5.5 million patients
are affected by
NVAF* worldwide1
NVAF patients have a
2-7X greater risk of
ischemic stroke2
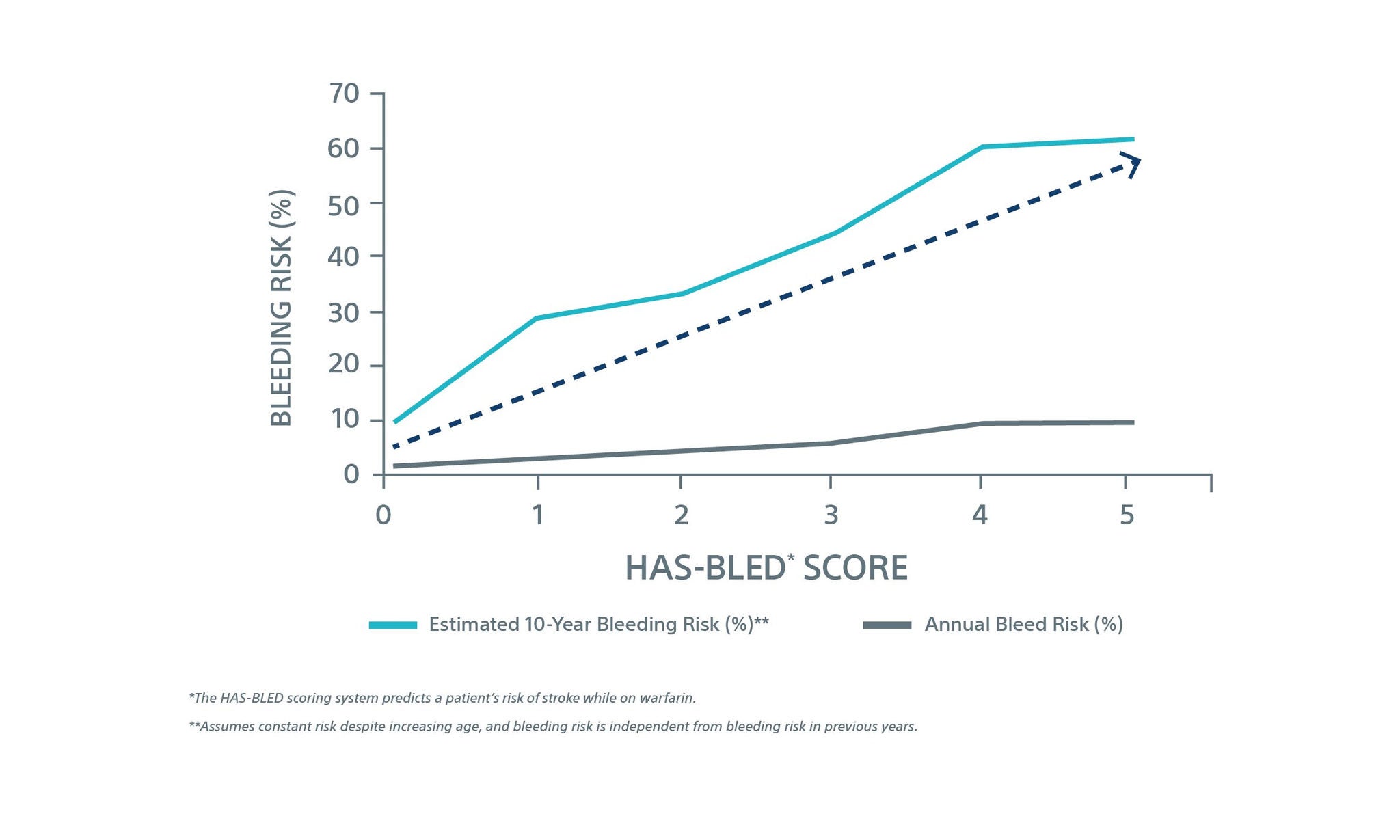
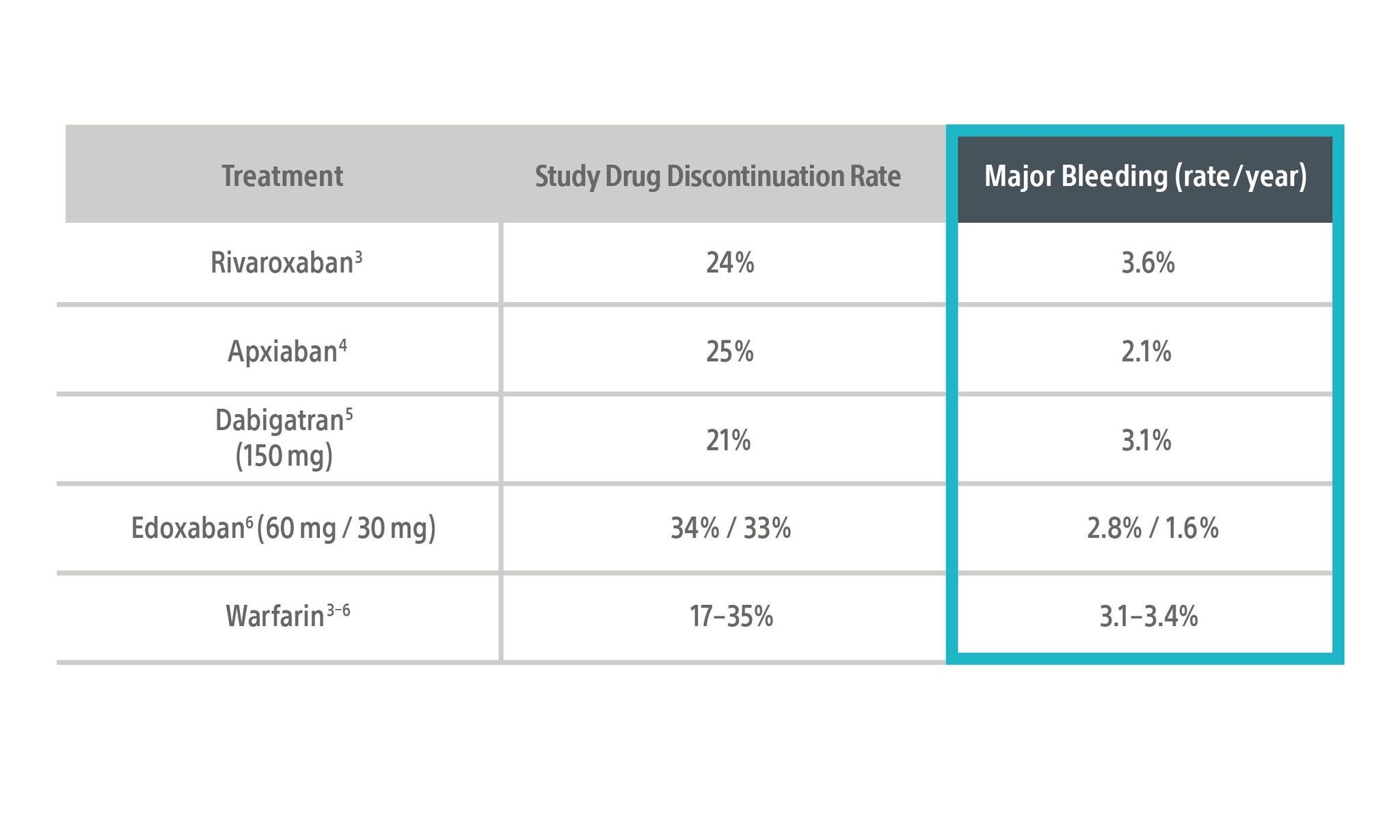
Current guidelines state that non-vitamin K antagonist oral anticoagulant (NOAC) therapy is the first line option for treatment of stroke risk reduction in non‑valvular atrial fibrillation (NVAF) patients. However, bleeding risk on these therapies compounds year over year, and are comparable across current pharmacotherapy options.