InTouch Newsletter Issue 9

Congresses and Events
PCR London Valves Symposia: Extending treatment options to address evolving needs in TAVI

Moving to a patient tailored TAVI: Dr Van Mieghem’s interview from PCR London Valves

Sentinel Cerebral Embolic Protection System, now Part of Boston Scientific´s TAVI Portfolio

SYNERGY™ - High-bleeding risk patients and DAPT: implementing the latest clinical evidence into real-world practice
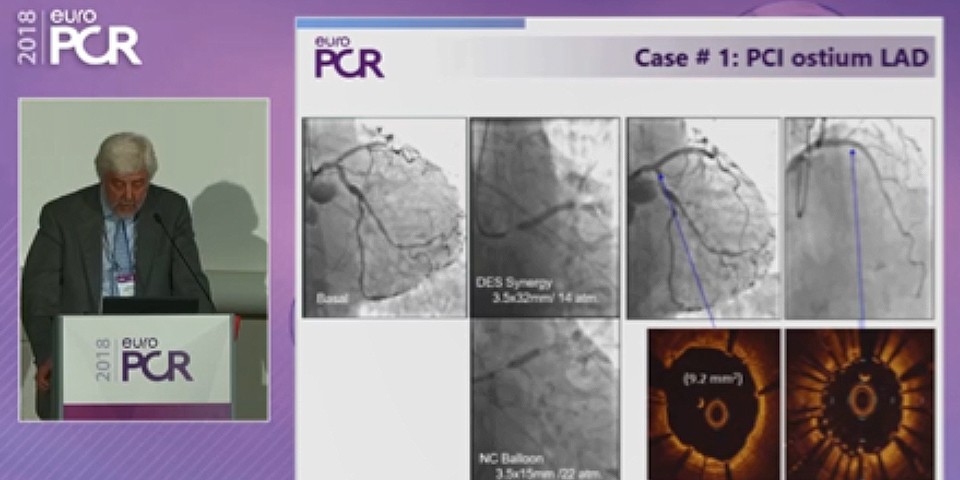
Educational Corner
PCR webinar: The new rotational atherectomy system for treating undilatable coronary lesions
Attend this PCR webinar to discover how to avoid and manage most common complications and learn more about the European Consensus Document, the "new rules" and the procedural recommendations.
WATCHMAN™ in shallow anatomies: a case with Prof Bergmann

InCathlab Live Case: Challenges of PCI and DAPT in an elderly patient with ASA allergy
SAFARI²™ - #1 TAVI Wire in Europe

As part of Boston Scientific’s TAVI portfolio, the SAFARI2™ is a pre-shaped TAVI guidewire designed for predictable, reliable performance across a broad spectrum of transcatheter devices. *,†, ‡,§,**
" SAFARI gave us a lot more forgiveness and I think it really protected the patients." Listen to Dr. Meduri, Co-Medical Director, Marcus Heart Valve Center, as he shares his experiences with SAFARI2™.
Clinical Studies
FAME 2 Study: Five-Year Outcomes with PCI Guided by Fractional Flow Reserve
EVOLVE II & SENIOR Trial Data Continues to Impress. Prof Meredith discusses the results

Bench testing performed by Boston Scientific. Bench test results may not necessarily be indicative of clinical performance. Data on file.
* Curve Retention test data with SAFARI2 Extra Small, Confida™ Brecker, Amplatz Extra Stiff™, and Amplatz Super Stiff™ Guidewires (n = 1).
† Guidewire Compression test data with SAFARI2 Extra Small, Confida Brecker, Amplatz Extra Stiff, and Amplatz Super Stiff Guidewires (n = 1).
‡ Nosecone Tracking test data with SAFARI2 Extra Small, Confida Brecker, Amplatz Extra Stiff, and Amplatz Super Stiff Guidewires (n = 1).
§ Simulated Use Static Friction, Dynamic Friction, and Distal Curve Displacement test data with Lotus™ Valve System with Safari Large and Amplatz Super Stiff Guidewires (n = 4).
** Simulated Use Friction, Dynamic Friction, and Distal Curve Displacement with Evolut® R with SAFARI2 Extra Small and Confida Brecker Guidewires (n = 3).
The Safari™ Guidewire is manufactured by Lake Region Medical and distributed by Boston Scientific Corporation.
All cited trademarks are the property of their respective owners. CAUTION: Federal (U.S.) law restricts this device to sale by or on the order of a physician.














