The CHAMPION-AF clinical trial met its’ primary and secondary endpoints at 36 months.
The trial results showed WATCHMAN FLX™ provided statistically superior protection from bleeding and similar efficacy compared to NOACs in patients with NVAF.
The CHAMPION-AF clinical trial met its’ primary and secondary endpoints at 36 months.
The trial results showed WATCHMAN FLX™ provided statistically superior protection from bleeding and similar efficacy compared to NOACs in patients with NVAF.
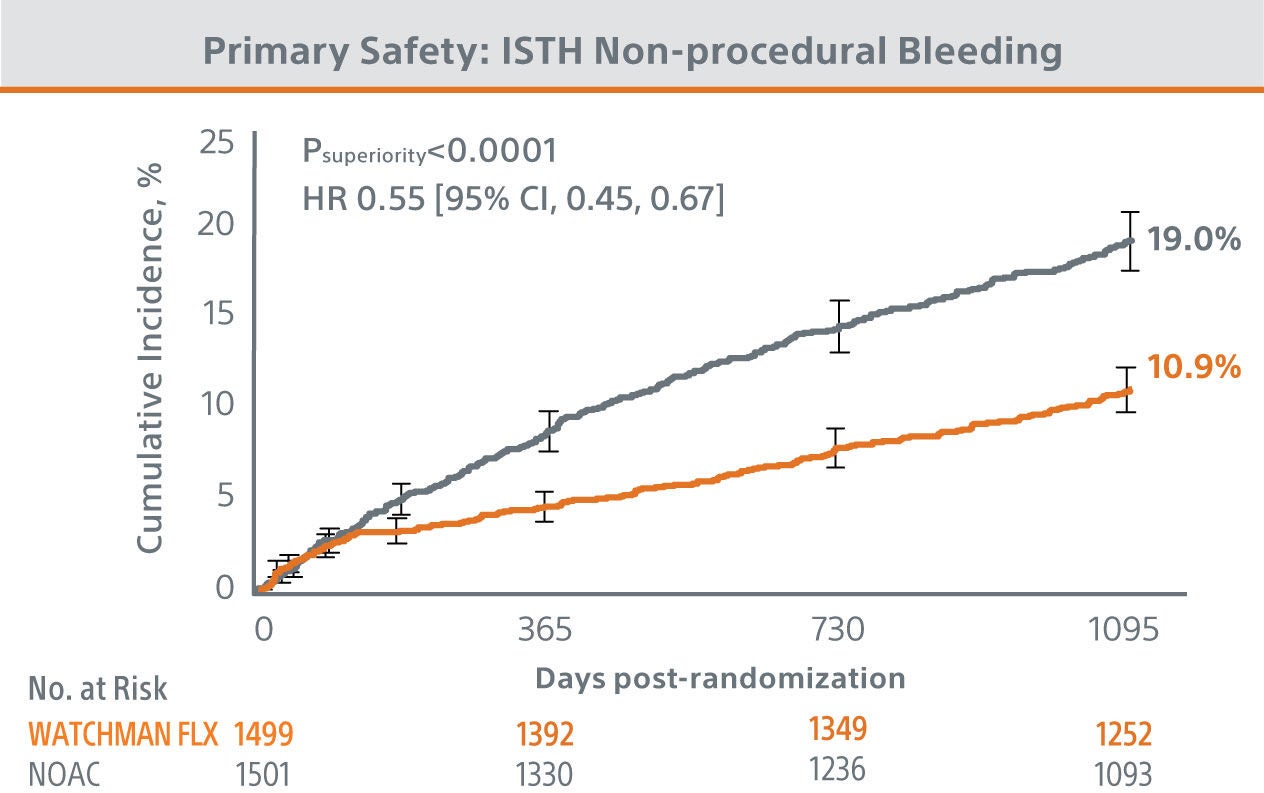
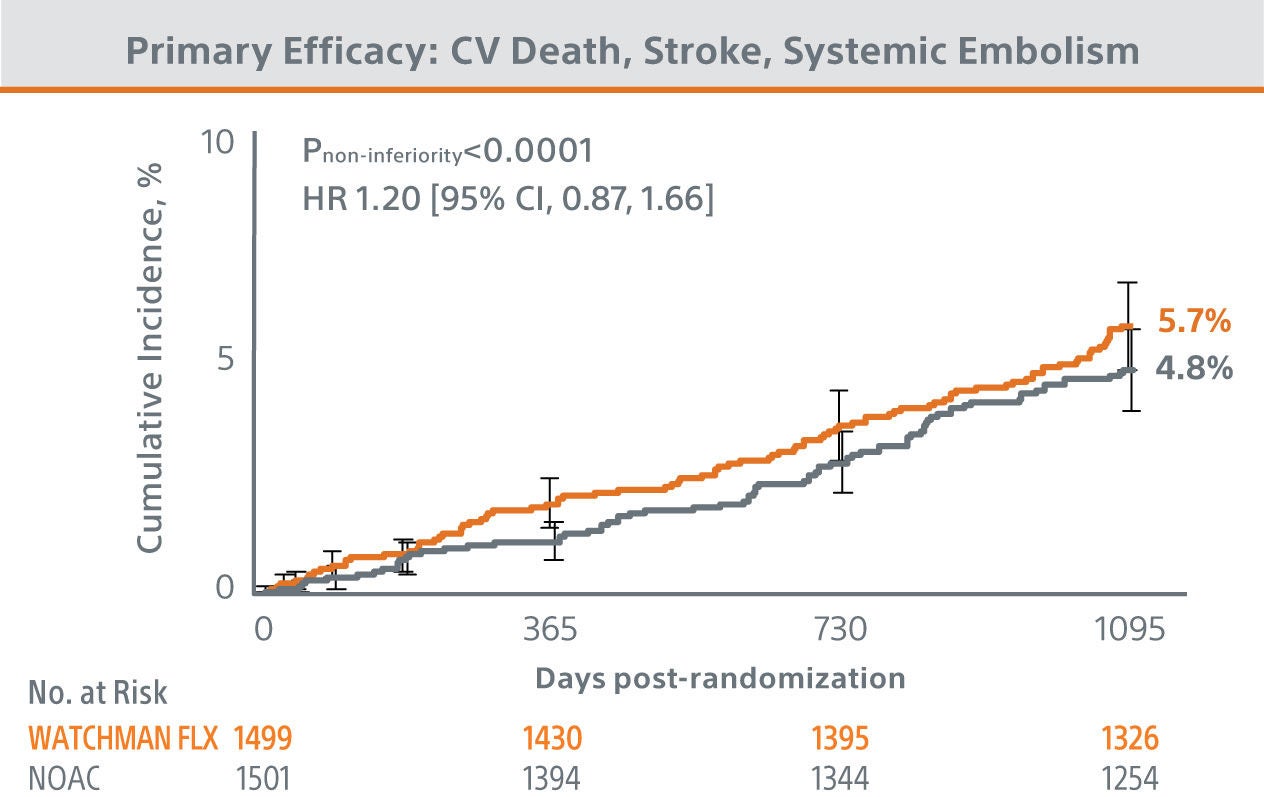
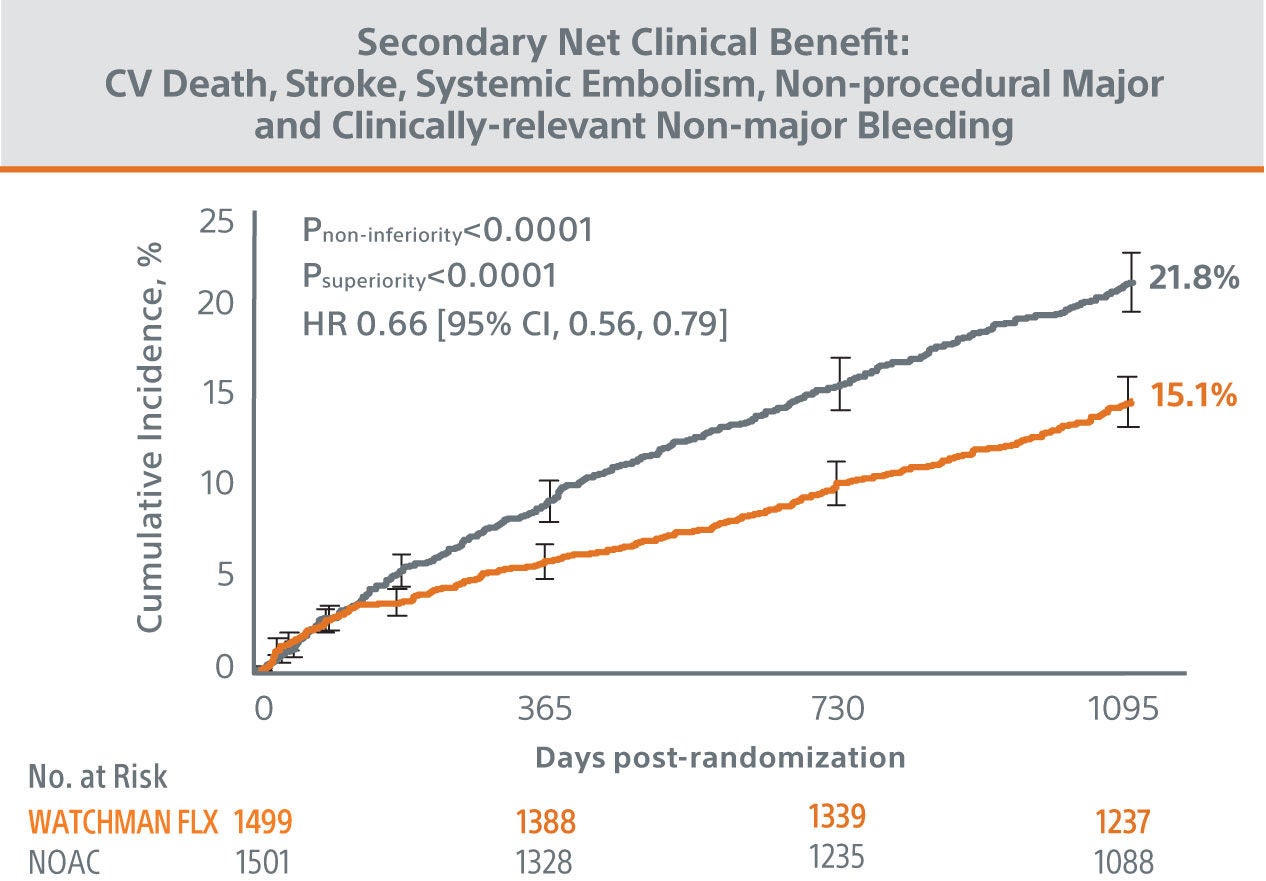
*Modified ISTH clinically relevant non-major bleeding: does not fit criteria for the ISTH definition of major bleeding but requires hospitalization or increased level of care.
References
S.K. Doshi, M.D. et.al. Left Atrial Appendage Closure or Anticoagulation for Atrial Fibrillation. The New England Journal of Medicine. (2026). https://www.nejm.org/doi/full/10.1056/NEJMoa2517213
CAUTION:
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.