* Caution: Investigational Device. Limited by US law to investigational use only. Not available for sale.
** The manufacturer recommended VF conversion testing during EMBLEM S-ICD implant, replacement, and concomitant device implants is being evaluated based on the PRAETORIAN DFT trial results and additional available data.
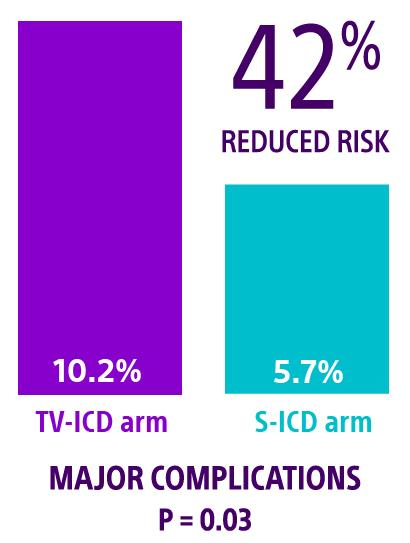
† In the ATLAS trial, serious complications were defined as moderate-severe or severe tricuspid regurgitation, hemothorax/pneumothorax, cardiac perforation, tamponade, pericardial effusion or pericarditis, ipsilateral upper extremity deep vein thrombosis and lead dislodgement or loss of sensing or pacing requiring revision.
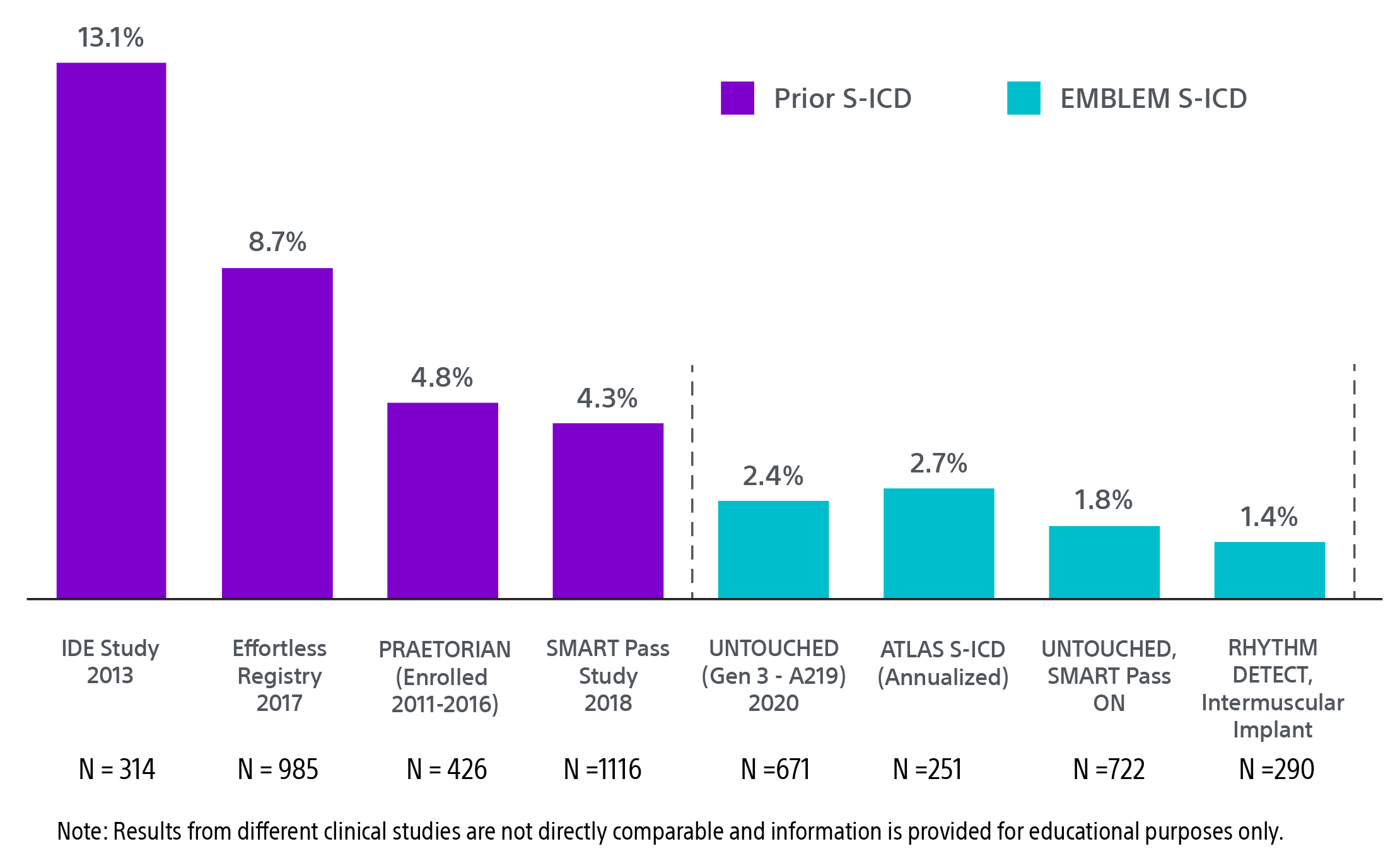
‡ IAS graph references for S-ICD (see at bottom of Clinical data tab)
IDE study: Weiss R, Knight BP, Gold MR, et al. Safety and efficacy of a totally subcutaneous implantable-cardioverter defibrillator. Circulation. 2013;128(9):944-953. doi:10.1161/circulationaha.113.003042
Effortless Registry: Lambiase PD, Theuns DA, Murgatroyd F, et al. Subcutaneous implantable cardioverter-defibrillators: long-term results of the EFFORTLESS study. Eur Heart J. 2022;43(21):2037-2050. doi:10.1093/eurheartj/ehab921
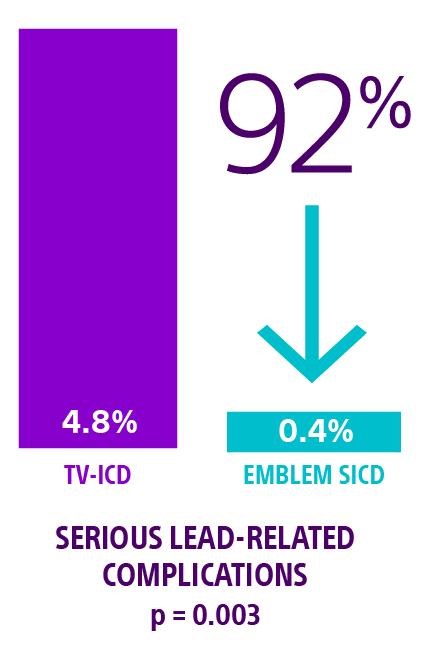
PRAETORIAN: Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526-536. doi:10.1056/NEJMoa1915932
SMART Pass: Theuns D, Brouwer TF, Jones PW, et al. Prospective blinded evaluation of a novel sensing methodology designed to reduce inappropriate shocks by the subcutaneous implantable cardioverter-defibrillator. Heart Rhythm. 2018;15(10):1515-1522. doi:10.1016/j.hrthm.2018.05.011
UNTOUCHED Gen 3: Gold MR, Lambiase PD, El-Chami MF, et al. Primary results from the Understanding Outcomes With the S-ICD in Primary Prevention Patients With Low Ejection Fraction (UNTOUCHED) trial. Circulation. 2021;143(1):7-17. doi:10.1161/CIRCULATIONAHA.120.048728
ATLAS S-ICD: Healey JS, Krahn AD, Bashir J, et al. Perioperative safety and early patient and device outcomes among subcutaneous versus transvenous implantable cardioverter defibrillator implantations: a randomized, multicenter trial. Ann Intern Med. 2022;175(12):1658-1665. doi:10.7326/M22-1566
UNTOUCHED SMART Pass: Boersma LVA, Aasbo J, Knops RE, et al. The impact of SMART pass algorithm status on inappropriate shock rates in the UNTOUCHED study. Europace. 2022;24(suppl 1):euac053.391. doi:10.1093/europace/euac053.391
RHYTHM DETECT: Botto GL, Ziacchi M, Nigro G, et al. Intermuscular technique for implantation of the subcutaneous implantable defibrillator: a propensity-matched case–control study. Europace. 2023;25(4):1423-1431. doi:10.1093/europace/euad028
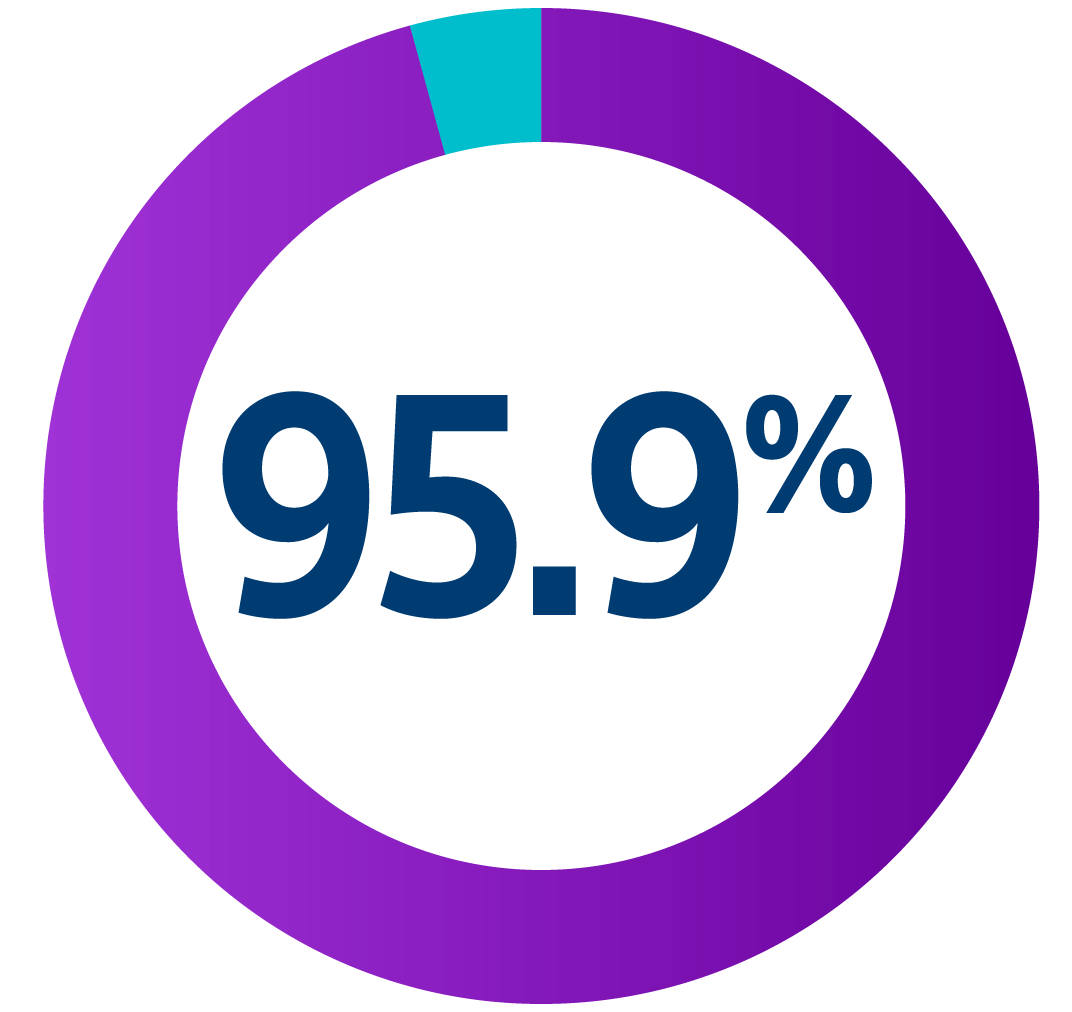
§ Communication testing assessed a pass/fail of communication at the programmed S-ICD telemetry setting and was required at the 6-month visit in four body postures. The test had to pass in more than 88% of attempts, across all postures, to be defined as successful.
‖ For full warranty terms and conditions go to bostonscientific.com/en-US/pprc/warranty-info-forms.html
REFERENCES:
- Healey JS, Krahn AD, Bashir J, et al. Perioperative safety and early patient and device outcomes among subcutaneous versus transvenous implantable cardioverter-defibrillator implantations: a randomized, multicenter trial. Ann Intern Med. 2022;175(12):1658-1665. doi:10.7326/M221-566.
- Olde Nordkamp LRA, de Veld JA, Ghani A, et al. Device-related complications in transvenous versus subcutaneous defibrillator therapy during long-term follow-up: the PRAETORIAN-XL trial. Circulation. 2025;0(0). doi:10.1161/CIRCULATIONAHA.125.074576.
- Leong DP, Dokainish H, Mondesert B, et al. Effects of implantable cardioverter-defibrillator leads on the tricuspid valve and right ventricle: a randomized trial. JACC Clin Electrophysiol. 2024;10(9):2088-2096. doi:10.1016/j.jacep.2024.04.034.
- Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary. Circulation. 2018;138(13):e210-e271. doi:10.1161/CIR.0000000000000548.
- Russo AM, Desai MY, Do MM, et al. ACC/AHA/ASE/HFSA/HRS/SCAI/SCCT/SCMR 2025 appropriate use criteria for implantable cardioverter-defibrillators, cardiac resynchronization therapy, and pacing. J Am Coll Cardiol. 2025;85(11):1213-1285. doi:10.1016/j.jacc.2024.11.023.
- Bardy G, Smith W, Hood M, et al. An entirely subcutaneous implantable cardioverter-defibrillator. N Engl J Med. 2010;363(1):36-44.
- Gold MR, El-Chami MF, Burke MC, et al. Postapproval study of a subcutaneous implantable cardioverter-defibrillator system. J Am Coll Cardiol. 2023;82(5):383-397. doi:10.1016/j.jacc.2023.05.034.
- Gold MR, Lambiase PD, El-Chami MF, et al. Primary results from the UNTOUCHED trial. Circulation. 2021;143(1):7-17. doi:10.1161/CIRCULATIONAHA.120.048728.
- Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med. 2020;383(6):526-536. doi:10.1056/NEJMoa1915932.
- Knops RE, van der Stuijt W, Delnoy P, et al. Efficacy and safety of appropriate shocks and antitachycardia pacing in transvenous and subcutaneous ICDs: PRAETORIAN analysis. Circulation. 2022;145(5):321-329. doi:10.1161/CIRCULATIONAHA.121.057816.
- Lambiase PD, Theuns DA, Murgatroyd F, et al. Long-term results of the EFFORTLESS study. Eur Heart J. 2022;43(21):2037-2050. doi:10.1093/eurheartj/ehab921.
- Zeppenfeld K, Tfelt-Hansen J, de Riva M, et al. 2022 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death. Eur Heart J. 2022;43(40):3997-4126. doi:10.1093/eurheartj/ehac262.
- Payne JE, Gold MR. A substernal defibrillator lead with pacing capability. JACC Clin Electrophysiol. 2019;5(2):197-198. doi:10.1016/j.jacep.2018.12.006.
- Romers H, van Dijk V, Boersma L. Evolution of extravascular ICD therapy. Heart Rhythm O2. 2023;4(1):59-64. doi:10.1016/j.hroo.2022.09.021.
- Boston Scientific. CRM Product Performance Report. Q3 2024.
- Knops RE, Lloyd MS, Roberts PR, et al. A modular communicative leadless pacing-defibrillator system. N Engl J Med. 2024;391(15):1402-1412. doi:10.1056/NEJMoa2401807.
- Lloyd MS, Brisben AJ, Reddy VY, et al. Design and rationale of the MODULAR ATP global clinical trial. Heart Rhythm O2. 2023;4(7):448-456. doi:10.1016/j.hroo.2023.05.004.
- Boersma LVA, Aasbo J, Knops RE, et al. SMART Pass algorithm status and inappropriate shocks in UNTOUCHED. Europace. 2022;24(Suppl 1):euac053.39. doi:10.1093/europace/euac053.391.
- Boston Scientific. EMBLEM S-ICD and EMBLEM MRI S-ICD User’s Manual. 2021.
- van der Stuijt W, Williams JL, Brisben AJ, et al. Battery longevity and its predictors in subcutaneous implantable cardioverter-defibrillators (S-ICDs): A mathematical analysis derived from a large dataset. Heart Rhythm. Mar 2025;22(3):868-870. doi:10.1016/j.hrthm.2024.09.009.
- Akar J, et al. Use of remote monitoring and outcomes in ICD patients. Heart Rhythm Society Scientific Sessions. 2014;Abstract LB03-03.
- Botto GL, Ziacchi M, Nigro G, et al. Intermuscular S-ICD implantation: propensity-matched study. Europace. 2023;25(4):1423-1431. doi:10.1093/europace/euad028.
- Winter J, Siekiera M, Shin DI, et al. Long-term performance of intermuscular S-ICD implantation. Europace. 2017;19(12):2036-2041. doi:10.1093/europace/euw297.
- Brouwer TF, Miller MA, Quast ABE, et al. Evaluation of four S-ICD implantation techniques. Circ Arrhythm Electrophysiol. 2017;10:e004663. doi:10.1161/CIRCEP.116.004663.
- Droghetti A, Locatelli A, Casiraghi B, et al. Totally submuscular S-ICD implantation in obese patients. Clin Case Rep. 2016;4(10):1009-1011. doi:10.1002/ccr3.652.
- Migliore F, Allocca G, Calzolari V, et al. Intermuscular two-incision S-ICD technique: registry results. Pacing Clin Electrophysiol. 2017;40(3):278-285. doi:10.1111/pace.12987.
- Knops RE, Olde Nordkamp LR, de Groot JR, et al. Two-incision S-ICD implantation technique. Heart Rhythm. 2013;10:1240-1243. doi:10.1016/j.hrthm.2013.05.016.
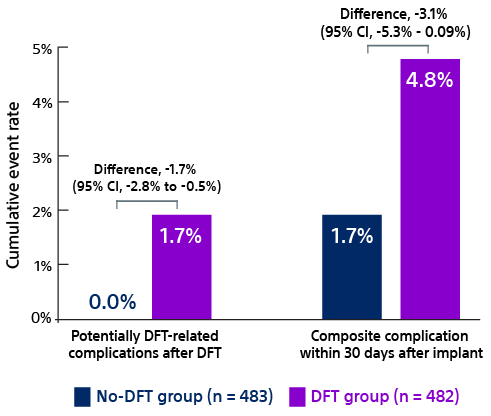
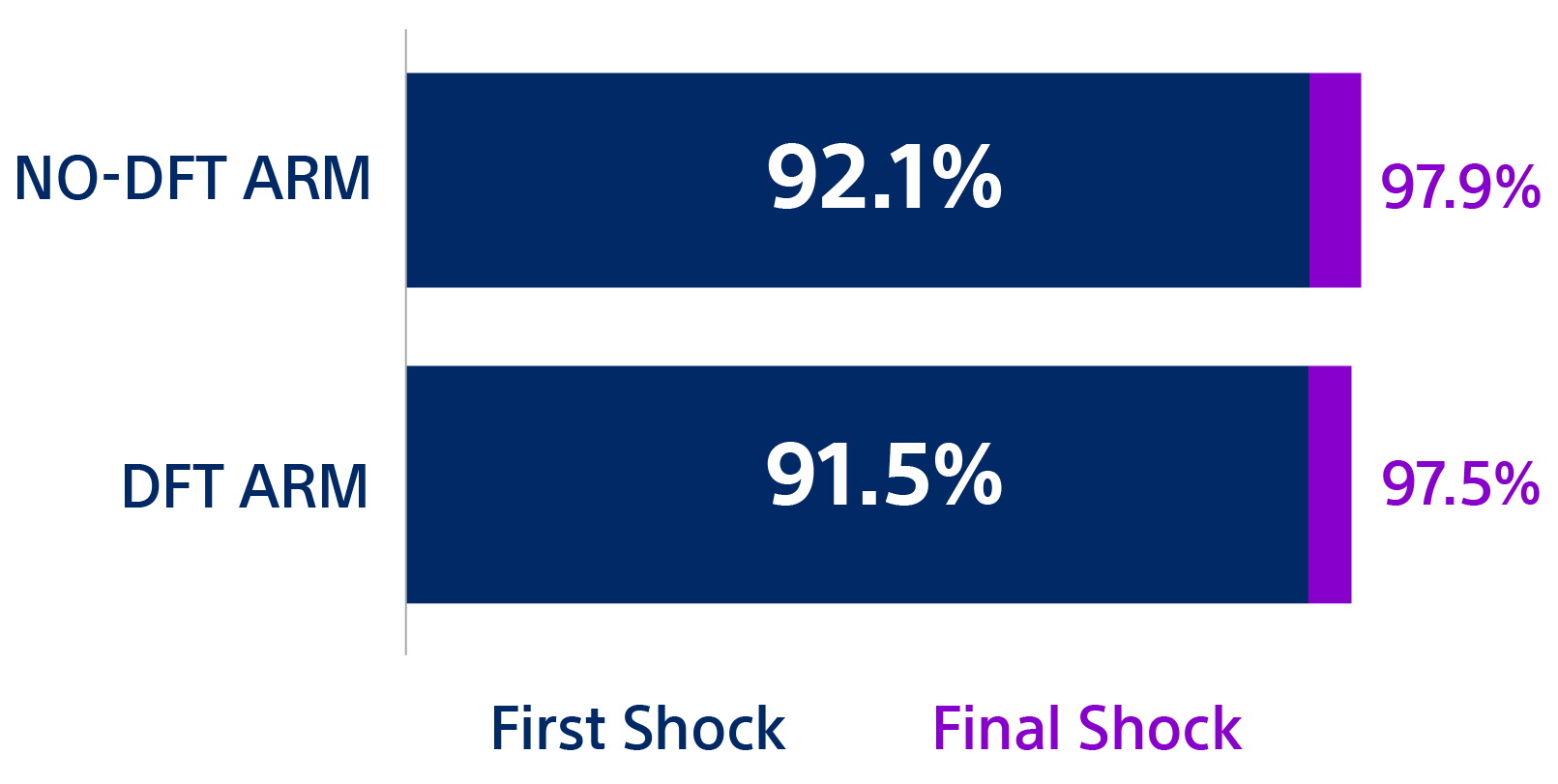
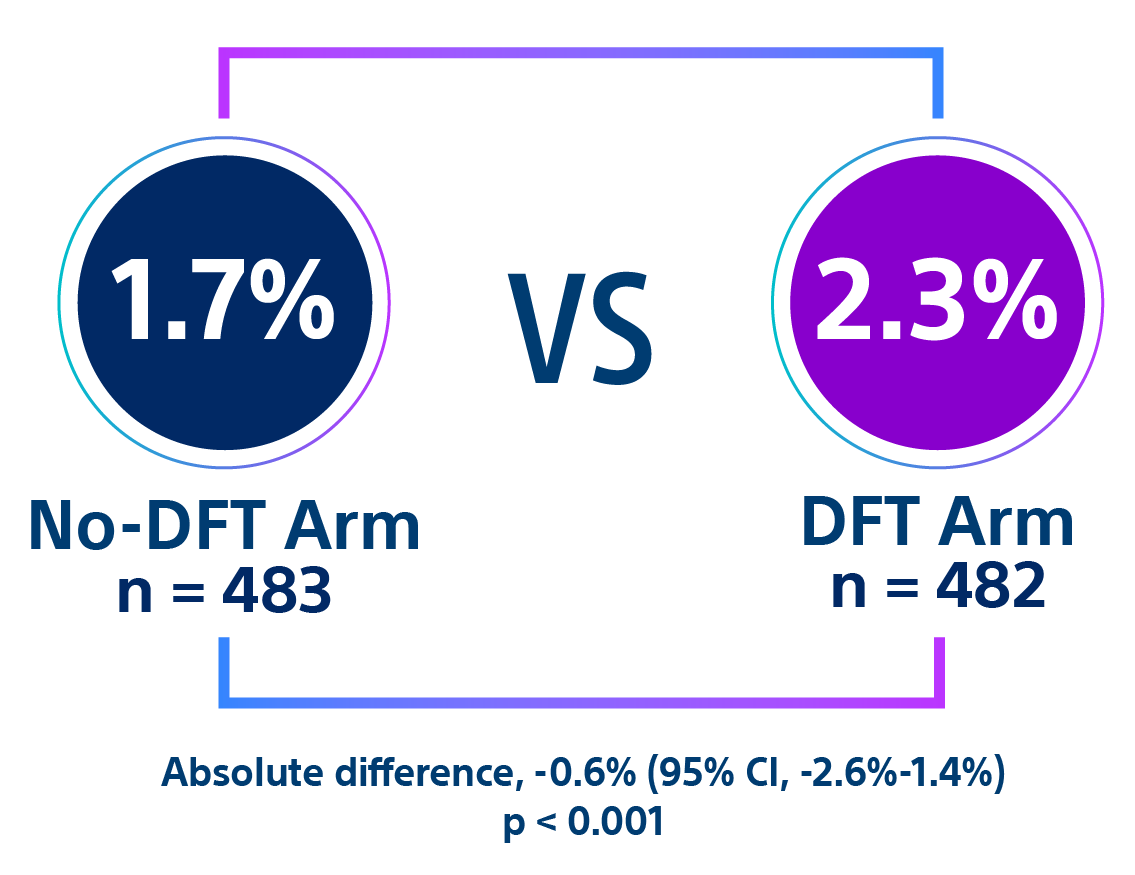
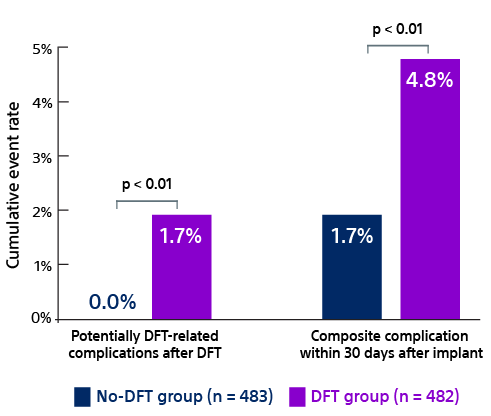
- Quast ABE, Baalman SWE, Betts TR, et al. Rationale and design of the PRAETORIAN-DFT trial: A prospective randomized CompArative trial of SubcutanEous ImplanTable CardiOverter-DefibrillatoR ImplANtation with and without DeFibrillation testing. Am Heart J. 2019;214:167-174. doi:10.1016/j.ahj.2019.05.002.
- Knops R. A randomized comparison of subcutaneous implantable cardioverter defibrillator implantation with or without defibrillation testing: the primary results of the PRAETORIAN-DFT Trial. HRS 2026. LB-525395.
- Friedman P, Murgatroyd F, Boersma LVA, et al. Chronic safety and performance of the EV-ICD: pivotal study. Heart Rhythm. 2023;20(7):1091-1092. doi:10.1016/j.hrthm.2023.04.038.
- Schuger C, Joung B, Ando K, et al. APPRAISE ATP randomized clinical trial. JAMA. 2024;332(20):1723-1731. doi:10.1001/jama.2024.16531.
- Lloyd MS, Reddy VY, Roberts P, et al. One-year outcomes of the MODULAR ATP trial. Circ Arrhythm Electrophysiol. 2025. doi:10.1161/circep.125.014395.