ClinicalEVIDENCE newsletter #2 – May 2023
Welcome to the latest edition of ClinicalEVIDENCE, your go-to source for the latest clinical data on heart diseases, diagnostics, and monitoring.
Our aim is to provide you with cutting-edge information that will keep you up-to-date on the most recent advancements in the field of heart failure management.
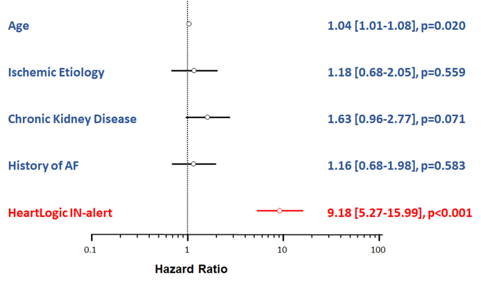
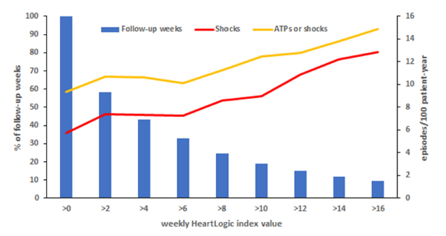
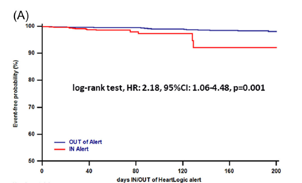
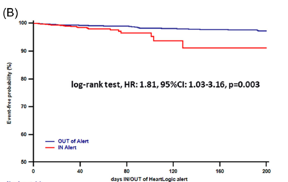
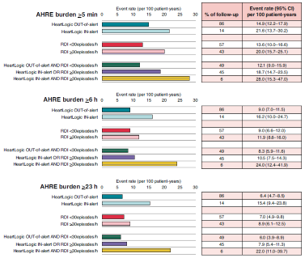
In this issue, we focus on HeartLogic tool, a multiparametric approach to heart failure diagnosis that is gaining increased attention in today’s world.
We hope you will find this issue informative and engaging, as we strive to deliver the most up-to-date and relevant content to our readers.