LUX-Dx™
ICM System
Signals that are astonishingly clear from the moment of insertion
Incorporating clear S-ECG signal quality from the moment of insertion, the LUX-Dx™ Insertable Cardiac Monitor (ICM) makes event reviews more efficient and confident. Signal quality is so clear that P-waves are visible in 90% of heart cycles and consistent over time1.
Key Resources
Explore
Product Details


1. ICM Device
The insertable cardiac monitor is a small, leadless electronic device inserted just under the skin in the left pectoral region. It uses two electrodes to monitor the patient’s subcutaneous ECG (S-ECG) data when specific arrhythmias are detected. Arrhythmia detection parameters are based on the Reason for Monitoring entered during device setup or can be programmed manually by the clinician. The device is MR-conditional.
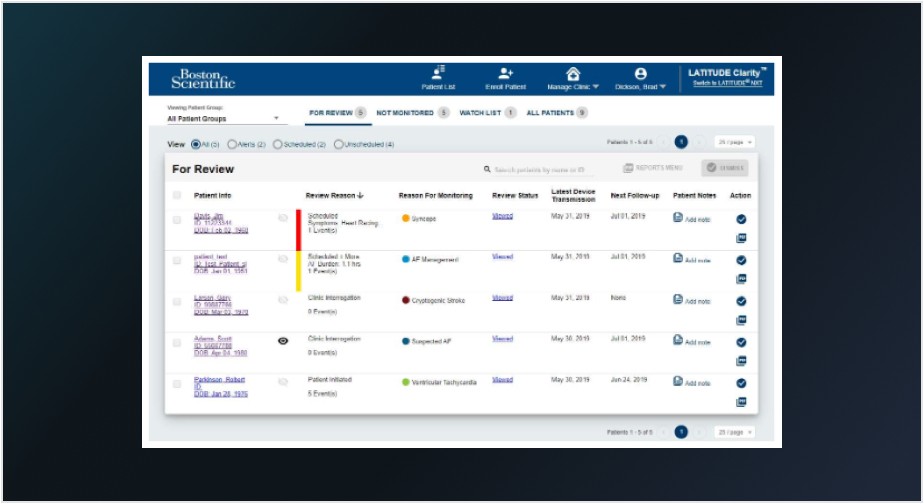
2. LATITUDE Clarity™ Data Management System
Built exclusively for the LUX-Dx ICM, the LATITUDE Clarity Data Management System helps clinics streamline their workflow, review data efficiently, accelerate critical decision making and adjust device settings remotely without bringing patients in for another appointment.
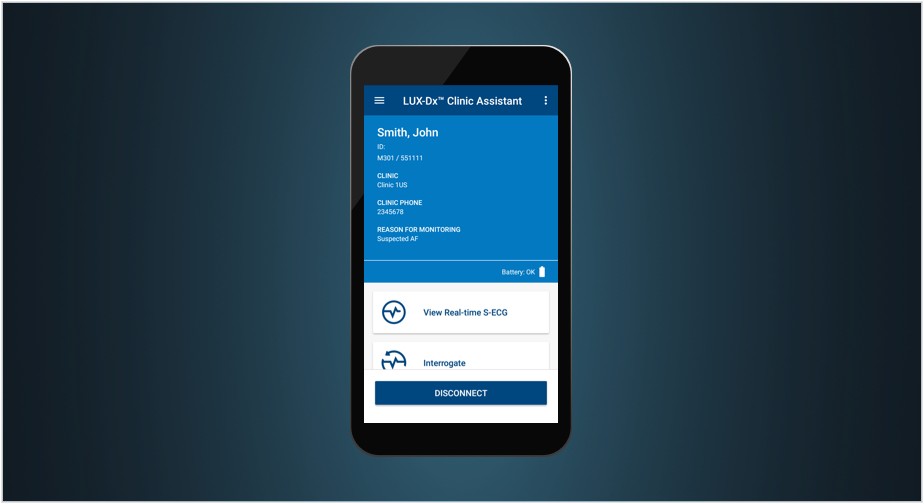
3. LUX-Dx™ Clinic Assistant App
Designed to enhance workflow and maximize efficiencies, the LUX-Dx Clinic Assistant app lets you:
- Connect to any LUX-Dx ICM device within 6 feet (2 meters)
- View ICM device status and real-time S-ECG
- Interrogate LUX-Dx ICM devices
- Apply programming changes
LUX-Dx ICM Product Specifications
| Parameters | Value |
|---|---|
| Volume | 1.2 cm3 |
| Mass | 3 g |
| Dimensions W X H X D | 7.2 mm x 44.8 mm x 4.0 mm |
| Surface Area of Can Electrode | 75.3 mm2 |
| Surface Area of Header Electrode | 10.2 mm2 |
| Battery Longevity | 3 years* |
*3 years projected longevity, under the following usage scenarios:
- Average of 1 auto-detected event per day
- Average of 1 patient-initiated event per month
- Less than or equal to 6 months shelf life between device manufacture and insertion
Notes:
- At the maximum shelf storage time of 18 months, longevity is reduced by approximately 4 months
- Projected longevity is 2 years when the Bluetooth Manual Connection is configured to not require a magnet
Implant Procedure
See how the LUX-Dx ICM was designed to offer a familiar insertion process to help you confidently implant the device.