Early neo2 Registry core-lab results
First large, real-world clinical data on the ACURATE neo2™ Aortic Valve System*: The Early neo2 Registry
Independently studying 85% of the original study’s 554 patients, the Early neo2 Registry core-lab analysis confirmed minimized paravalvular leak rates with the ACURATE neo2 Valve and its Active PVsealTM sealing skirt.
6.0%
new permanent pacemaker
2.7%
moderate to severe PVL1
8.0mmHg
mean gradient
See presentation replay
COMALIGN Study
First real-world analysis of patient-specific implantation techniques using different devices. The COMALIGN study demonstrated that neo-commissural alignment and optimized valve design can improve procedural success.
Best-in-class commissural alignment: ACURATE neo2 was the only self-expanding valve to 100% avoid commissural misalignment.2

0% moderate or severe commissural misalignment
ACURATE neo2 Valve
20/20 successful consecutive cases
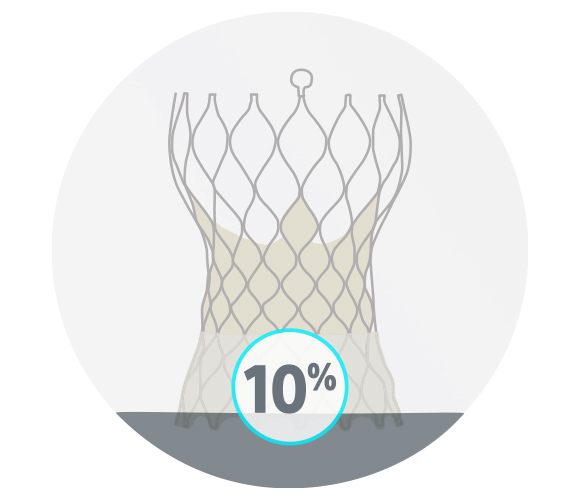
10% moderate or severe commissural misalignment
Evolut™ R/PRO Valve
17/20 successful consecutive cases; 3 cases with non-optimal rotations
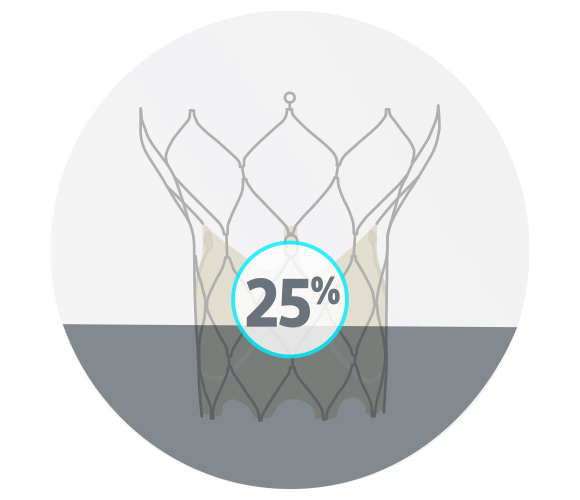
25% moderate or severe commissural misalignment
PORTICO™ Valve
15/20 successful consecutive cases; 5 cases difficult to assess
RE-ACCESS Study
First systematically evaluated analysis of coronary access before and after TAVI in an all-comers’ population
With 0 incidences of unsuccessful coronary cannulation, ACURATE neo demonstrated an extremely high coronary access success rate.
100%
ACURATE neo™ Valve Platform
100% successful coronary cannulation
n = 723
82%
Evolut™ R/PRO
82% successful coronary cannulation
n = 1233
99%
SAPIEN Platform
99% successful coronary cannulation
n = 963
1. Möllmann H, Holzhey DM, Hilker M, et al. The ACURATE neo2 valve system for transcatheter aortic valve implantation: 30-day and 1-year outcomes. Clin Res Cardiol. 2021;110(12):1912–1920.
2. Bieliauskas G, Wong I, Bajoras V, et al. Patient-specific implantation technique to obtain neo-commissural alignment with self-expanding transcatheter aortic valves. J Am Coll Cardiol 2021;14(19):2097-2108.
3. Reobtain Coronary Ostia Cannulation Beyond Transcatheter Aortic Valve Stent (RE-ACCESS); NCT04026204. J Am Coll Cardiol Intv. 2020.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings and instructions for use can be found in the product labelling supplied with each device. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France. Product available in the European Economic Area (EEA) only. Please check availability with your local sales representative or customer service
















