people globally have atrial fibrillation1

people worldwide affected by heart failure2,3

Sudden cardiac arrest is the third leading cause of death in Europe4-6
Utilising Boston Scientific technologies gives you access to on-demand support, via RhythmCARETM, expertise, with ADVANTICSTM, and learning through EDUCARETM .
What makes us different?
Our solutions across the entire patient pathway
Our Digital Solutions
We innovate driven by clinical need
We are continuously evolving our portfolio of devices and digital solutions to meet your complex challenges
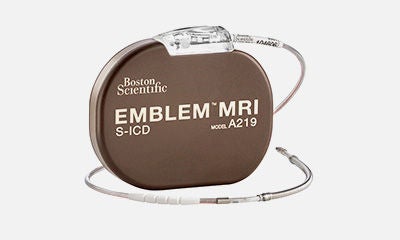
S-ICD past, present and future
The EMBLEM™ MRI S-ICD, is the only complete extrathoracic device proven to avoid the serious risks of more invasive lead, leaving the heart and the vasculature UNTOUCHED12-14.
All EMBLEM S-ICDs are by design, compatible with the EMPOWERTM Leadless Pacemaker17*.
Together, we set the pace and advance patient care
Working together, so patients have access to the optimal healthcare solutions

Changing the heart failure landscape
Our HeartLogicTM algorithm can predict potential heart failure events, with a median alert of 34 days in advance15-16.
By tracking trends, clinicians are alerted proactively ahead of an event, without having to wait for patients to report symptoms.
Designed for real life, backed by data
For almost 40 years, our technologies have been improving and changing patients’ lives

Our high voltage devices are designed with patients in mind to ensure we can provide reliable therapies, underpinned by patient safety and real-world evidence.
Real-world LATITUDETM data shows that the actual projected longevity of the EMBLEMTM S-ICD is nearly 9 years18, reducing the number of times a patient will need to undergo device replacements.
We don’t just develop technologies, we deliver meaningful impact
From personalised learning journeys to expert advice, you can access resources and support, when you need it most

Supporting you beyond clinical devices
When you use our devices, you unlock access to much more than our technology. RhythmCARE™ and EDUCARE offer educational and real time support:
- Personalised Coaching
- Remote Patient Management
- Access to insightful data and regular reports via LATITUDETM NXT
- Online medical education via EDUCARE
- Lippi G, Sanchis-Gomar F., et al. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. Int J Stroke. 2021; Feb 16:217-221.
- Groenewegen A, Rutten FH, Mosterd A, Hoes AW. Epidemiology of heart failure. Eur J Heart Fail. 2020; 22: 1342–1356.
- GBD 2017 Disease and Injury Incidence and Prevalence Collaborators . Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018; 392: 1789–1858.
- Grasner JT, Wnent J, Herlitz J, et al. Survival after out-of-hospital cardiac arrest in Europe - results of the EuReCa TWO study. Resuscitation 2020;148:218-226
- Andersen LW, Holmberg MJ, Berg KM, Donnino MW, Granfeldt A. In-hospital cardiac arrest: a review. JAMA. (2019) 321:1200–10. 10.1001/jama.2019.1696
- Kiguchi T, Okubo M, Nishiyama C, et al. Out-of-hospital cardiac arrest across the World: First report from the International Liaison Committee on Resuscitation (ILCOR). Resuscitation 2020; 152: 39–49. Article. doi: 10.1016/j.resuscitation.2020.02.044
- Product manual, RESONATE™HF, RESONATE™, RESONATE™X4, VIGILANT™, VIGILANT™X4, MOMENTUM™, MOMENTUM™ X4 - Manual 360198-002
- Product Performance report 2020 Q3 Edition (99.6% survival probability at 6 years)
- Boston Scientific ICDs and CRT-Ds with contemporary battery tehcnology have 1.9 Ah. Medtronic ICDs and CRT-Ds have 1.1 Ah. PHYSICIAN'S TECHNICAL MANUAL RESONATE™ HF ICD, RESONATE™ EL ICD, PERCIVA™ HF ICD, PERCIVA™ ICD, VIGILANT™ EL ICD, MOMENTUM™ EL ICD. Part Number: 51114084-001 EN US 2021-10-27. PHYSICIAN'S TECHNICAL MANUAL RESONATE™ HF CRT-D, RESONATE™ CRT-D, RESONATE™ X4 CRT-D, VIGILANT™ CRT-D, VIGILANT™ X4 CRT-D, MOMENTUM™ CRT-D, MOMENTUM™ X4 CRT-D. Part Number: 51113879-001 EN US 2021-10-27. Medtronic Cobalt™ XT DR ICD MRI SureScan™ Model DDPA2D4 device manual. Medtronic Cobalt™ XT VR ICD MRI SureScan™ Model DVPA2D4 device manual. Medtronic Cobalt™ XT HF Quad CRT-D MRI SureScan™ DTPA2QQ 2019-04-23 device manual
- Williams JL, Harley B, Williams G, “First Demonstration of Cardiac Resynchronization Therapy Defibrillator Service Life Exceeding Patient Survival in a Heart Failure with Reduced Ejection Fraction Cohort,” J Innov Cardiac Rhythm Manage. 2020; 11(12): 4325–4332.
- Haarbo J, Hjortshoj S, Johansen J, Jorgensen O, Nielsen J, Petersen H. Device Longevity in Cardiac Resynchronization Therapy Implantable Cardioverter Defibrillators Differs Between Manufacturers: Data from the Danish ICD Registry. Presented at HRS 2014. http://ondemand.hrsonline.org/common/presentation-detail.aspx/15/35/1241/9000. Boston Scientific = 136 patients, Medtronic = 6 51 patients, St. Jude Medical = 1,587 patients, Bitronik = 369 patients. Time to exchange of the device because of battery depletion or device failure recorded in the Danish ICD Registry was the endpoint. The four-year survival rate for devices in the Danish Registry study was 81.1% for Medtronic and 95.7% for Boston Scientific (P<0.01).
- Knops RE, Olde Nordkamp LRA, Delnoy PHM, et al. Subcutaneous or transvenous defibrillator therapy. N Engl J Med 2020;383:526–36.
- Knops RE, van der Stuijt W, Delnoy PPHM, et al. Efficacy and safety of appropriate shocks and antitachycardia pacing in transvenous and subcutaneous implantable defibrillators: analysis of all appropriate therapy in the PRAETORIAN trial
- Healey JS et al. Subcutaneous Versus Transvenous Defibrillators: The Atlas Trial. Heart Rhythm Society Late Breaking Clinical Trials LB-733 Randomized Clinical Trials. 2022
- Boehmer JP, Hariharan R, Devecchi FG, et al. A Multisensor algorithm predicts heart failure events in patients with implanted devices: results from the MultiSENSE study. JACC Heart Fail. 2017 Mar;5(3):216-25.
- Hernandez AF, Albert N, Allen L, et al. Multiple cardiac sensors for management of heart failure (MANAGE-HF) Phase I results. Abstract presented at: European Society of Heart Failure 2021 World Congress on Acute Heart Failure: June 29-July 1, 2021. Virtual.
- Data on File. CLIINICAL STUDY. The MODULAR ATP Clinical Study is designed to demonstrate the safety, performance and effectiveness of the mCRM™ Modular CRM System (EMBLEM™ S-ICD System and EMPOWER™ Modular Pacing System).
- Boston Scientific. Data on file: Data from LATITUDE™
*Disclamer for non-available technology in EMEA with no CE mark available
CAUTION
The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.