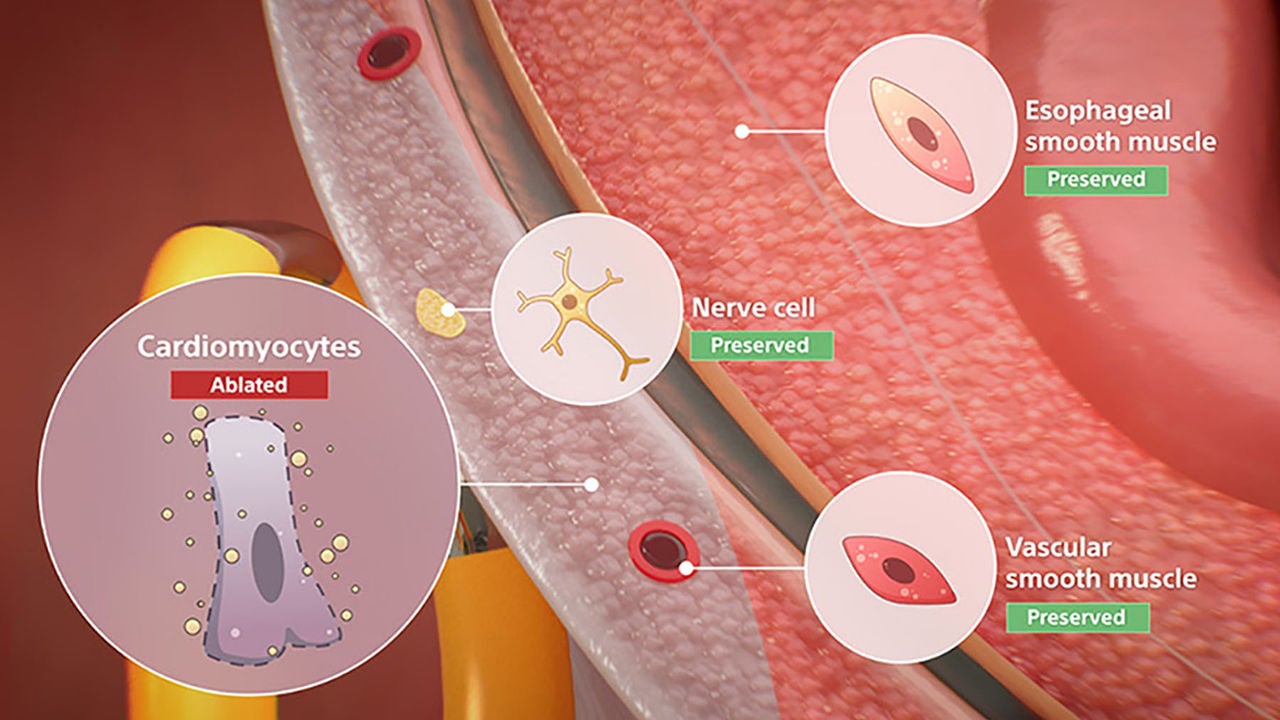
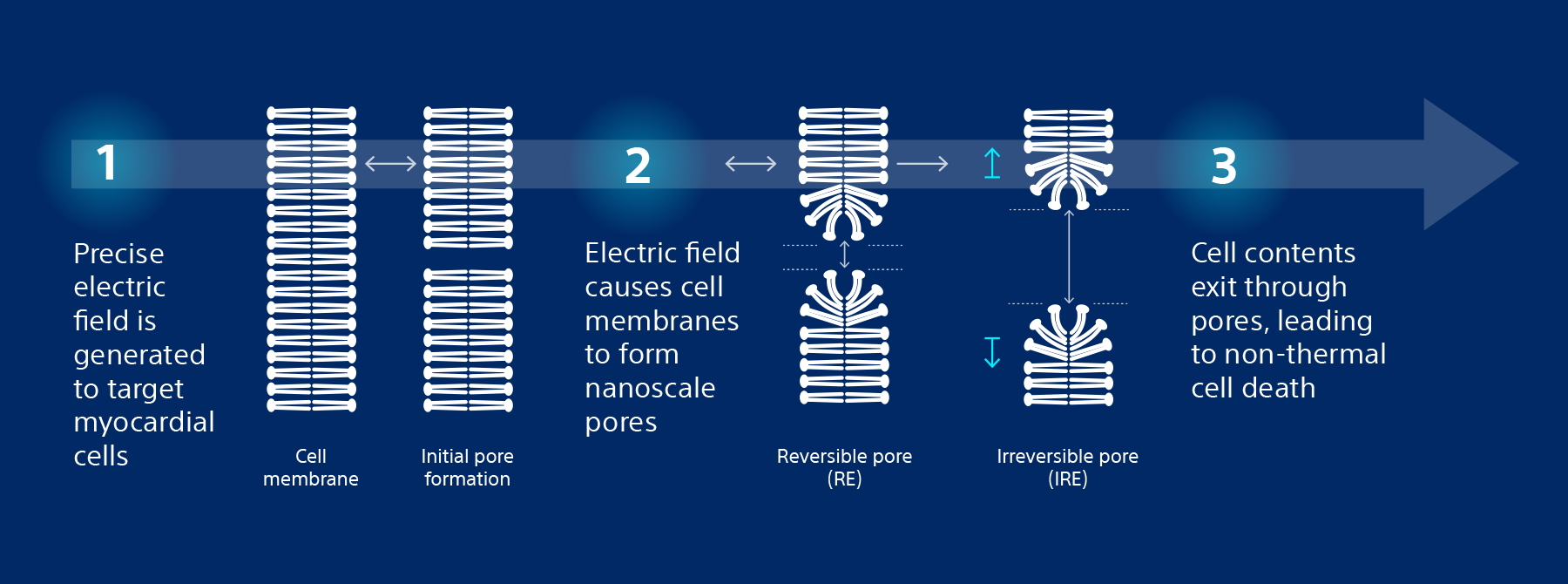
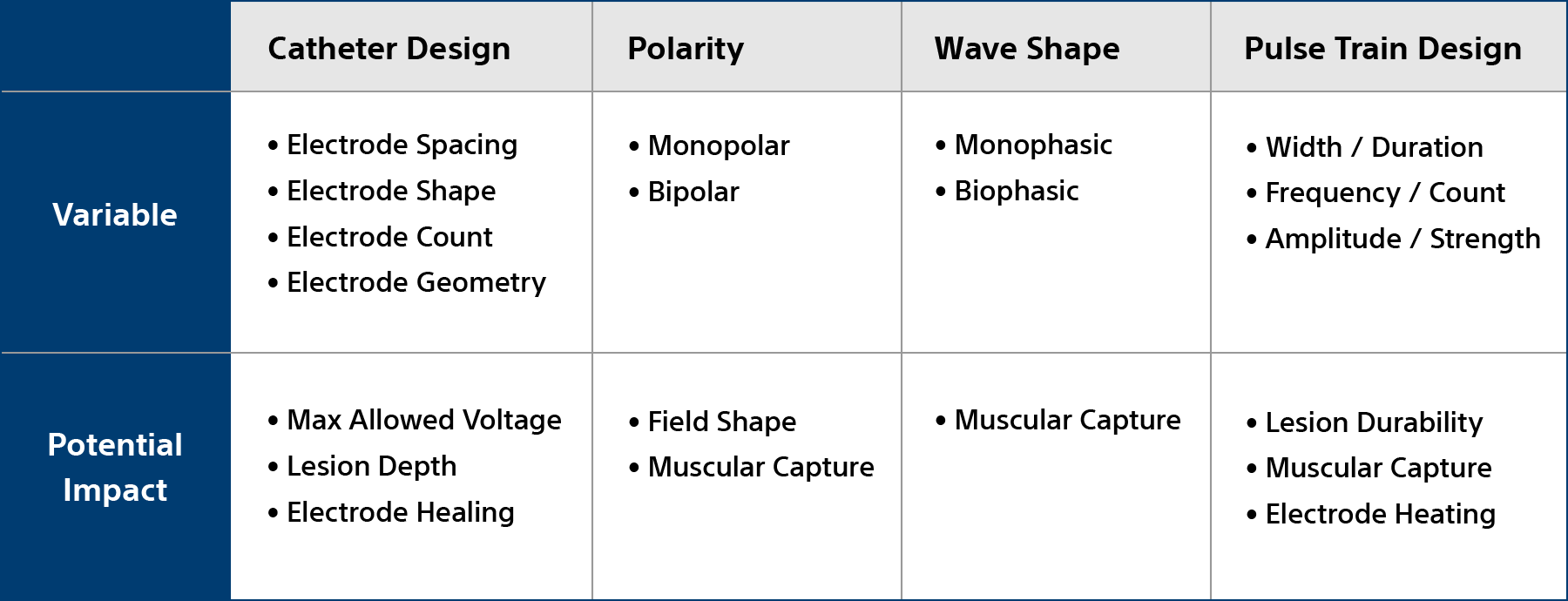
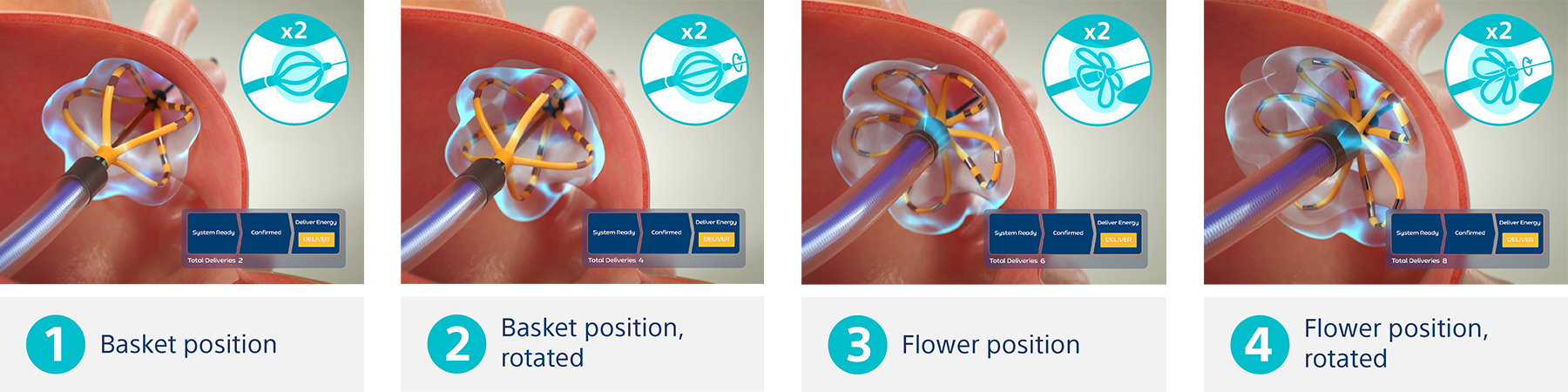
Non-thermal mechanism of action for cardiac ablation
Unlike thermal cardiac ablation methods, pulsed field ablation (PFA) therapy uses short bursts of electrical pulses to treat atrial fibrillation (AFib). The goal of PFA therapy is to isolate and target specific characteristics of cardiac tissue for irreversible electroporation (IRE), inducing cell death and durable lesions using a non-thermal energy source.