AGENT™
Drug-Coated Balloon
Optimized Drug Transfer
- TransPaxTM coating ensures targeted delivery of Paclitaxel2.
- Optimal dose of 2 µg/mm2 compared to 3 µg/mm2 of market leading DCBs3.
Tissue Absorption
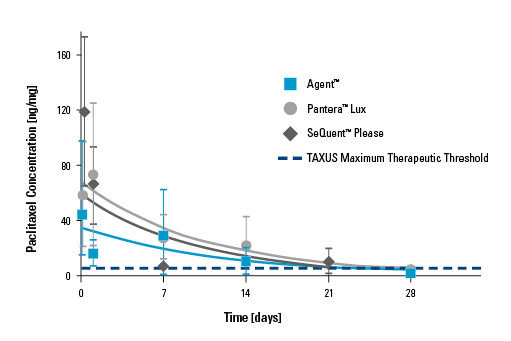
Drug Dissolution
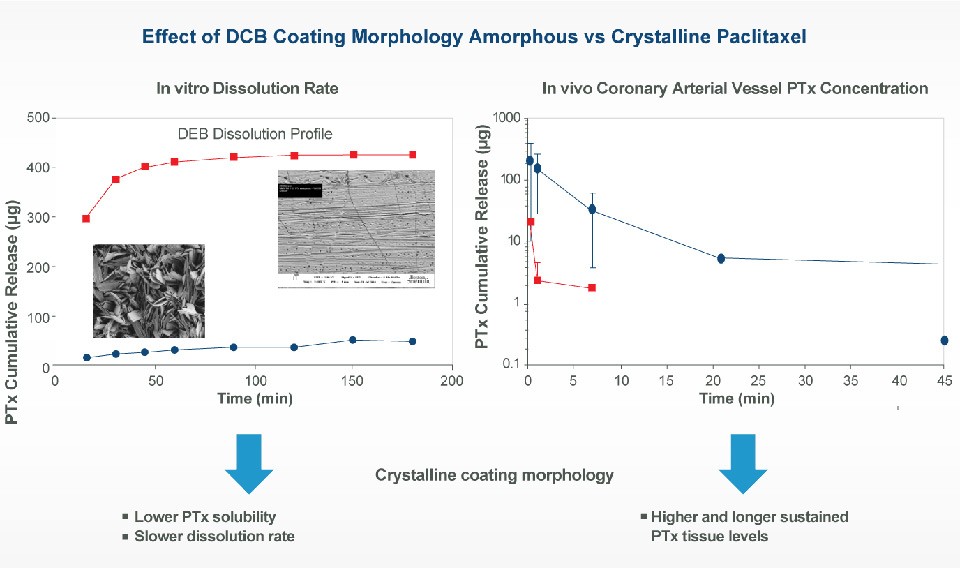
Drug-Coated Balloon

