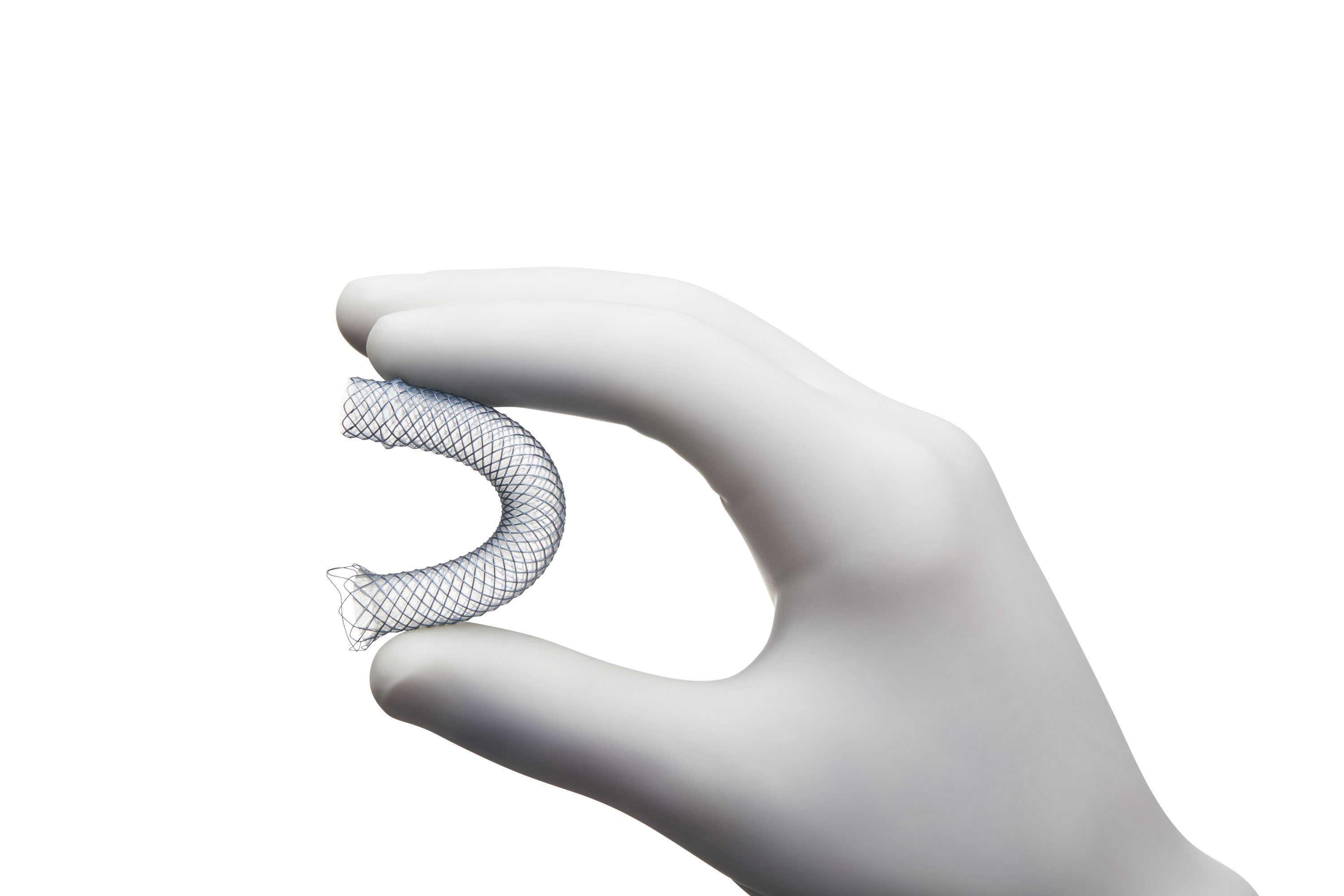
*Compared to WallFlex Biliary RXFully Covered Stents.
Indications for use:
WallFlex Biliary RX Stent System, WallFlex Biliary PLUS RX Stent System: indicated for use in the palliative treatment of biliary strictures produced by malignant neoplasms and relief of malignant biliary obstruction prior to surgery.
WallFlex Biliary RX Stent System RMV: indicated for use palliative treatment of biliary strictures produced by malignant neoplasms, relief of malignant biliary obstruction prior to surgery and for treatment of benign biliary strictures.
Warning: The safety and effectiveness of this device for use in vascular system has not been established.
Sources:
1. The conformability of the WallFlex Biliary PLUS RX Fully Covered Stent System was assessed through axial force measurements in DV bench tests conducted by Boston Scientific, demonstrating that its axial force is lower than that of other self-expandable metal stents, as referenced in the literature by Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49. Data on file. Bench testing is not indicative of clinical performance.
2. The radial force of the WallFlex Biliary PLUS RX Fully Covered Stent System was assessed through radial force measurements in DV bench tests conducted by Boston Scientific, demonstrating that its radial force exceeds the threshold set by other self-expandable metal stents, as defined in the literature by Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49. Data on file. Bench testing is not indicative of clinical performance.
3. Soderlund C, Linder S. Covered metal versus plastic stents for malignant common bile duct stenosis: a prospective, randomized, controlled trial. Gastrointest Endosc. 2006;63(7):986-995.
4. Moss AC, Morris E, Mac Mathuna P. Palliative biliary stents for obstructing pancreatic carcinoma. Cochrane Database Syst Rev. 2006;(1):CD004200. Published 2006 Jan 25.
5. Isayama H, Nakai Y, Hamada T, Yamamoto N, Koike K. Development of an ideal self-expandable metallic stent design. Gastrointestinal Intervention. 2015 ;4 :46–49.
6. Yokota Y, Fukasawa M, Takano S, et al. Partially covered metal stents have longer patency than uncovered and fully covered metal stents in the management of distal malignant biliary obstruction: A retrospective study. BMC Gastroenterol. 2017;17:105.
CAUTION:The law restricts these devices to sale by or on the order of a physician. Indications, contraindications, warnings, and instructions for use can be found in the product labelling supplied with each device or at www.IFU-BSCI.com. Products shown for INFORMATION purposes only and may not be approved or for sale in certain countries. This material not intended for use in France.
All images owned by Boston Scientific. All trademarks are the property of their respective owners.