InTouch Newsletter Issue 5

Congresses and Events
The SYNERGY™ Stent: complete revascularization solutions in complex PCI

In this Symposium from EuroPCR 2017, a panel of experts discuss about the recommended requirement for use of DES in clinical practice and the potential benefits of bioabsorbable polymer DES in high-bleeding risk patients. Watch the webcast and learn how the SYNERGY™ stent is being used in complex patient subsets.
LOTUS™ Valve: expanding treatment options in real-world practice

LOTUS™ Valve expands treatment options in real-world practice. From EuroPCR 2017, a webcast dedicated to the LOTUS™ program, reviewing a range topics from our trial data to some really good clinical examples.
Boston Scientific has closed Symetis Acquisition
Boston Scientific Corporation has announced the close of its acquisition for $435 million of Symetis SA. With the completion of the acquisition, the Company will immediately begin selling the ACURATE TA™ and ACURATE neo™/TF™ valve systems, expanding its Structural Heart Portfolio and increasing the Offerings for the treatment of Patients with Valvular Heart Disease.
Educational Corner
PCR Edu Online: How to treat patients with undilatable/calcified coronary artery lesions

An educational webcast from PCR Edu Online to learn how to treat patients with undilatable and calcified coronary artery lesions using the Rotational Atherectomy System. Watch the video presentation and join the course.
InCathlab Live Case: a safe plaque modification at Bournemouth Hospital
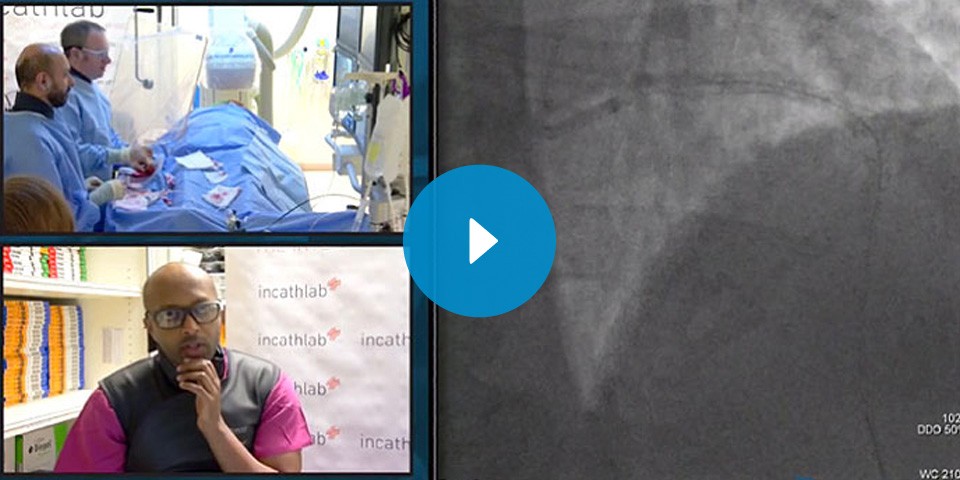
A Rotational Atherectomy Live Case to master a safe plaque modification with a step-by-step procedure: from setting up the system to practical procedural recommendations. Watch the video and compare techniques and approach strategies of different experts.
Learning with clinical case: Master plaque modification to treat diffused calcified lesions with IVUS support
Clinical Studies
New data from the EWOLUTION 1 year registry: WATCHMAN™ LAAC Device is effective in reducing the risk of stroke

One-year data from the EWOLUTION registry confirmed that the WATCHMAN™ LAA Closure device had a high implant success rate and was effective in stroke reduction for patients with nonvalvular atrial fibrillation. It also found that LAAC with WATCHMAN™ followed by dual antiplatelet therapy significantly reduced the risk of stroke and lowered the risk of major bleeding by more than half as compared to Warfarin use.
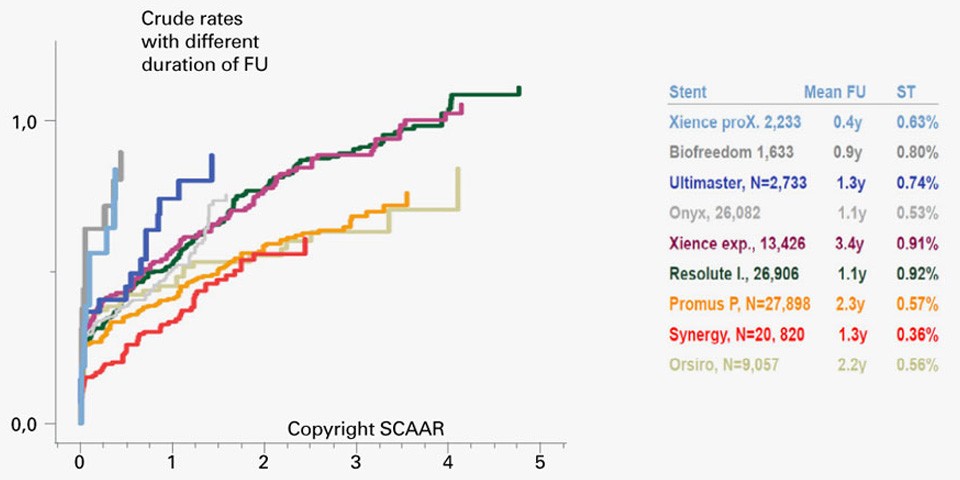
SCAAR Registry from EuroPCR 2017: SYNERGY™ Stents presents very low definite ST rates
Latest updates from EuroPCR 2017: the EVOLVE II Diabetes Substudy
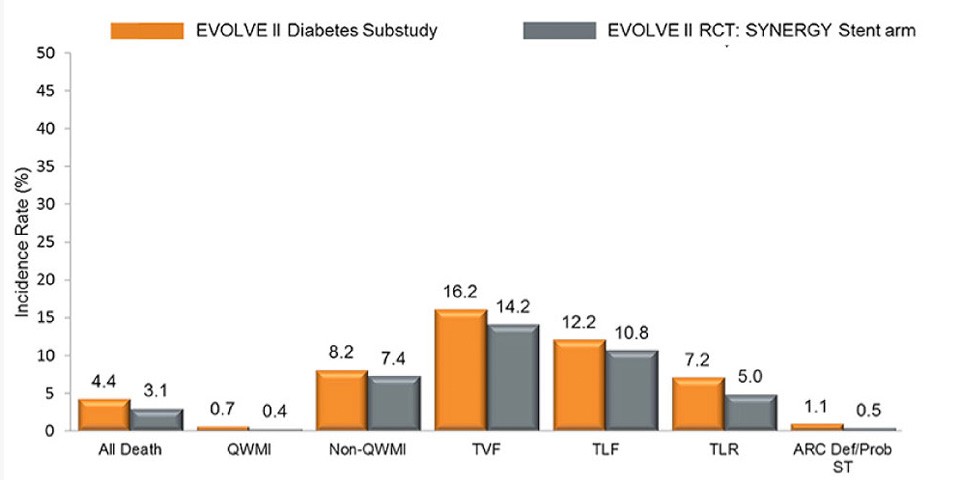
EVOLVE II Diabetes Substudy is a consecutive, multicenter, single-arm, non-randomized study with the SYNERGY™ Stent in medically-treated diabetic patients. SYNERGY™ BP Stent showed excellent performance as compared to all patients in the EVOLVE II Trial at three years:
- 1.1% def/Prob Stent Thrombosis at 3 years
- Patients with DM treated with the Synergy™ BP Stent had 0% definite/probable ST after 30 days
LOTUS™ Valve: REPRISE III 1 Year data from EuroPCR 2017

LOTUS™ Valve showed superiority to CoreValve® TAVI System Platform in the primary effectiveness endpoint at 1 year and non-inferiority in the primary safety endpoint at 30 days. This is the result of the REPRISE III 1 year data trial. Watch the REPRISE III data highlights animation and read the full deck presented by Dr. T. Feldman at EuroPCR 2017.