Further Evidence for Early Healing with the SYNERGY™ Stent
How does SYNERGY compare with other stents regarding endothelial cell coverage, endothelial cell function, and thrombogenicity?
% Endothelial Cell (EC) Coverage at 21 Days in Cell Assay
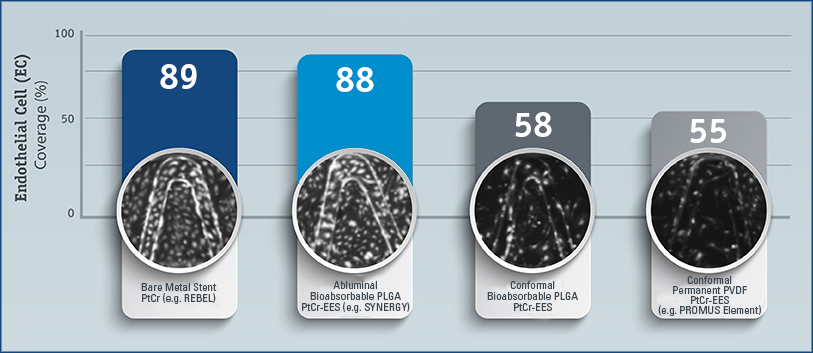
Endothelial Cell (EC) Function
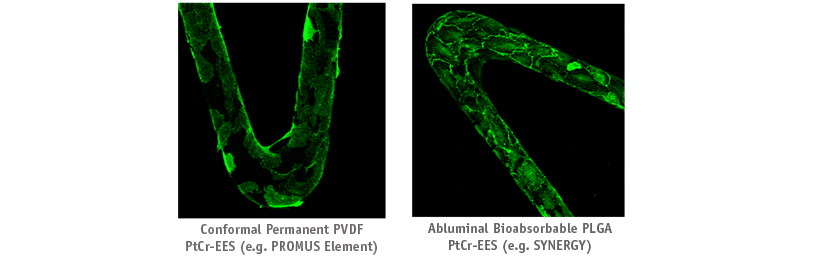
In a pre-clinical evaluation, SYNERGY displays greater localization of VE-Cadherin at cell junctions indicating improved EC function. Green stain indicates functional endothelial cells.
















