"With ACURATE neo2 we have the lowest PPI rate, always below 10%. We are having a great load of patients undergoing TAVR and we need to discharge them earlier. The very low rate of PPI is making our life easier for dealing with the logistics of this kind of patients."
Dr. Ignacio J. Amat Santos
The ACURATE neo2 Platform is designed to drive efficiency for the entire TAVI programme.
Treating more patients in an optimised environment and a more cost-efficient way is critical. Using the right valve can help. ACURATE neo2 allows you to broaden the treatable patient population, deliver faster and more efficient TAVI procedures with optimised outcomes, and drive towards safe and early patient discharge. 1-4
Faster, leaner and more efficient TAVI*¹⁺²

3.9 min
Average Device Usage Time1,2

97.5 %
High Procedural Success1,2
Leading the way to safe, early hospital discharge ¹⁺²
Procedural outcomes with the ACURATE Platform are associated with reductions in post-operative length of stay, which may reduce hospital costs, improve long-term clinical outcomes and enable you to treat more TAVI patients.
Early discharge with the ACURATE Platform means TAVI programme efficiency without compromising on safety so that your patients have less disruption and a return to their normal life as quickly as possible.

Reduced length of hospital stay
More patients discharged within 2 days vs Evolut R 5
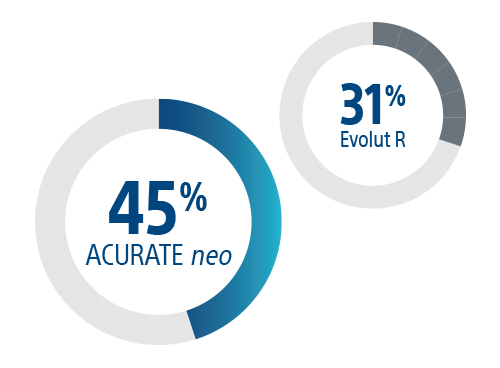
Safe early hospital discharge
More patients safely discharged the next day vs SAPIEN 3
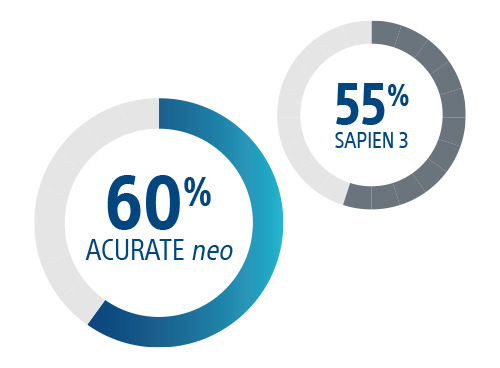
*compared to ACURATE neo
1+2. Möllmann, H., Holzhey, D.M., Hilker, M. et al. The ACURATE neo2 valve system for transcatheter aortic valve implantation: 30-day and 1-year outcomes. Clin Res Cardiol 110, 1912–1920 (2021).
3. Moriyama N, et al. Safety of Next-Day Discharge After Transfemoral Transcatheter Aortic Valve Replacement With a Self-Expandable Versus Balloon-Expandable Valve Prosthesis: ACURATE neo vs. SAPIEN 3. Cardiovascular Interventions. 2019;12.
4. Routine use of ACURATE neo self-expanding valve is associated with improved procedural outcomes and reduced per-operative length of stay: insights from a single-center registry. Presented at EuroPCR 2019 by R.A. Kotronias
5. Kotronias RA, Banning AP, Kharbanda RK. Oxford University Hospital. Presented at EuroPCR 2019 by R. A. Kotronias.
CAUTION: The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com.
Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries.
This material not intended for use in France.
















