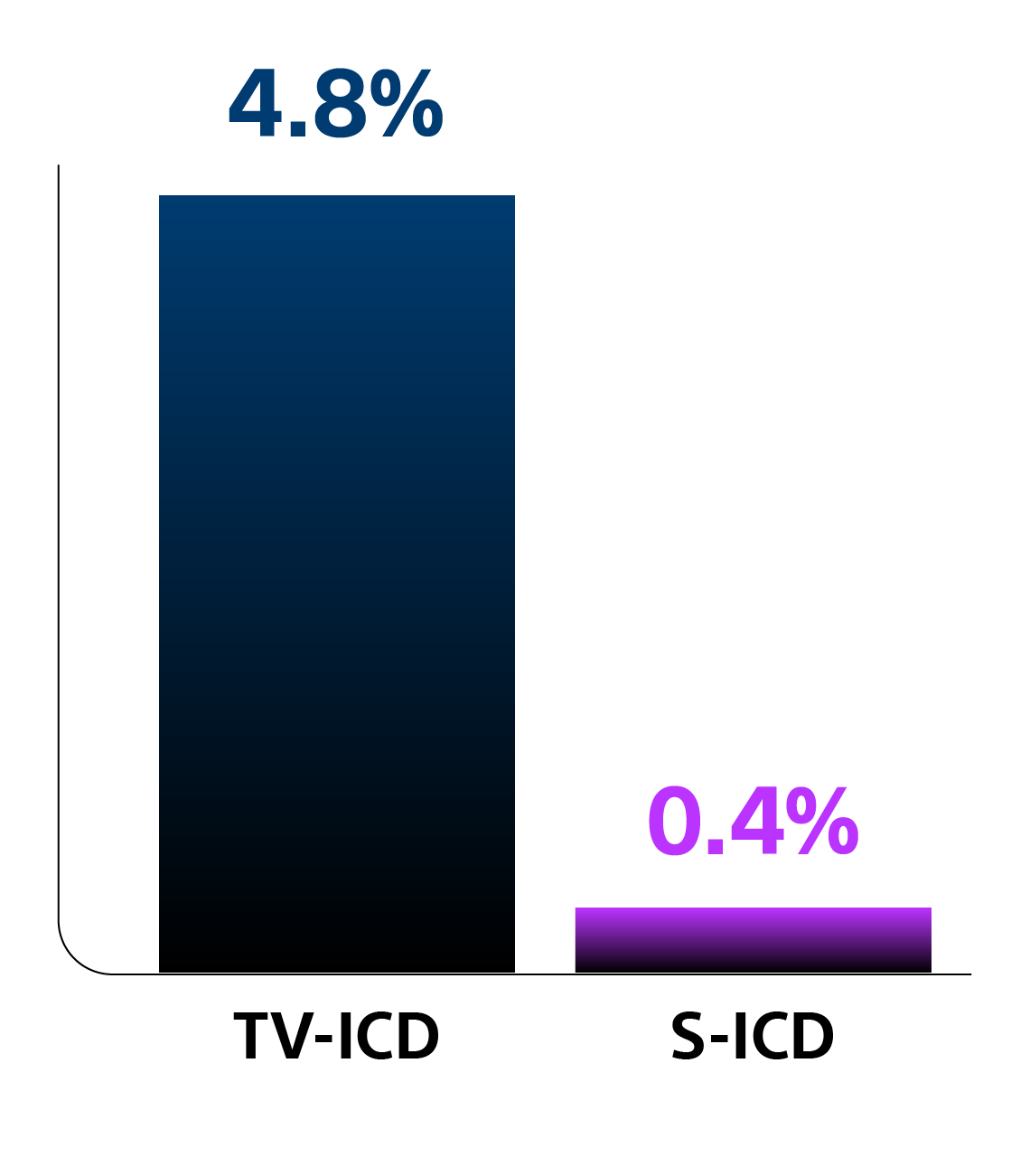
Secondary Analysis2
Based on data from nearly 450 patients, this analysis assessed the severity of tricuspid regurgitation (TR) six months post-implant.
Key Results
Patients with an S-ICD were seven times less likely to experience worsening TR compared to those with a TV-ICD (p<0.001).