Stroke Matters. Protect the Brain
Stroke is #1 fear of patients undergoing TAVI while it is unpredictable and can be an economic burden for the healthcare system and the society.
SENTINEL Cerebral Protection System is safe and effective, with clinical data showing no major complications and the potential to reduce disabling strokes.
It only takes one piece…
The PROTECTED TAVR study
New data showed a 21% relative risk reduction in all stroke and a 60% significant relative risk reduction in disabling stroke through 72-hours

PROTECTED TAVR the biggest TAVI trial to date, enrolled 3000 patients in more than 50 sites globally.
Discover here the outcomes and more on new upcoming BHF PROTECT TAVI Study planned for 2025.

A significant milestone was recently achieved: more than 100.000 patients have been protected with Sentinel world wide.
SENTINEL is safe and fast to deploy
4 minutes median deployment time
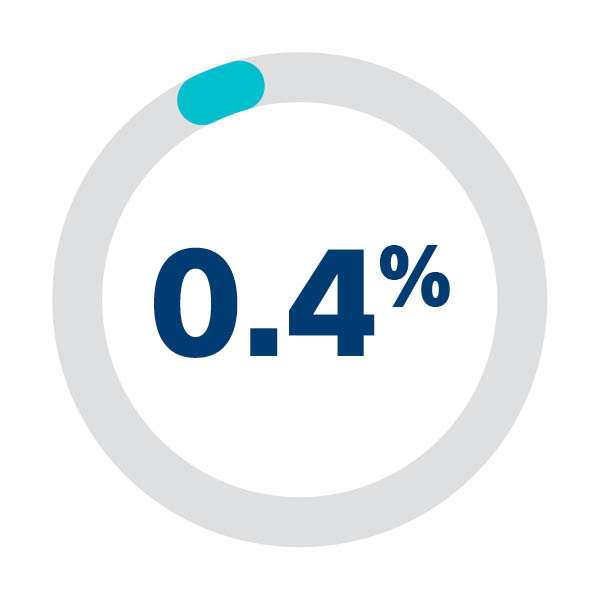
0.4% Access related vascular complications
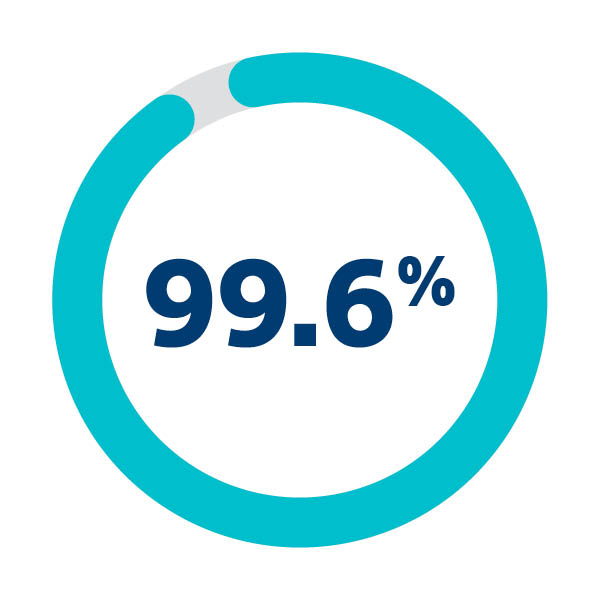
99.6% Successful deployment and retrieval
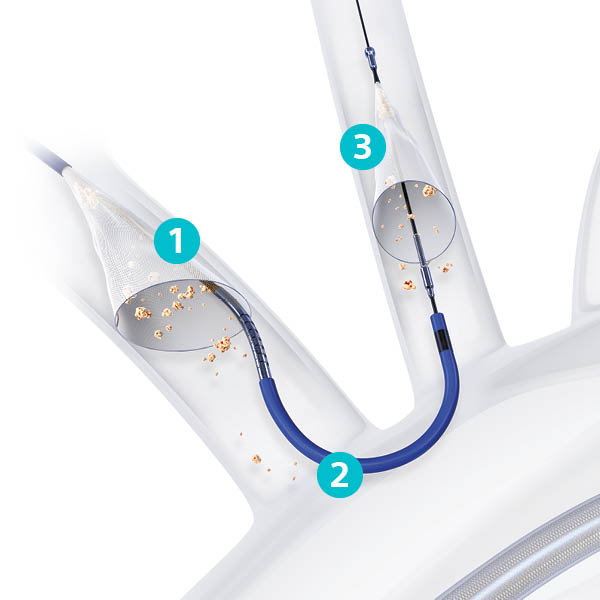
- Proximal Filter delivered to the brachiocephalic artery
- Articulating Sheath allows to accommodate wide range anatomies minimizes TAVI device interaction
- Distal Filter delivered to left common carotid
















