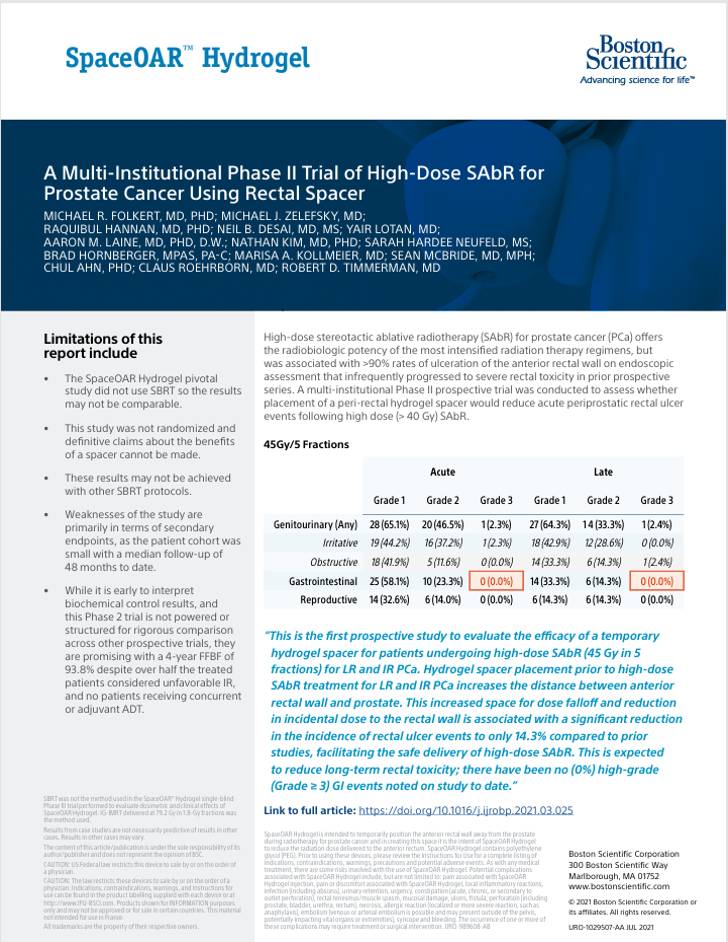
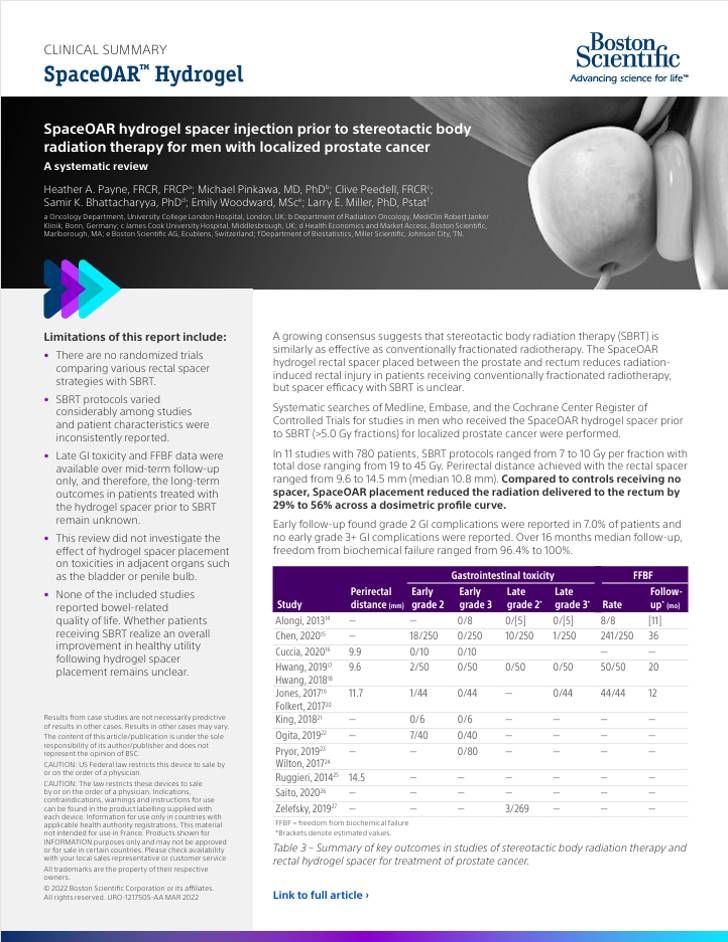
Clinical Summaries
SABRE TRIAL
Study objective
Primary endpoint:
- Proportion of patients experiencing late GI toxicity between 3 and 24 months after SBRT with or without placement of SpaceOAR Vue Hydrogel.
Secondary endpoint:
- Proportion of patients experiencing a decrease in Expanded Prostate Cancer Index Composite (EPIC-26) bowel score greater than or equal to the minimal important difference (MID) in EPIC-26 bowel score from baseline to 24 months post-SBRT initiation.