Unlike DES, Drug Coated Balloons have a very short (30 seconds) vessel transfer window for the drug.
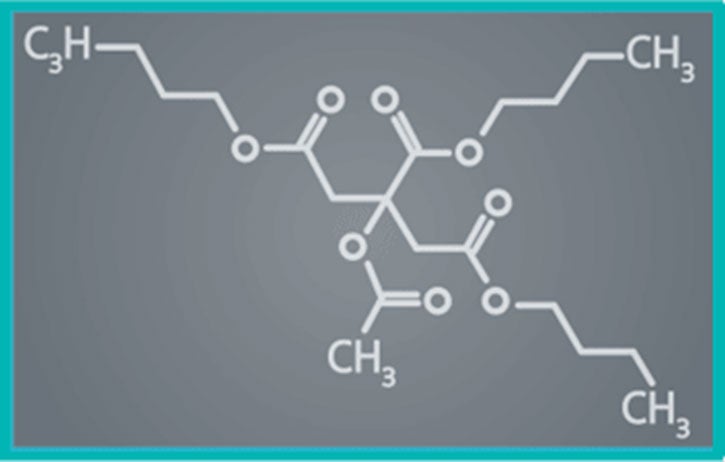
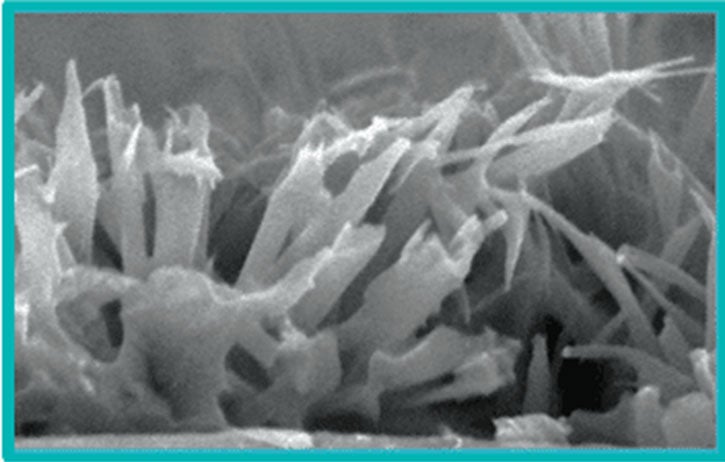
PTx is ideally suited as it is hydrophobic (highly durable during tracking), more lipophilic (rapidly absorbed into tissue) and chemically stable (long half-life provides sustained retention).
Paclitaxel drug has become the most extensively studied in coronary DCB therapy and remains the most widely used and trusted in clinical practice1.