* Exclusion criteria includes patients who have had a prior major bleeding event in ISTH definition within the 30 days prior to randomization. Lack of resolution of related clinical sequelae or planned and pending interventions to resolve bleeding/bleeding source, are a further exclusion regardless of timing of the bleeding event.
CHAMPION-AF Clinical Trial
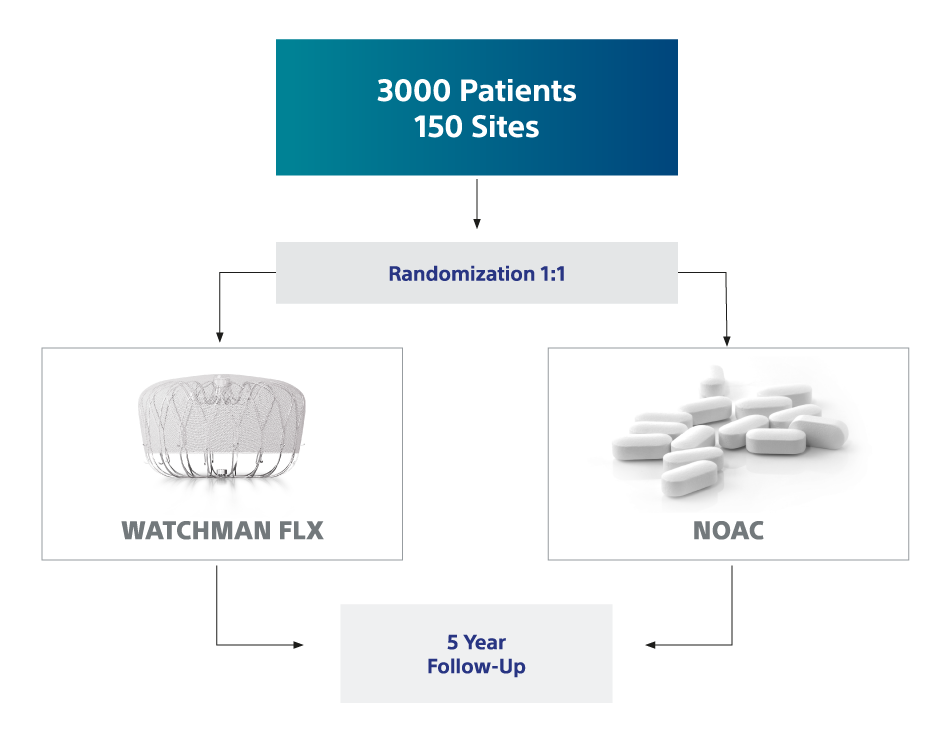
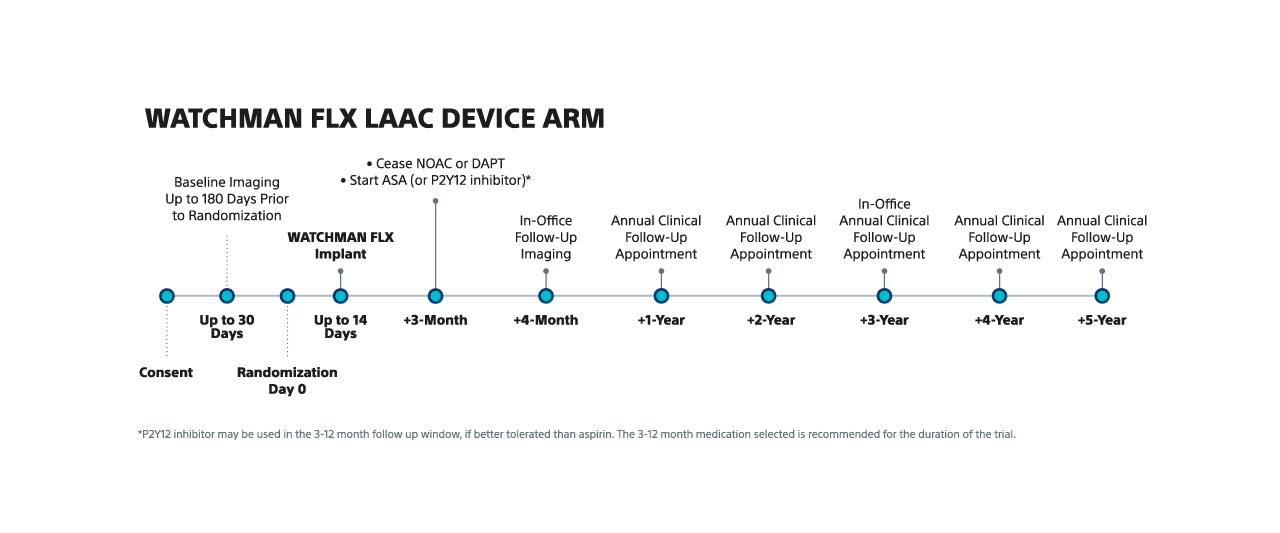
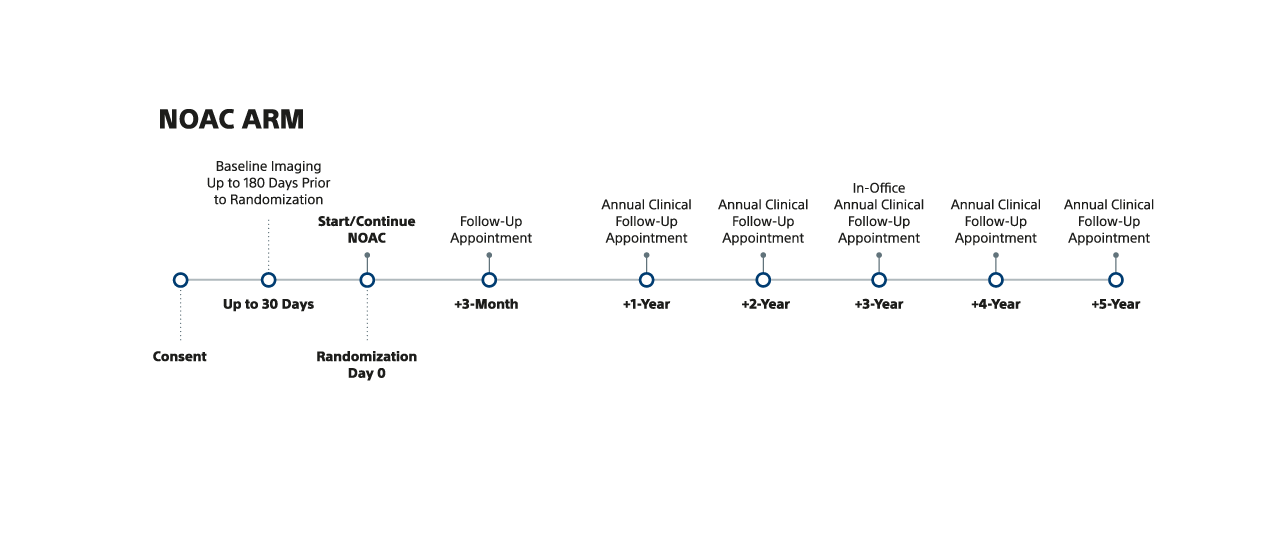
NOTE: For Device and NOAC arms, annual clinical follow-up visits can be via phone or in-office, with the exception of the 3-year clinical follow-up visit, which must be in-office.

CAUTION:
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.















